Abstract
This study describes signalment, history, antibiotic administered, clinical signs observed, therapy, and outcome of anaphylactic events within 4 h following ophthalmic administration of an antibiotic to cats. Data came from survey responses (45 cats) or Federal Drug Administration reports (16 cats). Cat age (7 weeks—19 years), breed, and gender ranged widely. Most were healthy (87%) prior to anaphylaxis. Ophthalmic antibiotics commonly were administered for conjunctival (65%) or corneal (11%) disease, or ocular lubrication (7%) and contained bacitracin, neomycin, and polymyxin B (44%), or oxytetracycline and polymyxin B (21%). Polymyxin B was present in all cases. Vaccines or other drugs were also administered to 51% of cats. In 56% cases, anaphylaxis occurred within 10 min of drug application. Most (82%) cats survived. Although a causal association was not proved, ophthalmic antibiotic administration preceded anaphylaxis in all cats. Like other drugs, ophthalmic antibiotics should be used only when indicated.
Ophthalmic preparations containing antibiotics are indicated for treatment of blepharitis, conjunctivitis, keratitis, or dacryocystitis when a bacterial etiology is known or suspected. They are also used for bacterial prophylaxis when anatomic or physiologic defense mechanisms are impaired, such as occurs with keratoconjunctivitis sicca, ulcerative keratitis, or surgical intervention involving the ocular surface or intraocular structures. In addition, antibiotics formulated in petrolatum or mineral oil bases have been used to lubricate the ocular surface when protection afforded by eyelids or tears is impaired. Therefore, topical ophthalmic antibiotics are frequently prescribed in a wide diversity of species.
Appropriate antibiotic selection is ideally based upon examination of differentially stained cytological samples together with culture and sensitivity results. 1 However, in many cases, such analysis is not immediately available or practical, and initial antibiotic selection is empirical. In North America, neomycin, bacitracin, polymyxin B, and gramicidin are commercially available in several combinations as ophthalmic formulations and are commonly used to treat ocular conditions in a variety of species. Warnings that one or more of these ophthalmic antibiotics may be associated with anaphylactic reactions in cats have appeared in non-peer-reviewed literature 2 and conference presentations for which proceedings were not produced (personal communication, E Kathryn Meyer, VMD). However, the authors are aware of only one case report in the peer-reviewed literature describing anaphylaxis suspected to be due to topical application of an ophthalmic antibiotic. 3 An ophthalmic solution containing neomycin, polymyxin, and a corticosteroid was suspected to have been the cause; however, the cat had also received topically applied proparacaine prior to administration of the antibiotic, making a causal association between antibiotic and anaphylaxis impossible to ascertain. That report also referred to five other cases of suspected anaphylaxis following topical ophthalmic preparations containing bacitracin, neomycin, and polymyxin, but provided no further details regarding these occurrences. Based upon quantity-marketed information reported to the Food and Drug Administration, Center for Veterinary Medicine (FDA CVM) between 1996 and 2009, an average of approximately 1,850,000 units (bottles/tubes) of veterinary ophthalmic antibiotics approved for use in cats were sold per year. However, quantity marketed data do not equate to doses actually administered to cats. Many of these antibiotics are also approved for use in other species, including dogs, horses, cattle, and sheep. Regardless, given the relative frequency with which veterinary antibiotic ophthalmics are prescribed and the current reporting rates of anaphylactic-type events, it appears that these events are sufficiently uncommon that a prospective study of this event would likely be unfeasible. Therefore, we retrospectively assessed cases of anaphylaxis in cats noted soon after they received an ophthalmic antibiotic. Specifically, the present study was designed to collect and analyze detailed clinical data from veterinarians who had directly observed anaphylactic events in cats within 4 h of topical administration of at least one of neomycin, bacitracin, polymyxin B, or gramicidin. In particular, we wished to describe the cat's signalment and history, and the incriminated drug, clinical signs, therapy, and outcome in such cases.
