Abstract
Research on endurance locomotion has mainly focused on elite athletes rather than common middle-aged subjects. Our report describes the physiological and hematological adaptation of a healthy, active 62-year-old man who trekked alone along a 1300 km/3 month course of Alpine paths (Via Alpina). The following procedures were conducted: pre- and post-trekking and fortnightly field anthropometry (total and lean body mass), functional tests (isometric maximal voluntary force, spontaneous walking speed, relative metabolic cost, and peak oxygen consumption) and clinical chemistry/hematological measurements with laboratory instruments; daily self-administered effort measurements using portable devices along the route (walked distance, ascent, descent, time, metabolic consumption, and cost). Despite the tough trekking route, the subject completed the trek without any worsening of his performance, or any significant health or functional problems. In addition, his peak oxygen consumption increased by 13.2%. His successful adaptation may be attributed to his constant, repeated middle-intensity and extensive exercise and lengthy exposure to high altitude. The clinical chemistry/hematological measurements documented his physiological adaptation. In conclusion, we show how an active, middle-aged man can successfully face endurance trekking, not only without any harm to his health or functions but also with an increase in his capacity to support specific effort.
Introduction
Both endurance walking and running are used in basic life functions, such as retrieving food, colonizing new habitats, mating, and escaping hazards. 1 −3 Walking and running have been included in modern exercise recommendations by several authoritative guidelines. 4 It is widely acknowledged that the study of extremes helps us understand general biological phenomena. Extreme conditions, including endurance performances, refer to long-distance racing, 1 ,5−10 challenging environmental conditions (eg, desert, mountain, and polar expeditions 1 ,5,8,9,11), and strenuous physical activity (eg, man-hauling 1 ,8,11). Investigations into endurance methods in extreme conditions have included walking,1,8 running, 1 ,5,7−9 swimming, 9 cycling, 1 ,6,7,12 kayaking, 9 and multi-discipline races. 9 The research into extreme endurance locomotion has involved both able-bodied and disabled subjects. 6
Traditionally, these studies tend to enlist top athletes. However, studying the physiological characteristics of amateurs may reveal traits which may benefit nonelite athletes. The aim of this investigation is to describe the physiological changes that were recorded in a 62-year-old man who trekked a very long distance (> 1300 km) following the mountain path route that is known as the Via Alpina. 13
Methods
The subject
