Abstract
A 2-year-old, female spayed, domestic shorthair cat presented to the University of Missouri-Veterinary Medical Teaching Hospital (UMC-VMTH) with an approximately 11-month history of fluid-draining pockets along her ventral thorax and axillae. The skin in these regions was erythematous, and multiple areas drained a serous to serosanguinous fluid. Fluid-filled, nodules formed along the ventrum, but these nodules disappeared as fluid drained spontaneously. Histologic assessment of skin biopsies revealed areas of vascular proliferation extending along the deep margin of the section and rare instances of invasion into the superficial dermis. These vascular channels were devoid of cells, lined by variably pleomorphic endothelial cells which had a low mitotic index. Based on the mild to moderate pleomorphism, positive staining with prospero-related homeobox gene-1 (PROX-1), and the locally aggressive nature of the tumor, a final diagnosis of lymphangiosarcoma was made.
Lymphangiomas and lymphangiosarcomas are often architecturally comparable to hemangiomas and hemangiosarcomas but are characterized by a paucity of erythrocytes within the vascular channels. 1 These tumors can be further differentiated by the expression of lymphatic vessel endothelial receptor-1 (LYVE-1) and prospero-related homeobox gene-1 (PROX-1) in lymphatic vessels only.2,3 Lymphangioma (also described as a hamartoma) and lymphangiosarcoma are uncommon neoplasms in the cat that originate from the endothelial cells of lymphatic vessels. In humans, lymphangiosarcomas frequently arise in areas of chronic lymphedema. 4 This case report describes the clinical presentation, morphologic features, and outcome in a cat with a locally aggressive lymphangiosarcoma.
A 2-year-old, female spayed, domestic shorthair cat, presented to the University of Missouri-Veterinary Medical Teaching Hospital (UMC-VMTH) with an approximately 11-month history of fluid-seeping pockets along her ventral thorax and axillae. Firm nodules would form in these areas, but nodules would eventually empty when they would drain a serous or serosanguinous fluid. The cat had completed numerous therapeutic trials with various anti-microbial drugs and corticosteroids without appreciable benefit. Additionally, the cat had undergone multiple wound debridement procedures and surgical excision of nodules. The histopathologic diagnosis from tissue excised during one of these procedures was panniculitis. The cat continued to behave normally until 4 days prior to presentation, at which time she became lethargic. Abnormalities noted on physical examination included erythematous skin along her ventral thorax and axillae with alopecia in the affected areas (Fig 1). The skin in these areas was moist with serous and serosanguinous fluid oozing from the surface of the skin. There was a circular, draining tract, resembling a puncture wound, approximately 7 mm in diameter located in the right axillary region that could not be attributed to any previous biopsy. Our differentials at this time were an infection (an atypical mycobacterium, nocardia, or actinomyces) and neoplasia.

Ventral thorax and axillary region, revealing erythematous skin with a moist surface and multiple areas of alopecia.
Initial evaluation included complete blood count (CBC), serum chemistry profile, retroviral testing, and skin biopsy for culture and histopathologic assessment. CBC was unremarkable and showed a mild lymphopenia (0.66 × 103/μl; 1.5–7) (Sysmex XT 2000iV, Sysmex America, Mundelein, IL). Chemistry panel abnormalities consisted of a mild hyponatremia (145 mEq/l; 149–159), mild hypochloremia (110 mEq/l; 114–122), hypoproteinemia (5.5 g/dl; 6.4–8.8), and a very mild hyperphosphatemia (5.4 mg/dl; 2–5.3) (AU 400, Beckman Coulter, Brea, CA). Retrovirus testing via an in-clinic enzyme-linked immunosorbent assay (ELISA) was negative. Under general anesthesia, two dermal punch biopsy (6 mm) samples were obtained for tissue maceration culture and sensitivity testing and fungal culture. A 1.5 × 1.0 × 0.2 cm elliptical incisional biopsy was submitted for histopathologic review. The cat was discharged while awaiting the culture and biopsy results. Trimethoprim—sulfamethoxazole (15 mg/kg PO q 12 h) and doxycycline (5 mg/kg PO q 12 h) were prescribed for potential mycobacterial infection. Buprenorphine (0.02 mg/kg PO q 12 h) was also prescribed to be used as needed for pain the 2–3 days following biopsy.