Materials and methods
To investigate a temporal association between anaphylaxis and ophthalmic antibiotic administration, a survey was designed for veterinarians who had seen such cases (Supplementary data {TAB Survey file}). Prior to release, the survey was validated by 22 veterinarians, including the authors, a veterinary epidemiologist, and several veterinarians who had seen suspected cases. This survey was designed without input from the FDA. The survey was made available electronically through the Veterinary Information Network (VIN), and invitations to participate sent by email to 40,251 VIN members who had previously indicated that they were willing to receive surveys. The survey was also made available to 219 diplomates of the American College of Veterinary Ophthalmologists who subscribed to the College's list serve. Approximately 17 days later, 27,326 VIN members and all American College of Veterinary Ophthalmologists list serve members were sent a reminder email, and the survey link was distributed to 68,812 VIN members via a weekly newsletter. Veterinarians who became aware of the survey via any alternate methods were also eligible to respond. Data were entered anonymously; however, respondents were encouraged to add their contact details so that additional information could be solicited if necessary. Veterinarians reporting more than one case were asked to complete a separate survey response for each cat. To minimize duplicate reporting and indirect observations, veterinarians who were aware of but had not primarily managed cases were asked not to complete the survey. The survey was accessible by respondents for 38 days in February and March, 2010. Nineteen respondents who had not completed the survey when it was withdrawn received personal emails asking them to complete data input; six subsequently provided complete data. Data from all completed survey responses were compiled into a spreadsheet (Excel) for analysis.
In addition, the FDA CVM was asked to identify reports of anaphylactic events following administration of an ophthalmic antibiotic that were voluntarily submitted to the agency between 1996 and 2009. Following identification, review, and analysis of these reports by FDA CVM staff, all pertinent data were added to the same spreadsheet. Because reports to the FDA CVM had not included all information sought through the survey, data for some questions were incomplete. A number of measures were undertaken to ensure that duplicate data were not retrieved from survey respondents and FDA CVM records. First, a question in the VIN survey asked respondents if cases had been reported to the FDA or any other organization. Second, all data sets within the spreadsheet were assessed for overlapping signalment, reporting region, or incident date. Where duplicate entries within or between the two means of reporting were suspected, the more complete data set was analyzed and the suspected duplicate was discarded.
The definition of anaphylaxis used in this study was ‘a severe, fatal or potentially fatal, systemic, allergic reaction that occurs suddenly after contact with an allergy-inducing substance.’ 4 All reports from both sources were critically reviewed to ensure they fulfilled this definition as completely and accurately as possible. Cats were included only if they demonstrated clinical signs affecting two or more organ systems, or affecting only one organ system but severe enough to cause death. Cats which had an ophthalmic antibiotic applied to a location other than the eye, those in which the anaphylactic event occurred more than 4 h or at an unknown time after antibiotic application, and cases where the respondent suspected that another drug was responsible for anaphylaxis were excluded. In addition, specific actions were taken to reduce limitations inherent in retrospective studies conducted by survey. First, survey questions always included response options such as ‘unsure’ or ‘unknown’, so as to limit solicitation of definitive responses in situations where respondents could not answer confidently. With the same intent, answers were required only for those questions that determined inclusion criteria. Finally, all reports where the data provided were insufficient to permit the authors to fully verify inclusion criteria were excluded.
Results
A total of 998 veterinarians completed the survey. Of these, 922 respondents reported never witnessing anaphylaxis following ocular administration of topical antibiotics to a cat, while 76 respondents reported 86 anaphylactic events in this context. An additional 23 events were reported to the FDA CVM; one of which was also reported via the survey. Thus a total of 108 apparently unique cases of anaphylaxis following ocular administration of a preparation containing at least one of neomycin, bacitracin, polymyxin B, or gramicidin were reviewed. Of these, 47 cases failed to meet all inclusion criteria. Therefore, the following is a summary of data regarding 61 cats collected from 41 survey respondents (45 cats) and 16 FDA CVM reports (16 cats) which fulfilled all entry criteria and for which sufficient data were available.
