Abstract
Objective
The current study evaluated multiple metabolic and inflammatory hormone responses in recreational climbers (7 men and 3 women, age 26–49 years) over 9 days. In particular, acylation-stimulating protein (ASP), which influences fat storage in adipose tissue, has not been measured at high altitude.
Methods
Serial measurements were taken at sea level (SL), or 353 m, on day 0, 4000 m on day 3, 4750 m on day 6, and 5300 m on day 9 of the expedition.
Results
Body mass index (BMI) decreased upon ascent to 5300 m from SL (SL 23.2 ± 1.5 kg/m2; 4000 m 23.2 ± 1.4 kg/m2; 4750 m 22.9 ± 1.3 kg/m2; 5300 m 22.3 ± 1.2 kg/m2; P < .001). Similarly, plasma non-esterified fatty acids and triglycerides increased, while HDL cholesterol decreased (P < .05 to < .001) from SL to 5300 m. Acylation-stimulating protein (SL 42.2 ± 40.2 nm; 4000 m 117.0 ± 69.6 nm; 4750 m 107.9 ± 44.5 nm; 5300 m 82.2 ± 20.2 nm; P = .019) and adiponectin (SL 10.4 ± 6.5 ng/mL, 4000 m 13.9 ± 8.5 ng/mL, 4750 m 18.3 ± 8.3 ng/mL, 5300 m 14.7 ± 8.0 ng/mL; P = .015) increased, as did insulin and Interleukin-6 (IL-6) levels (up to 71% and 168%, respectively; P < .05) with no change in leptin, complement C3 (C3), high sensitivity C-reactive protein (hsCRP) or cortisol levels throughout the mountain ascent from SL to 5300 m.
Conclusion
Acylation-stimulating protein and adiponectin are increased during a 9-day period of high altitude (SL to 5300 m) exposure despite weight loss in healthy mountaineers.
Introduction
It has been reported that individuals exposed to altitudes of 5000 m or more above sea level (SL) experience decreased hunger, decreased food intake, and weight loss.1,2 Increased energy expenditure associated with strenuous physical activity from mountaineering, increased exposure to cold and inclement weather, and decreased oxygen saturation are a few factors that could explain the loss of weight and appetite experienced by high altitude climbers. 1 There are, however, conflicting reports regarding the physiological/hormonal responses behind weight loss at high altitude. The changes, if any, of metabolic-related hormones such as ghrelin and leptin at high altitude have been controversial. 3 These discrepancies may be explained by the large variations in the methodology employed to study human physiology at high altitude. For instance, exposure to hypoxia (altitude-induced vs hypobarometric chamber), duration and elevation of stay, and population (experienced vs recreational climbers) are all factors that must be taken into account when comparing studies. Nonetheless, recreationally fit individuals climbing to altitudes greater than 4000 m under their own power for an acute period of time (less than 14 days) generally experienced decreased total caloric intake as well as decreased body mass index (BMI).4,5
A decrease in calorie intake and, consequently, weight loss may be mediated by the release of adipose tissue hormones. 6 Leptin, a classic adipokine produced almost entirely by adipose tissue, regulates satiety and is closely correlated to BMI. 7 Likewise, adiponectin is an adipose tissue-secreted hormone known to play a role in fatty acid oxidation and is strongly and inversely associated to adipose tissue mass. 7 Finally, acylation-stimulating protein (ASP) may also play a role in mediating weight loss. It is produced by adipose tissue and regulates storage of triglycerides in this tissue. In addition, systemic concentrations of ASP may be directly regulated by other cytokines and inflammatory markers such as high-sensitivity C-reactive protein (hsCRP), adiponectin, and interleukin-6 (IL-6) and vice versa. 8 –10 Plasma IL-6 has been reported to increase during brief bouts of exercise (0.5–2.5 hours) and is inversely related to exercise intensity independently of lipid, glucose, and insulin concentrations.