Culture failed to identify a causative infection. Fungal and anaerobic cultures produced no growth. Light growths of two different staphylococcal organisms identified by the aerobic culture (Staphylococcus aureus and Staphylococcus epidermidis) were attributed to skin contamination or colonization. The epidermis was moderately thickened with small to moderate numbers of lymphocytes, plasma cells, and mast cells present in the superficial dermis. There was an irregular area of small vessel proliferation extending along the deep margin of the section and rarely invading into the superficial dermis. Vascular channels were formed by small, plump endothelial cells growing directly on dermal collagen bundles, dissecting them and forming irregular clefts (Fig 2). These channels sometimes surrounded and entrapped adnexal structures. The endothelial cells had rounded, hyperchromatic nuclei with very rare binucleate cells and a mitotic index of 1/10 40 × fields. Most of the vascular spaces were clear, but some contained a small number of red blood cells. There were multifocal areas of neutrophil accumulation, particularly where the vascular channels approached the surface or contained entrapped hair follicles. No organisms were identified on the hematoxylin and eosin or acid-fast stains.

Skin biopsy. The superficial dermis is infiltrated by irregular vascular channels lined by small plump endothelial cells. Hematoxylin and eosin. Bar = 100 μm.
Slides were immunohistochemically labeled for vimentin, CD31, and PROX-1 for identification of mesenchymal cells, endothelial cells and lymphatics, respectively. For immunohistochemistry, paraffin-embedded samples were positioned on slides, deparaffinized, hydrated, and placed in Tris buffer for at least 5 min prior to staining. Antigen retrieval was either by proteinase K digestion (vimentin) for 5 min or steamed in a decloaking chamber (CD31 and PROX-1) (Biocare Medical, Concord, CA) at 98° for 30 min. Slides were treated with 3% H2O2 for 15 min, rinsed, then subjected to protein block (Sniper, Biocare Medical, Concord, CA) for 20 min and drained. Sections were incubated in primary antibody mouse monoclonal anti-vimentin (M7020, Dako, Carpinteria, CA 1:200) for 20 min, primary antibody mouse monoclonal anti-CD31 (M0823, Dako, Carpinteria, CA 1:40) for 60 min, or primary antibody polyclonal rabbit anti-PROX-1 (11-002; AngioBio, Del Mar, CA 1:800) for 90 min. The detection system used was either Mach2HRP (CD31 and vimentin) (M2U522, Biocare Medical, Concord, CA) or LSAB+ (labeled streptavidin biotin) (Prox-1) (Dako, Carpinteria, CA) for 30 min. The chromogen used was Romulin Red (Biocare Medical, Concord, CA) for 10 min. Slides were then counterstained in CAT Hematoxylin (Biocare Medical, Concord CA) at a 1:10 dilution for 5 min, dehydrated and cover slipped. Normal feline skin or lymph node was used as a positive control. Negative controls were performed under identical conditions, incubating with Mouse IgG (Sigma, St Louis, MO) at a 1:1000 dilution for 30 min.
Special stains for vimentin, CD31, and vWF were positive for the cells lining the spaces, confirming their vascular origin, but these stains cannot differentiate between vascular types (Fig 3). Therefore, PROX-1 staining was performed and was positive, indicating lymphatic endothelial cell origin (Fig 4). Tissue from the prior biopsy that had yielded the diagnosis of panniculitis was made available for evaluation and compared to biopsy samples obtained at UMC-VMTH. Upon review, the previous biopsy contained the abnormal vascular spaces; however, they were obscured by much larger areas of inflammation. Thus, the previous diagnosis of panniculitis, while accurate, was a mis-diagnosis of the underlying neoplasm.

Immunohistochemistry. Positive reaction for CD31 in the cells lining the clear spaces (brown cytoplasmic staining) indicating vascular origin. Bar = 50 μm.