The 41 survey respondents included 39 veterinary practitioners and two veterinary ophthalmologists. Data regarding specialty training were not available for the 16 cases reported to the FDA CVM. All 61 reports were collected from the United States (Southwest: 18, Southeast: 17; Northeast: 14; Midwest: six; Northwest: four); location was not noted in two cases. Reported anaphylactic events occurred between 1993 and 2010; year was unknown in 20 cases. Age of affected cats was reported for 51 cats with a median of 3.5 years (range 7 weeks to 19 years). Twenty-four (40%) were neutered males, 20 (33%) were spayed females, five (8%) were intact females, and two (3%) were intact males; in 10 (16%) cases respondents were unsure of the cat's gender. The population consisted of 31 (51%) domestic shorthair cats, five (8%) domestic longhair cats, three (5%) each Siamese, Himalayan, and Persian cats, and Ragdolls, one (2%) unspecified ‘purebred’, and 12 (20%) mixed breed cats.
Prior to the anaphylactic event, 8/61 (13%) cats had been diagnosed with systemic disease including upper respiratory disease (n = 5), diabetes mellitus (n = 1), restrictive cardiomyopathy with secondary arrhythmia (n = 1), and an abscess at an unidentified site and congestive heart failure subsequent to fluid overload (n = 1). Four cats were receiving other medications (lysine, amoxicillineclavulanic acid, methimazole, ivermectin, or chloramphenicol ophthalmic ointment) at the time of the anaphylactic event but were not reported to be systemically ill. In five cases the respondent was unsure if the cat was receiving other medications or nutritional supplements.
Reasons for use of the ophthalmic antibiotics administered in the 4 h prior to anaphylaxis were described as treatment of conjunctivitis or inflammation of the third eyelid (40 cats; 65%), ulcerative keratitis/corneal abrasions (seven cats; 11%), ‘ocular infection’ (four cats; 7%), lubrication for unspecified reasons or during general anesthesia (four cats; 7%), or ‘to facilitate eye examination’, for ‘intermittent epiphora’, or ‘following corneal surgery’ in one cat each. The reason for ophthalmic antibiotic administration was not specified for three cats (5%). Commonly-applied products were an ophthalmic ointment containing bacitracin, neomycin sulfate, and polymyxin B (27 cats; 44%) or oxytetracycline and polymyxin B (13 cats; 21%). In 11 cases (18%) bacitracin, neomycin sulfate, polymyxin B and hydrocortisone acetate ointment was implicated. Seven of 61 cats (11%) received neomycin sulfate, polymyxin B, and dexamethasone; four as an ointment and one as a suspension. A single case each was attributed to bacitracin, gramicidin, neomycin, polymyxin B ophthalmic solution; bacitracin, neomycin sulfate, and polymyxin B ophthalmic solution; and flumethasone, neomycin and polymyxin B ophthalmic gel. In 75% of 61 cats, the ophthalmic drug manufacturer was not known or provided. For the remaining 15 cats, a wide variety of commercial manufacturers was represented including Alcon (n = 4), Pfizer (n = 3), and Bausch and Lomb, Pharmaderm, and Evsco, Dechra (one case each). In four instances a generic brand was incriminated.
Information regarding known previous administration of at least one of the component drugs in the antibiotic preparation and unusual reactions to other medications was solicited only from survey respondents; this information was not available from the FDA CVM data set. Of the 45 cases collected via the survey, six (13%) cats were reported to have received the same antibiotic previously. These included two cats that had received preparations containing bacitracin, neomycin sulfate, and polymyxin B, two that had received neomycin sulfate, polymyxin B, and dexamethasone, one that had received dexamethasone, and one that had received an unspecified ‘triple antibiotic under the supervision of a pharmacist’. Four of 45 (9%) cats from the survey were reported to have previously demonstrated unusual reactions to other medications, including oxytetracycline and polymyxin B ophthalmic ointment (n = 1; several episodes of vomiting) and vaccines [n = 3; gastrointestinal signs and lethargy (one), vomiting, diarrhea, and hyperemic skin (one), and unspecified (one)]. A total of 31 (51%) cats received vaccines or drugs other than the ophthalmic antibiotic during the same visit at which the anaphylactic event occurred (Table 1). The most commonly administered medication was fluorescein stain, which was administered to 22 (36%) cats. In seven of these cats, fluorescein was applied along with ophthalmic proparacaine (n = 2), tropicamide (two), atropine (one), dexamethasone (one), or chloramphenicol ophthalmic ointment and oral L-lysine (one).