It has been hypothesized that leptin could be responsible for high altitude anorexia and weight loss, yet there is little data to support this; leptin does not appear to change in response to increasing altitude. 11 Cortisol, similar to leptin, does not consistently change in all studies at high altitude.3,12 We hypothesized that ASP may change with increasing altitude due to its role in regulating dietary triglyceride uptake, its release from adipose tissue, and its association to inflammatory markers which could impart satiety. 13
Therefore, based on published literature showing weight loss and increased satiety at high altitude, as well as the important role that adipokines play in weight loss, satiety, and exercise, assessment of adipokines would provide insight into the physiological processes that occur at high altitude. Adipokines and inflammatory factors were measured in recreationally fit individuals climbing to an altitude 5300 m under their own power over a period of 9 days.
Materials and Methods
Climbing and Data Collection Protocol
A group of 12 healthy individuals (8 men and 4 women, age range 26–49 years) were invited to participate in a scientific expedition in the heart of the Andes Mountain. Two (one man and one woman) did not complete the ascent and were eliminated from the analysis. The objective of the expedition was to reach the summit of the Huyana Potosi in Bolivia. Prior to departure and during the ascent, serial clinical and laboratory assessments were to be completed at sea level (SL; 353 m) on day 0, upon arrival to La Paz (4000 m, Bolivia) on day 3, at base camp (4750 m, Huyana Potosi) on day 6, and at high camp (5300 m, Huyana Potosi) on day 9. The climbers carried their personal belongings (restricted to approximately 35% of their body weight), which included clothing, tents, cookware, and climbing gear. In addition, all subjects were provided with detailed instructions prior to the start of the study to remain well hydrated, and avoid caffeine and alcohol during the duration of the expedition. A local guide and porters were hired to ensure the safety of the members of the expedition and to carry scientific material as well as food and water.
Subjects
All subjects met the following predetermined criteria: age range 18 to 55 years, no significant medical condition including diabetes and hypertension, no recent exposure to high altitude, and not involved in professional climbing/mountaineering activities. The study protocol was approved by the CRIUCPQ (Centre de Recherche de l'Institut de Cardiologie et Pneumologie de Québec) associated with Laval University. All subjects signed informed consent prior to engaging in the expedition.
Measurements of Body Composition
Changes in body mass were measured with a Tanita bioelectric impedence analyser (BIA) utilizing the foot-to-foot contact electrode method (Tanita TBF-300A Body Composition Analyzer/Scale, IL, USA). Measurements were taken at the same time as blood samples were drawn at SL (353 m, day 0), 4000 m (day 3), 4750 m (day 6), and 5300 m (day 9). Measurements were taken following the manufacturer's instructions and equations. Only data for total body weight are reported. Subject height was measured at SL. Body Mass Index was calculated as follows: weight (kilogram) / height2 (meter2).
Blood Sampling
Blood samples were drawn from the antecubital vein between 7 and 9
Plasma Analyses
Plasma glucose, insulin, triglyceride (TG), total cholesterol (TC), and high-density lipoprotein cholesterol (HDL-C) were measured in the hospital clinical biochemistry laboratory (Hôpital Laval, Quebec) according to validated clinical procedures. Low-density lipoprotein (LDL) cholesterol was calculated using the Friedewald formula. 14 Measurement of ASP, adiponectin, hsCRP, C3, IL-6, leptin, and non-esterified fatty acids (NEFA) was performed in the research laboratory. Adiponectin (intra-assay variation 3.9%, inter-assay variation 8.5%) and leptin (intra-assay variation 3.2%, inter-assay variation 7.8%) were measured using commercial radioimmunological assays according to the manufacturers' protocol (Millipore, MA, USA). Interleukin-6 (intra-assay variation 7.4%, inter-assay variation 7.8%) and hsCRP (intra-assay variation 4.6%, inter-assay variation 6.0%) were measured using commercially available enzyme-linked immunosorbent assays (ELISAs) according to manufacturer's protocol (Millipore, MA, USA). Acylation-stimulating protein was measured using an in-house ELISA method following methodology previously reported with the following modifications: standards (including the blank), samples and controls were diluted in a 1% w:v of bovine serum albumin (BSA): phosphate buffered saline (PBS) instead of PBS alone15,16 (intra-assay variation <4%, inter-assay variation <8%). Plasma NEFA was determined by colorimetric enzymatic assay (Wako Diagnostics, Richmond, VA). Plasma C3 concentration was determined by turbidimetric assay using a polyclonal anti-human antibody specific against C3 (inter-assay coefficient of variation was <4% and inter-assay CV was <8%; Wako Diagnostics, Richmond, VA).