The subject is an Italian male, aged 62, with a body mass of 75 kg, height 178.5 cm, and BMI of 23.5 (pre-trekking measurements). He is retired and physically active, mainly performing downhill skiing, open water (sea) swimming, and trekking. Testing procedures were explained to the subject and he gave informed consent. While trekking, he did not change his already healthy eating habits, but rather adjusted his calorie intake to match his increased caloric demands.
Equipment
The subject carried all his equipment in a rucksack on the route depicted in the Figure. Besides the necessary amount of water and food for each daily or multi-day stage (depending on each arrival venue, mostly lodges or villages), he carried clothing, a few personal items (eg, for personal hygiene), a tent, a sleeping bag, a mobile phone, a laptop with an internet key, and the portable measuring devices (see PROCEDURES) with all their accessories. The rucksack mass varied from 14 to 29 kg and its exact weight was always taken into account for making the specific calculations. The subject covered the route with the Nordic Walking gait, a worldwide method of walking aided by thrusts from hand-held poles that are similar to cross-country skiing poles. 14
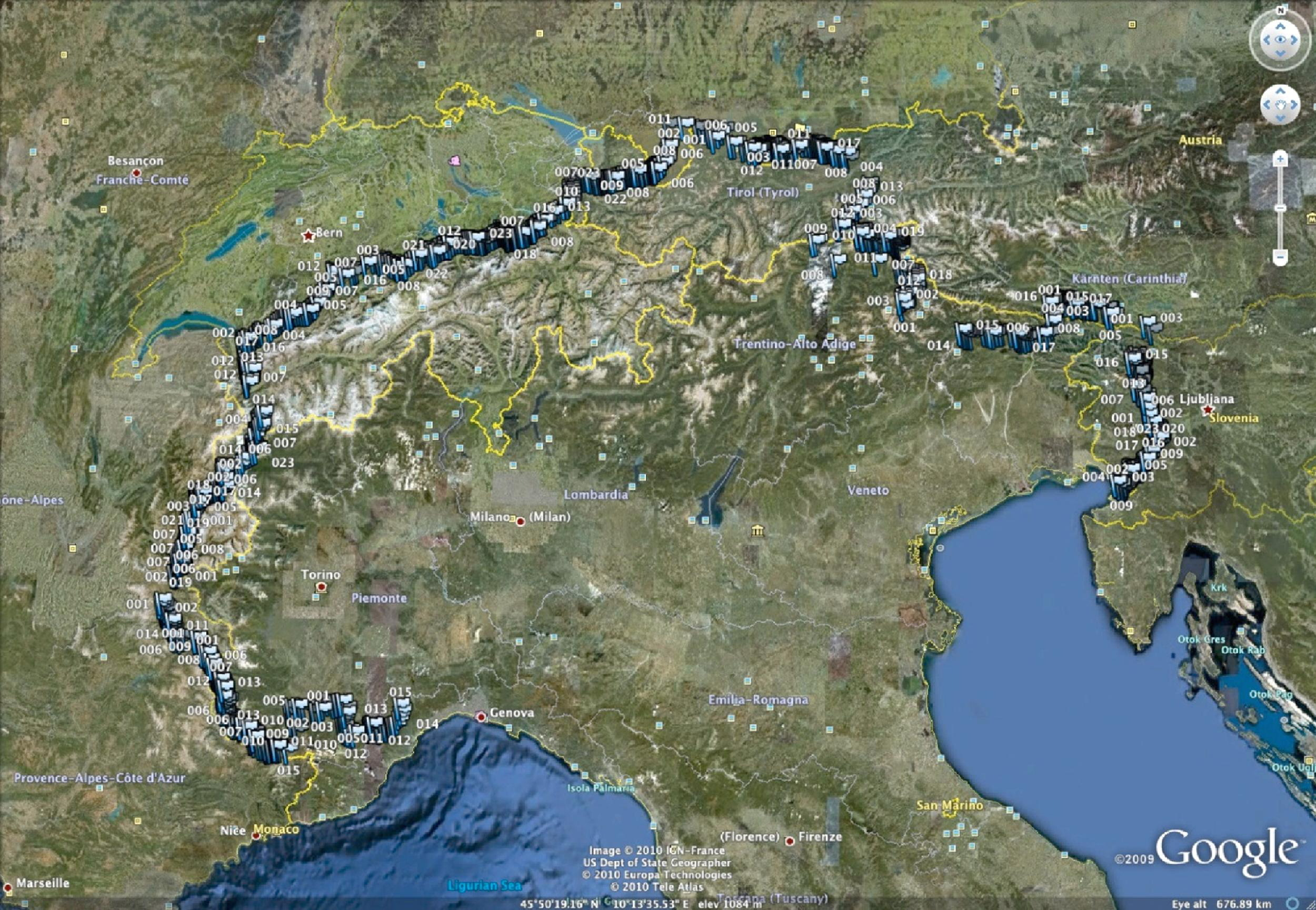
Trekking course as marked waypoints (light blue flags; image saved with Google Earth 5.1.3533.1731).
Procedures
Three sets of measurement procedures were conducted to follow the subject's progressive effort and the effects on his health and performance: Pre- and post-trekking laboratory measurements: anthropometry (total and lean body mass), force (knee extensor and elbow flexor isometric maximal voluntary contractions), spontaneous walking speed and relative metabolic cost, peak oxygen consumption, a broad panel of clinical chemistry (Modular System, Roche Diagnostics GmbH, Mannheim, Germany), and hematological tests (ADVIA 2120TM, Bayer Diagnostics, Newbury, UK), as described in Tables 1 and 2. Pre-trekking laboratory measurements were taken 5 days before departure and post-trekking measurements were taken 10 days after arrival. Approximate fortnightly field measurements: same as the pre- and post-trekking measurements except for peak oxygen consumption (the last field measurement was performed 2 days after completing the route). Daily field self-administered measurements including metabolic energy consumption and metabolic cost of transport in addition to walked distance, ascent, descent, and time.
Complete results of laboratory and field physiological testing
MVF maximal voluntary force; SWS spontaneous walking speed.
missing values due to shoulder pain from a past accident.
Complete results of clinical chemistry and hematological testing
Laboratory and field measurements
Body mass was measured using an electronic scale. In order to estimate the lean mass, a total of 7 skin folds (chest, midaxillary, triceps, subscapular, abdomen, suprailiac, and thigh) were measured with a skin fold caliper (Gima, Gessate, Italy) on the right side of the body.