Other medications and vaccines administered during the same visit, but prior to the anaphylactic event.
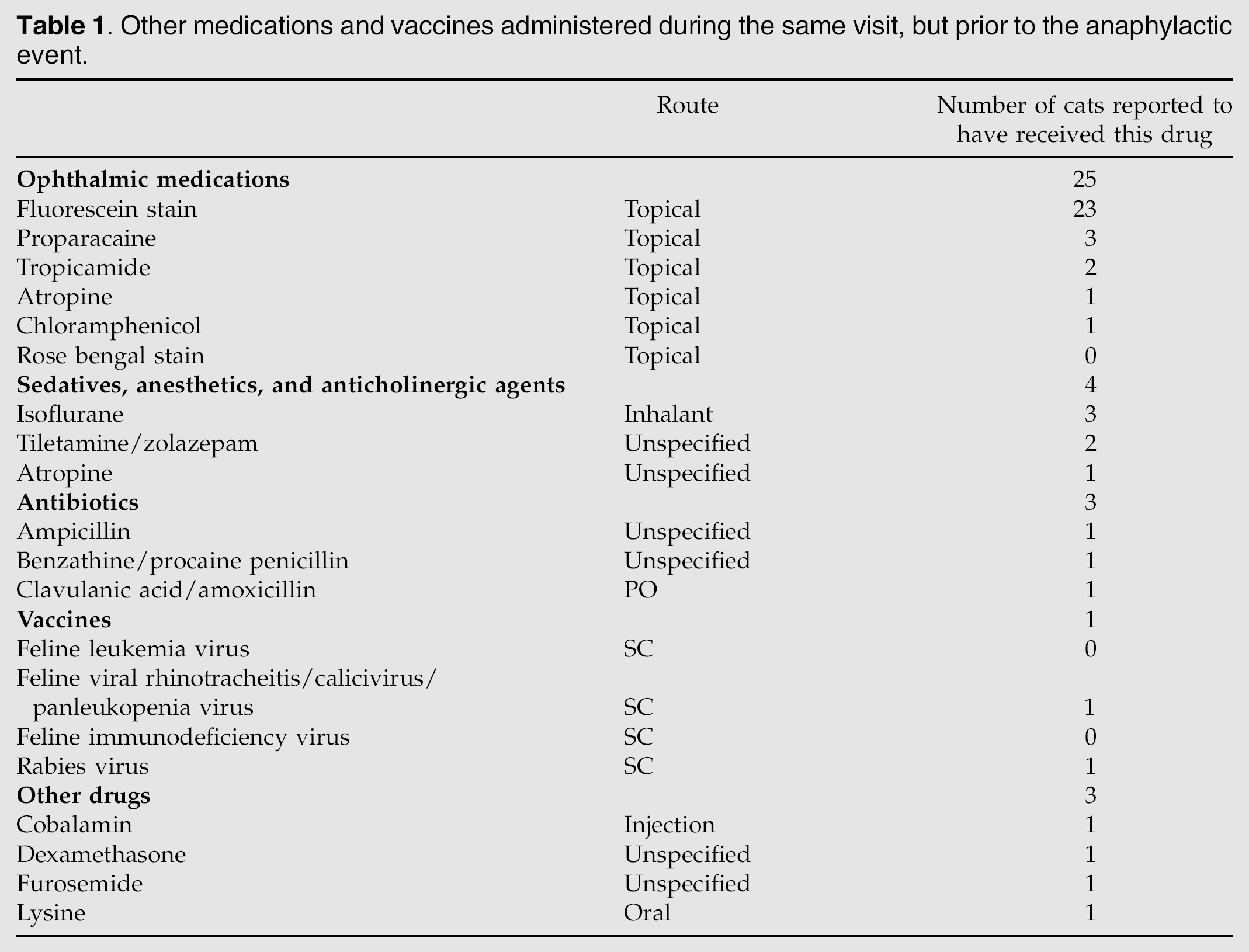
The remaining nine cats not receiving fluorescein, did receive ophthalmic proparacaine (n = 1) or systemically administered isoflurane (one), tiletamine, zolazepam and isoflurane (two), benzathine procaine penicillin and atropine (one), amoxicillineclavulanic acid (one), furosemide and ampicillin (one), FVRCP and rabies vaccines (one), or cobalamin/vitamin B12 (one).