Statistical Analysis
Results are presented in the Tables as mean ± standard deviation (SD) and in the figures as median, 25th and 75th percentile, and minimum and maximum values, as indicated in the figure legends. Statistical analyses and graphics preparation were performed using SigmaStat 3.5 (Systat Software Inc, CA, USA) and GraphPad Prism (GraphPad Software Inc, CA, USA). Changes in parameters across altitude were compared using repeated-measures one-way ANOVA with a post-hoc test using the Holm-Sidak method versus baseline (SL, day 0). For all analyses, P < .05 was considered statistically significant.
Results
Anthropometric Characteristics of Climbing Participants
Ten subjects, 7 men and 3 women age 37 ± 9 years (range 26–49 years) completed all tests at all altitudes. There was no significant difference in age between men and women (men 38 ± 9 vs women 32 ± 7 yrs, P = .27). Table 1 describes anthropometric characteristics of the participants at SL, 4000 m, 4750 m, and 5300 m, which correspond to days 0, 3, 6, and 9, respectively. There was a significant decrease in body weight at 4750 m and 5300 m compared to sea level (SL).
Anthropometric characteristics and plasma glucose and lipoproteins
Values for parameters are given as mean ± SD; BMI (body mass index); NEFA (non-esterified fatty acids); TG (triglycerides); TC (total cholesterol); LDL-C (LDL cholesterol); HDL-C (HDL cholesterol). Repeated measures one-way ANOVA (RM one-way ANOVA) were performed on the data with a Holm-Sidak post hoc test. Post-hoc p values, using baseline 350 m as the reference values, are reported under the means and SD for each altitude.
Mountain Ascent Altered Lipids but Did Not Affect Glucose
All lipid and glucose values are presented in Table 1. Triglyceride constantly increased at all altitudes compared to SL. Again, compared to SL, NEFA significantly increased at 5300 m whereas HDL cholesterol decreased. There was an overall significant difference in LDL cholesterol and TC (P = .009 and P = .002, respectively), yet the post-hoc test did not reveal any significant differences for any of the individual altitudes compared to SL. There was no change in glucose at any time.
Climbing from Sea Level to 5300 Meters Altered the Adipose Tissue-Secreted Hormones Acylation-Stimulating Protein and Adiponectin
Adiponectin was significantly higher at 4750 m compared to SL (analysis of variance [ANOVA] P = .02, post hoc analysis [post-hoc] SL vs 4750 m P = .002, Figure 1A). There was no statistically significant difference in the concentration of circulating leptin (Figure 1B). Insulin levels increased significantly at 4750 m compared to SL (ANOVA P = .03, post-hoc SL vs 4750 m P = .02, Figure 1C). There was an increase in ASP from SL to 4000 m (ANOVA P = .02, post hoc SL vs 4000 m P < .05, Figure 1D) while the precursor to acylation-stimulating protein, C3, did not change (Figure 1E). In order to determine if changes in C3 conversion to ASP were responsible for the change in ASP, we calculated the percentage of ASP to C3 (%ASP/C3), which revealed a statically significant increase (P < .04, Figure 1F).