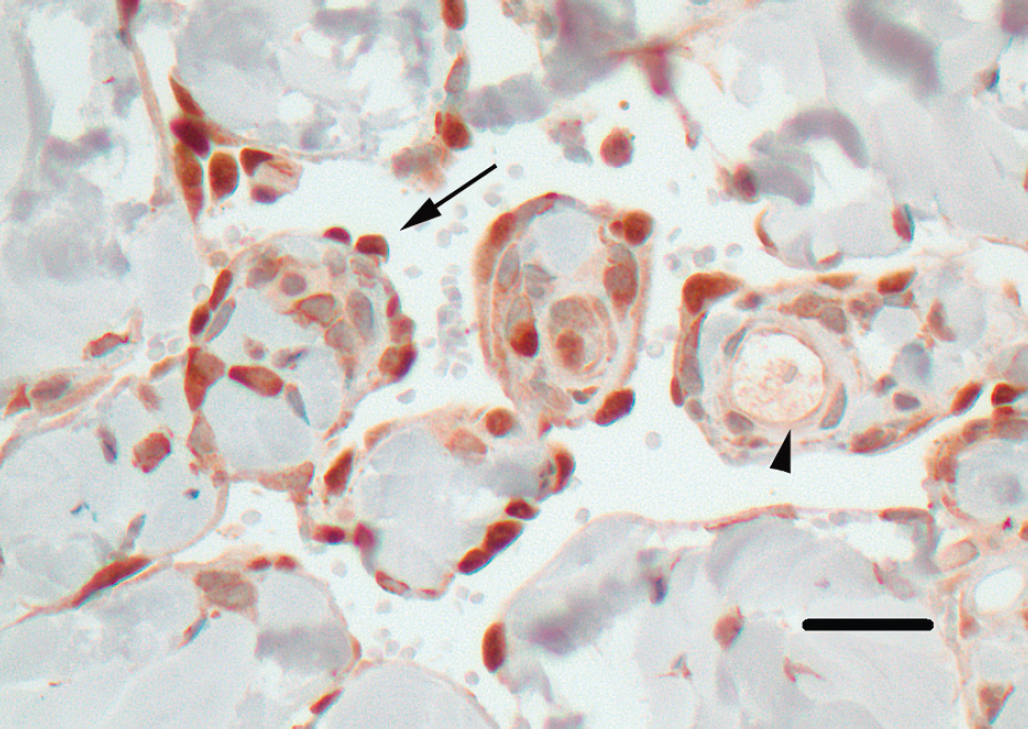
Immunohistochemistry. Positive reaction for PROX-1 in the cells lining the clear spaces (brown nuclear staining, arrow) but not in the blood vessel endothelium (arrow head) indicating lymphatic vessel origin. Bar = 25 μm.
Based on the clinical signs of chronic serosanguinous discharge, lack of hemorrhage, and a relative lack of red blood cells within the vascular channels, the lesions were likely of lymphatic origin. This was confirmed with positive staining of PROX-1. Lymphangiosarcoma was considered likely because of the severity and extensiveness of the lesions and the moderate cellular atypia, even though the cells exhibited a low mitotic rate. The clinical presentation was similar to ‘feline abdominal lymphangiosarcoma;’ although, this cat presented with more anterior lesions. 3 The final diagnosis was lymphangiosarcoma and secondary inflammation.
Lymphangiosarcomas are infrequently reported in the literature. 5 They are often associated with chronic lymphedema in humans; however, an underlying cause in animals remains unknown. Definitive diagnosis is based on histopathologic examination; however, as in our case, differentiation of lymphangiosarcoma from hemangiosarcoma can be difficult.5,6 Differentiation between the two often depends on the scarcity or presence of red blood cells within the channels as seen in lymphangiosarcoma and hemangiosarcoma, respectively. 1 One study reported the use of ultrastructural features to differentiate lymphatic from blood vessel endothelium. Lymphatic vessels are characterized by a discontinuous basal lamina or none at all, fewer intercellular junctions, and fewer pinocytotic vesicles. They also lack pericytes and may have gaps between endothelial cells. On the other hand, blood vessel endothelium is characterized by a continuous basal lamina, numerous micropinocytotic vesicles, well-developed intercellular tight junctions, and may be surrounded by pericytes. 7 Recently, immunohistochemistry using LYVE-1 and PROX-1 have been used to differentiate lymphatic vessels from blood vessels.2,3
Surgical excision and radiation therapy were offered as treatment options. Wide surgical excision is often recommended for the treatment of angiomas, but this may be difficult if the lesions are extensive or infiltrative. 8 Prior surgeries in this cat had not attempted to provide wide margins and were not successful in resolving the disease. Attempted resection of all involved tissue would have created a very large wound without adequate tissue to close the resulting defect. Radiation therapy was reportedly used in a single dog with lymphangioma after multiple failed surgical resections. Radiation was apparently successful as the treated dog remained well 2 years after treatment with no signs of tumor regrowth. 9 Laser photocoagulation was reportedly used to treat one dog and two cats with lymphangioma. Although one cat died from unrelated causes 6 months following treatment with no signs of recurrence, tumor regrowth occurred in the other two pets within 6 months. 8 The owner of the cat described in this report declined further treatment and elected euthanasia but declined necropsy examination.