As determined by inclusion criteria, clinical evidence of anaphylaxis was observed within 4 h after ophthalmic antibiotic application in all cats. In 24 (39%) cases, anaphylaxis occurred within 5 min of drug application, and of these, 12 (20%) occurred within 60 s. Ten events occurred within 5–10 min, eight within 11–15 min, three within 16–20 min, three within 21–30 min, one within 31–45 min, three within 46–60 min, and one within 3–4 h of drug administration. Eight respondents indicated that the event occurred ‘within minutes’ but were not more specific. Clinical signs associated with anaphylaxis varied widely but, as determined by inclusion criteria, always involved at least two body systems; most commonly gastrointestinal, respiratory, ocular, dermatologic, or cardiovascular systems in various combinations (Table 2). Respiratory and gastrointestinal signs were most commonly reported, and often occurred together (31 cats). Three cats exhibited multisystemic signs implicating simultaneous involvement of respiratory, ocular, dermatologic, neurologic, gastrointestinal, and cardiovascular systems; all three survived. Therapy typically involved administration of fluids, corticosteroids, antihistamines, epinephrine, and oxygen in various combinations; often in conjunction with flushing of the antibiotic preparation from the eye (Table 3).
Clinical signs observed during the anaphylactic event. (Only more frequently observed signs are listed.)

Treatment provided for anaphylactic event.
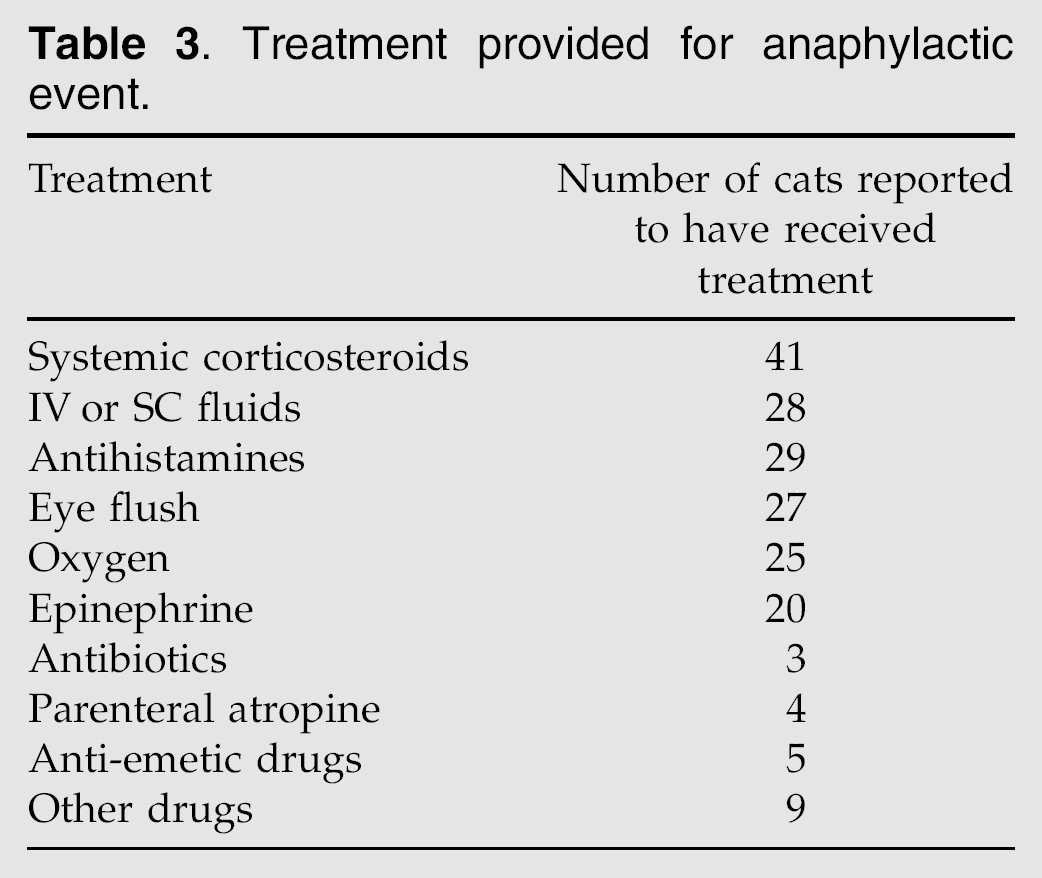
Fifty of 61 (82%) cats survived the anaphylactic event; however, one cat became blind and remained in a mentally debilitated state and another with a radiographic and echocardiographic diagnosis of restrictive cardiomyopathy was euthanased 7 days after the anaphylactic event. Outcome in three (5%) cases was not reported. The eight (13%) deaths occurred within ‘minutes’ (n = 1), 15 min (n = 1), 20 min (n = 1), 30–40 min (n = 1), 2 h (n = 1), 12 h (n = 1), or 24 h (n = 1) following application of the ophthalmic antibiotic. Time to death was not specified for one cat. Of the eight cats which did not survive, two experienced a combination of respiratory, neurologic, gastrointestinal, and cardiovascular signs; and one each exhibited