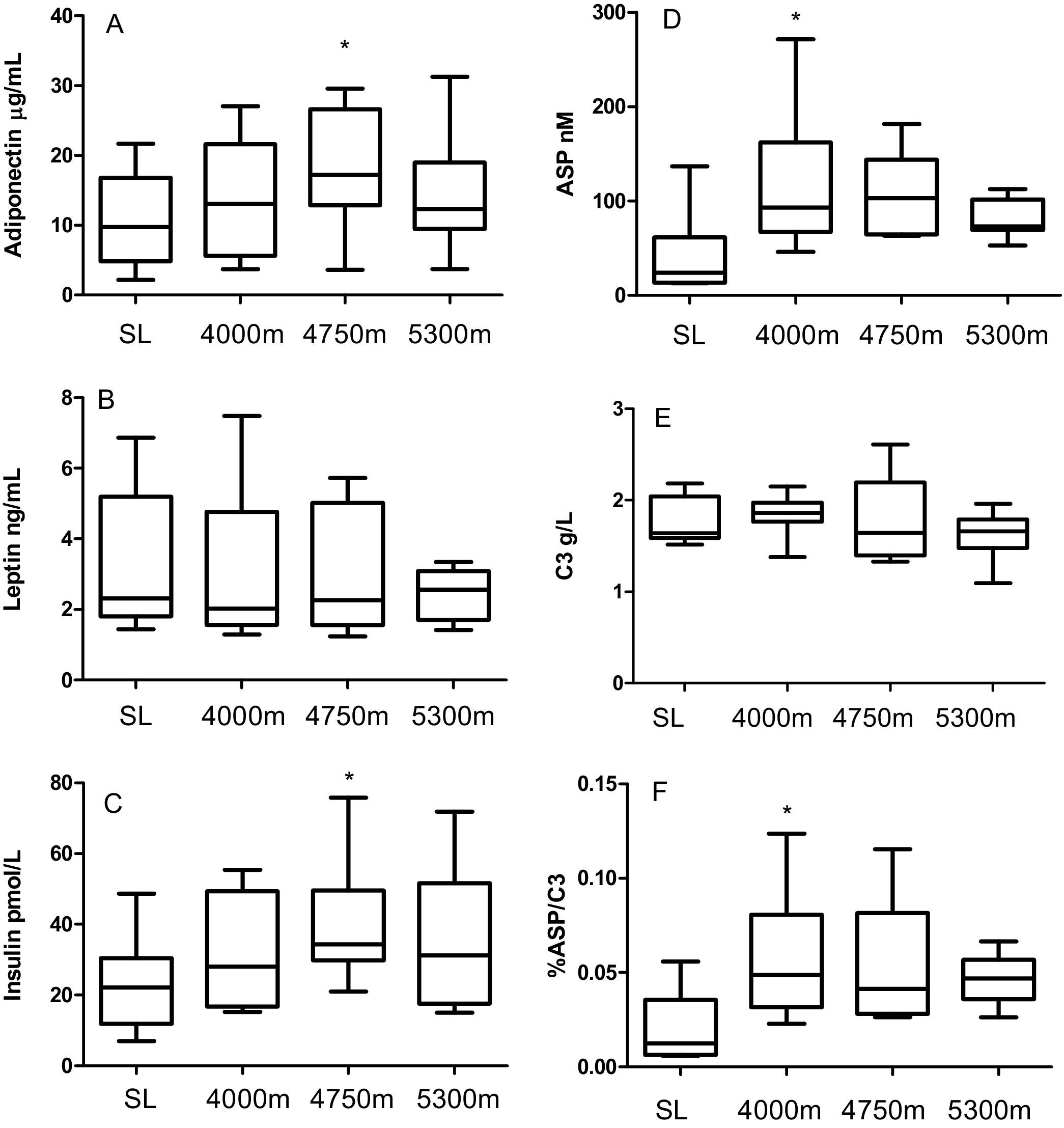
Adipokines measured at sea level (SL, day 1), 4000 m (day 3), 4750 m (day 6) and 5300 m (day 9) altitude (n = 10 subjects). Panel A: Adiponectin: Repeated measures one-way ANOVA (RM ANOVA) P = .02. Panel B: Leptin RM ANOVA pNS. Panel C: Insulin RM ANOVA P = .03. Panel D: Acylation-Stimulating Protein (ASP) RM ANOVA P = .02. Panel E: Complement C3 (C3) RM ANOVA P = .08. Panel F: Percentage ASP to C3 (%ASP/C3) RM ANOVA P = .04. Results are graphed as box (median, 25th percentile and 75th percentile) and whiskers (minimum and maximum) where * denotes P < .05 for Holm-Sidak post hoc test compared to SL value.