15
All measurements were taken by the same trained examiner. An average of 2 tests was used for analysis and, when the measurements differed by more than 1.0 mm, a third measurement was taken and the updated mean value was used. The isometric maximal voluntary force from the knee extensors (both legs together) and elbow flexors (both arms separately) was measured using a load cell mounted on a customized chair (546 QDT, DS EUROPE, Milan, Italy). The load cell was calibrated with known weights before each testing session and all the measurements were taken at the same time of the day. Pre- and post-trekking laboratory measurements of spontaneous walking speed and relative metabolic cost were taken on a standard athletic track (400 m, outdoor, all-weather), while field measurements were taken at each location on a flat and even course. All walking tests were conducted under acceptable environmental conditions (atmospheric pressure average ± SD: 718 ± 40 mmHg; temperature: 25 ± 4 °C; humidity: 42 ± 14 %). Field measurements were taken twice: without rucksack and, after a proper rest, with rucksack. The metabolic cost was measured as the difference between walking oxygen consumption and resting (standing) oxygen consumption, divided by walking speed.
16
Oxygen consumptions in ml O2 kg−1 min−1 were converted into energy units J kg−1 min−1 according to the measured respiratory quotients.
16
Oxygen consumption was measured using a portable breath-by-breath metabograph (K4b
2
, Cosmed, Rome, Italy) and the walking speed was measured using a small GPS receiver (Edge 305, Garmin, Olathe, USA). Peak oxygen consumption was measured during a 20-minute incremental walking protocol at a constant speed (1.4 m s−1) while slope increased from 0 to 21%, step 3%. The man walked on a motorized treadmill (Saturn 300/100r, h/p/cosmos, Nussdorf-Traunstein, Germany) and his oxygen consumption was measured using the aforesaid metabograph. Fortnightly tests were always conducted after waking (7:00
Self-administered measurements
The subject wore 2 portable devices when walking: an upper arm device for measuring effort-related variables (Armband: BodyMedia, Pittsburgh, USA17,18), and a pocket GPS receiver (Edge 305, Garmin, Olathe, USA). He downloaded recordings daily into a laptop and emailed them to the University lab by an internet key. The armband allowed measuring metabolic energy consumption, using different physical and physiological sensors, which was fed to a proprietary algorithm together with the whole mass input (total body + rucksack mass). Oxygen consumption was estimated by multiplying the armband measurements in MET by 3.5 ml O2. Metabolic cost (J per kg body mass per m traveled distance) was calculated assuming that 1 ml O2 is equivalent to 21 J. The GPS receiver allowed measuring the walked distance, ascent, descent, and time. The subject frequently marked his position (latitude, longitude, and altitude, together with the position-marking time, “waypoint”) to provide the data for a 3D calculation of distance, ascent, descent, and speed. The GPS 2D distances were calculated using the Haversine formula 19 and the 3D distances by applying Pythagorean theorem.
Results
Laboratory and field tests
Total and lean body mass decreased by 6.8 kg (−9.1%) and 1.7 kg (−1.7%) from pre-trekking to trekking on day 53 (pre-trekking value vs average of values from day 53 until post-trekking), respectively. Likewise, BMI and % fat decreased from 23.5 to 21.4 and from 20.3 to 14.9%, respectively. From day 53 until post-trekking, these anthropometric variables remained constant. Knee extensor and elbow flexor isometric maximal voluntary forces remained relatively constant over time, showing average values of 1030.6, 364.1, and 282.6 N for both knees, left elbow, and right elbow, respectively (Table 1). The walking tests showed constant values of spontaneous speed and relative metabolic cost over time: on average, 1.71 m s−1 and 3.15 J kg m−1 without rucksack, and 1.65 m s−1 and 4.31 J kg m−1 with rucksack (Table 1). The subject's total (walking and rest days summed) metabolic energy expenditure was 1 592 175 kJ, while his daily average metabolic power was 17 306 kJ d−1. Peak oxygen consumption per body mass unit increased from 40.97 to 50.43 ml O2 kg−1 min−1 (+23.1%) and, in absolute values, from 3.07 to 3.48 l O2 min−1 (+13.2%). The results of clinical chemistry and hematological testing are shown in Table 2.