respiratory, ocular, and neurologic signs; respiratory, neurologic, and cardiovascular signs; and respiratory and gastrointestinal signs. Two cats died after demonstrating signs implicating only one body system(respiratory or ocular). No specific clinical signs were listed for one cat which died within 15 min of product application. Although there was no obvious pattern linking specific clinical signs with outcome, 6/8 cats that subsequently died experienced vomiting, and 4/8 vomited and had concurrent respiratory signs. Of the eight cats that did not survive, none had their eyes flushed after event onset; 27/53 that survived did have their eyes flushed. Six of the eight (75%) cats that died received, as treatment for anaphylaxis, epinephrine in conjunction with other treatments including oxygen, dexamethasone sodium phosphate, and fluids (one), oxygen, dexamethasone sodium phosphate and atropine (one), oxygen and atropine (one), oxygen and fluids (one), dexamethasone sodium phosphate and atropine (one), and systemic steroids (one). Of the remaining two cats that died, one received an ophthalmic preparation of neomycin sulfate, polymyxin B, and dexamethasone as the only treatment and one cat did not receive any treatment for anaphylaxis. Twenty-two of the 50 cats (44%) that survived received epinephrine, in various combinations with fluids, eye flushing, oxygen, corticosteroids, and diphenhydramine. The most common combination of therapies used in cats that survived included fluids, flushing the eyes, systemic corticosteroids, and diphenhydramine (n = 10).
Three cats had necropsies performed. Necropsy findings were reported as ‘consistent with an acute anaphylactic reaction’ (n = 1), congestion of lungs, liver, small intestine, and kidneys (n = 1) or ‘unknown’ (n = 1). Of the 45 cats for which data were obtained by survey, the anaphylactic event was not reported to any organization in 25 (56%) cases and the respondent was unsure if a report had been made in a further 10 (22%) cases. Respondents indicated they had filed a report for 10 (22%) cats; eight to the drug manufacturer, two to the FDA CVM, one to United States Pharmacopeia practitioners' network, and five to an unspecified organization. In six (13%) cats, the event was reported to more than one organization.