The Inflammatory Factor IL-6 Increased With Ascent but Cortisol and C-Reactive Protein Did Not Change
There was no change in CRP at all altitudes during the ascent (Figure 2A). IL-6 increased overall, with a specific increase at 5300 m compared to SL (ANOVA P < .001, post-hoc SL vs 5300 m P = .0007, Figure 2B). Lastly, cortisol did not change at any altitude compared to SL (Figure 2C).
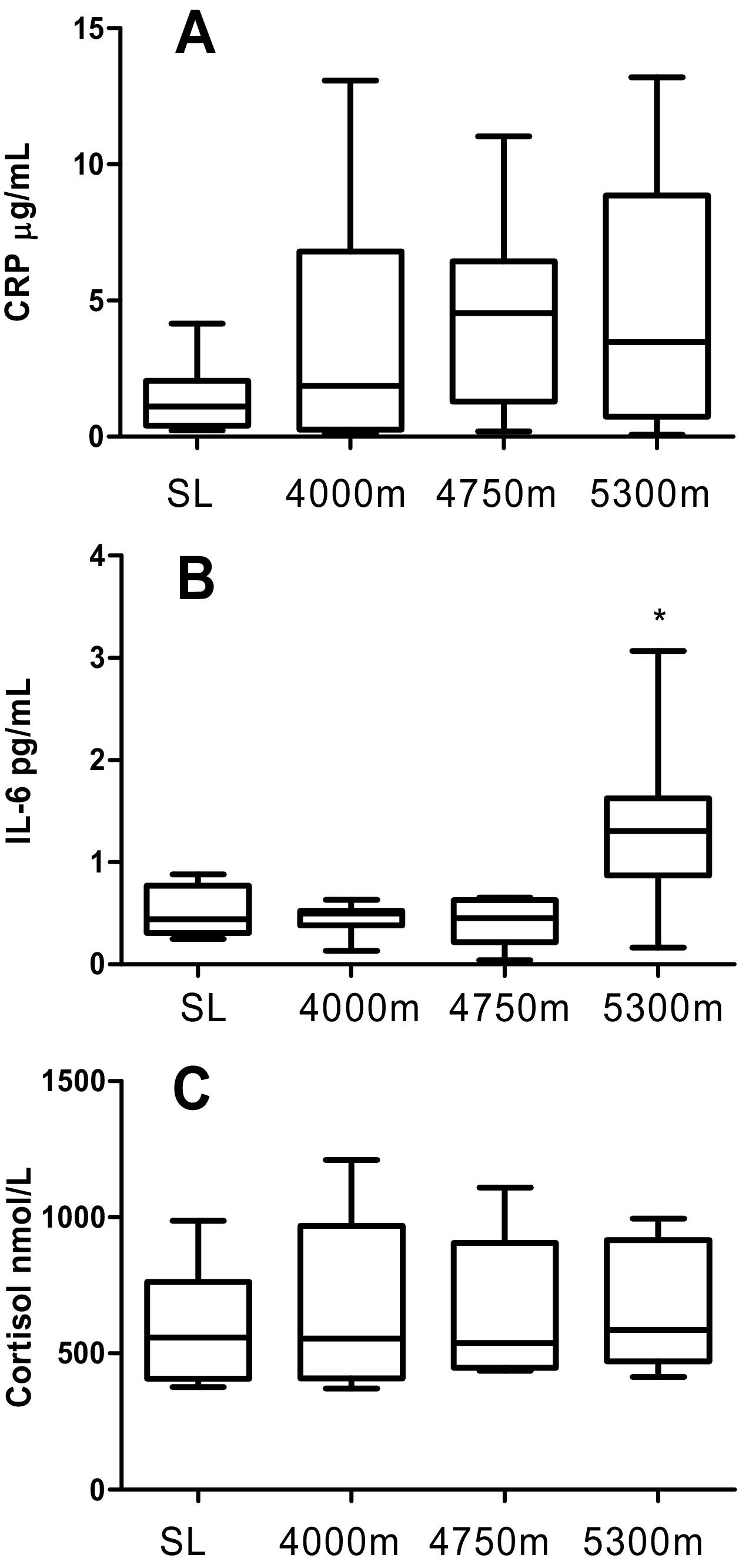
Inflammatory Factors measured at sea level (SL, day 0), 4000 m (day 3), 4750 m (day 6) and 5300 m (day 9) altitude (n = 10). Panel A: C-reactive protein (CRP) repeated measures one-way ANOVA (RM ANOVA) P = .12. Panel B: Interleukin 6 (IL-6) RM ANOVA P < .001. Panel C: Cortisol RM ANOVA P = .64. Results are graphed as box (median, 25th percentile and 75th percentile) and whiskers (minimum and maximum) where * denotes P < .05 for Holm-Sidak post hoc test compared to SL value.
Discussion
This study is the first to examine ASP at high altitude. The study suggests that changes in the adipokines ASP and adiponectin, the inflammatory marker IL-6, and insulin may be related to the high-altitude changes in metabolism highlighted by the changes in weight and plasma lipids. On the other hand, we did not observe changes in leptin, cortisol, or CRP, in contrast to other reports (as discussed below). It has been generally reported in the literature that most individuals lose weight during high altitude exposure. 17 –20 Our results are in accordance with what has been previously published, as all our subjects continuously lost weight throughout the ascent. Regardless of methodology, (BIA, skinfolds, or post-high altitude use of dual-emission X-ray absorptiometry [DEXA]), there is consensus in the literature that high altitude leads to body weight loss consisting of decreased body water as well as decreased lean body mass (LBM) and/or fractional muscle (FM). 21