Self-administered measurements
The subject walked 1312.7 km in 68 days (19.3 km d−1; range: 5.5–41.0 km d−1) with 24 days of rest for a total of 92 days trekking (see Figure). The total ascent and descent were 62 480 m (934 m d−1; range: 0–2526 m d−1) and 61 659 m (910 m d−1; range: 0–2647 m d−1), respectively. The total walking time was 30 033 min, (range: 1 hour and 17 minutes–12 hours and 46 minutes; mean 7 hours and 42 minutes per day), and average speed was 0.71 m s−1 (range: .29−1.38 m s−1). The subject showed an average oxygen consumption of 18.89 ml O2 kg−1 min−1 (46.1% of pre-trekking peak value) and a metabolic cost of 10.35 J kg m−1. The daily average oxygen consumption and metabolic cost did not change much over time (Consumption [ml O2 kg−1 min−1] = −.03 Trekking day # + 20.16; average ± SD 18.89 ± 2.51; and Cost [J kg−1 m−1] = −.01 Trekking day # + 10.70; average ± SD 10.35 ± 3.82). Due to a technical problem, the waypoint marking times were not recorded on 3 specific walking days (along an estimated total distance of 64.7 km) and, consequently, the respective partial consumptions and average daily costs could not be calculated.
Discussion
The subject walked along a mountainous course of about 1300 km and was able to record anticipated measurements himself. His total and daily metabolic energy expenditures were similar to other human record endurance performances.1,8
Metabolic expenditure and physical activity
Functional results such as knee extensor and elbow flexor isometric maximal voluntary forces, spontaneous walking speed, and relative metabolic cost remained quite constant over successive trekking days. The dispersion of the measured forces was within the specific intrinsic measurement variability. Apparently, prolonged trekking did not seem to be an effective conditioning stimulus for these variables. Conversely, the peak oxygen consumption increase was striking. This may be attributed to: 1) constantly repeated middle-intensity extensive exercise; and 2) lengthy exposure to altitude (with several overnight stays in lodges at over 2000 m).
Anthropometry, biochemistry, and hematology
Total and lean body mass and biochemical and hematological values did not deteriorate substantially or approach alert or critical values. The decrease in both total and lean body mass until day 53 (from May 31st until July 22nd) might have been caused by other environmental difficulties that he had to face—for example, heavy snow. An increase was observed in high-density lipoprotein cholesterol (from 52 to 90 mg dL−1) accompanied by a substantial decrease in triglycerides (from 173 to 48 mg dL−1) during the trek (all rapidly reversed at the end of the trekking days). High-density lipoprotein increase is a well-known effect of physical exercise; training programs that elicit 1200 to 2200 kcal wk−1 can increase high-density lipoprotein cholesterol 8 mg dL−1). 20 A very similar sharp decrease had been previously observed after exercise cessation; Dressendorfer et al 21 reported that HDL cholesterol levels increased by 18% in 12 male marathon runners after one week of running, but a 3-day rest period acutely reversed the change.
Finally, the increase observed in C-reactive protein (up to 15.6 g L−1) is likely attributable to the distance-related acute phase response to aerobic exercise. Similar results were earlier described in long-distance runners by Strachan et al, 22 who observed an increase of C-reactive protein concentration, reaching a median of 27 mg L−1.
In conclusion, we have described how a mature adult successfully faced an extreme long-distance trek with a significant improvement of aerobic capacity and an ability to support extensive endurance effort. Further research is required to generalize the results to the overall population.
Financial disclosures
Armband was provided by Sensormedics Italia Srl, Milan, Italy; GPS receiver provided by Garmin Italia, Rozzano, Italy. The Faculty of Motor and Sport Science, Verona University, provided partial economic support for the subject's trekking.
Footnotes
Acknowledgments
The authors thank the subject for his trekking, as well as for his work as a self-operator; the Faculty of Motor and Sport Science, Verona University, for their partial economic support for his trekking; Mrs Chiara Begnini for her assistance with data collection and analysis; Carlo Capelli, MD, and Antonio Cevese, MD, for their assistance with the project planning and data collection (Mrs. Begnini, Dr Capelli and Dr Cevese from the Faculty of Motor and Sport Science, Department of Neurological, Neuropsychological, Morphological and Movement Sciences, Verona University); Antonio Cevese for his English language reviewing; Gian Cesare Guidi, MD, chairman of the Board for the Department of Morphological-Biomedical Sciences, Faculty of Medicine and Surgery, Academic Hospital of Verona, for his support for clinical chemistry and hematological tests.