Among the hormones we measured that are related to adipose tissue, we found that adiponectin and ASP increased with increasing altitude. However, the precursor to ASP (C3) did not change, thereby suggesting an increased conversion of C3 to ASP. While the exact mechanism influencing the conversion of ASP to C3 is not known, it has been shown that dietary fat (especially chylomicrons) and some pharmaceutical agents can increase ASP production. 22 This suggests regulation at the level of conversion of C3 to ASP, serving to increase the level of bioactive ASP rather than having an effect on the C3 precursor protein that is present at much higher concentrations. No other study has evaluated C3 or ASP at high altitude, and ASP not only plays a role in adipose tissue TG storage but may also influence satiety. Acylation-stimulating protein-deficient mice (C3KO) display increased food intake and delayed dietary TG clearance, which is normalized upon injection of ASP into C3KO. 23 Our preliminary results in rats show that central injection of ASP into the intracerebroventricular cavity decreased food intake. 24 ASP has been shown to change in response to several physiological conditions in humans, including decreasing with chronic exercise training, and increasing with chronic metabolic derangements such as cardiovascular disease and Type II diabetes. 25 Thus, several possible hypotheses can be proposed for the change in ASP at high altitude: a response to changes in energy substrate released from adipose tissue, a compensation due to increased TG or NEFA, a response to the changes in body weight and suspected increased satiety, or a result of the influence of other adipokines (such as adiponectin).
To the best of our knowledge, only one other study has measured adiponectin at high altitude, where no changes were observed regardless of caloric intake. 2 The same study also reported a decrease in adiponectin at SL when food intake was restricted to quantities reported with high altitude anorexia. 2 Acute exercise does not affect adiponectin concentration whereas certain conditions, such as obesity, reduce adiponectin.26,27 Therefore, the increased adiponectin reported here may be related to stimulation of substrate oxidation for energy usage, or in response to changes seen in other adipokines, potentially ASP. These hypotheses remain to be tested.
Two other inflammatory factors measured in this study, hsCRP and C3, also did not change with altitude. Imoberdorf et al also reported no change in CRP in subjects who passively ascended the mountain. 28 Bailey and colleagues found that, in healthy subjects, CRP did not increase and only those individuals who exhibited signs of acute mountain sickness had increased CRP. 29 On the other hand, we did observe an increase in IL-6. Previous studies support our findings of increased IL-6 with increasing altitude 28 ,30-32 although, again, Bailey and colleagues only found an increase in IL-6 among those individuals exhibiting signs of acute mountain sickness, and not among the healthy participants. 26 Interestingly, IL-6 typically increases rapidly with physical activity and can have a direct influence on increasing the production of CRP by the liver. 33 Despite this, in our climbers, IL-6 did not increase until 5300 m and was not associated with a simultaneous increase in hsCRP. In addition, it has been reported that muscle-derived IL-6 (such as that from exercise) may play a role in increasing gluconeogenesis in the liver. 34 Overall, the increases in ASP and %ASP/C3 occurred first, followed by increases in insulin and adiponectin and, lastly, by increases in IL-6.
Cortisol has a strong influence on skeletal muscle catabolism; 35 however, we found no change in cortisol. Cortisol response to high altitude remains controversial, with no change reported by some researchers, 3 whereas others have found an increase in cortisol. 12 ,36,37 Imoberdorf and colleagues found an increase in cortisol whether subjects actively (walking) or passively (air-lifted) ascended to 4559 m. 38 Measurements of fractional muscle synthesis rates increased in the active ascent group despite the increased cortisol. 38 Overall, changes in cortisol appear not to be related to body weight changes nor to energy intake at high altitude, as reported by the current study and previously published literature.35,4
We also found deleterious modulations of the lipid profile in our climbers as demonstrated by an early increase in TG (that was maintained), a gradual continuous increase in NEFA, and a later decrease in HDL-C. These changes in lipids that occurred simultaneously with the hormone changes mentioned above may be associated with altered energy utilization in response to the negative energy balance. Increased NEFA may lead to increased TG lipoprotein secretion from the liver. 39 There was no change in plasma glucose yet insulin levels were increased, suggesting insulin resistance; however, this was not caused by elevated cortisol. Potentially, increased insulin secretion may also contribute to increased satiety and weight loss. Whether these changes are dictated by adipokines and cytokines, or vice versa, requires further investigation.
Due to the observational nature of this article, it is not possible to identify specific mechanisms. Further, it was not possible to examine effectively the role of gender in body composition and metabolic changes at altitude due to our small sample size and inclusion of only 3 women, although there was no significant difference in the high altitude response between men and women. However, based on our findings, we hypothesize that the increased physical demands combined with altitude-induced hypoxemia during this expedition led to the increased ASP, adiponectin, insulin, IL-6, TG, and NEFA. Using a simulated Everest ascent (decompression chamber), analysis of subcutaneous adipose tissue demonstrated a decrease in lipolysis, suggesting a potential mechanism by which adipose tissue-related hormones may change. 40 Due to the efficacy of high altitude exposure to induce increased satiety and weight loss, it would be tempting to propose high altitude exposure or the use of barometric chambers, possibly in combination with exercise, as a weight loss tool. However, based on the findings of this study, the combined physical stresses of activity, cold, and hypoxia have led to negative metabolic consequences including increased inflammatory markers (IL-6), increased insulin levels, and worsening lipid profile. Therefore, short-term exposure to altitude-induced hypoxemic conditions may have deleterious metabolic consequences despite weight loss.
Conclusion
Ascent to an altitude of 5300 m over a 9-day period induced changes in metabolism-related hormones including increases in ASP, adiponectin, insulin, and IL-6. These changes may be related to the weight loss seen in our participants or to changes in plasma lipids.
Footnotes
Acknowledgment
The authors would like to thank Dr Peter Bogaty for his scientific guidance in the preparation of this manuscript.