Discussion
To the authors' knowledge, this represents the largest retrospective analysis of data from cats experiencing anaphylaxis within 4 h following application of an ophthalmic antibiotic. Response to the call for information was enthusiastic with over 1000 surveys submitted in 38 days. Although the survey invitation encouraged responses from only practitioners who had seen relevant cases, only 8% of respondents reported observing this during a 17-year period, and only 45 cases fulfilled all study inclusion criteria. In an overlapping 13-year period, only 16 additional cases reported to the FDA CVM fulfilled all entry criteria. Thus, although this study was not designed to assess incidence of this adverse event in cats, these data suggest that such events occur uncommonly. Assuming this event is relatively uncommon, a prospective study would be impractical. Rather, retrospective analysis of data collected from multiple centers was required. However, such a study has inherent limitations. First, our survey required that veterinarians supplied detailed case information sometimes several years after the event. We attempted to minimize effects of incomplete records or altered recollection of events by including an ‘unsure' or ‘unknown' category in survey questions, by recommending that the medical record was available during survey completion, and by requiring that only veterinarians who had primarily managed the anaphylactic event complete surveys. However, ideally data should be collected immediately after adverse events occur when fact recall is likely to be clearest, further patient data can be collected prospectively in a standardized manner, and suspected drugs can be submitted for analysis.
The retrospective nature of this study also prohibited demonstration of a definitive causal association between ophthalmic antibiotic administration and anaphylaxis; in large part because 31 (51%) cats received vaccines or drugs other than the ophthalmic antibiotic during the visit. For example, approximately half of all cats in this study were known to (23 cats) or may (seven cats) have received topically administered fluorescein stain around the time anaphylaxis occurred. Fluorescein is recognized as a cause of anaphylaxis in humans 5 and cats 6 when injected intravenously, and when used as an ocular surface stain in people. 7 In addition, ophthalmic proparacaine, chloramphenicol, vaccines, and their associated inactive ingredients were administered to these cats prior to anaphylaxis and have been associated with hypersensitivity in humans. 8–11 Although the role of these other compounds in our population is unclear, ophthalmic antibiotics were the only consistently administered drugs in this study and approximately half of all cats received only these drugs. Specifically, administration of an ophthalmic ointment containing oxytetracycline and polymyxin B (13 cats), or bacitracin, neomycin sulfate, and polymyxin B with (11 cats) or without (27 cats) hydrocortisone accounted for 51 (84%) of all reported cases. Further, polymyxin B was the only antibiotic present in all medications implicated in the study. At least three hypotheses might explain these observations: first, a range of topically applied ophthalmic antibiotics (acting alone or in various combinations) was responsible for the anaphylactic events; second, that one or more inactive ingredients were responsible; or, finally, that polymyxin B alone was responsible. Parenteral administration of polymyxin B is utilized in in vitro and in vivo studies to induce histamine and serotoninmediated increased vascular permeability.12,13 Additionally, polymyxin B is commonly associated with contact dermatitis in humans. 14 However, to the authors' knowledge, polymyxin B is not reported to be associated with anaphylaxis following topical ocular or dermatologic application in other species. By contrast, there are numerous reports implicating the combination of neomycin and bacitracin in immunologic responses, including anaphylaxis, in humans.15–17 Identification of specific allergens in humans has been performed using dermal skin testing or specific IgE assays, 18,19 but results of these tests were not done or reported for any cats in the present study.
Lack of known previous exposure to the incriminated ophthalmic antibiotic in all but 6/45 cats in this study warrants discussion. First, previous exposure may have occurred but have been unknown to, or not reported, by the owner or veterinarian. In particular, it is possible that some cats may have been exposed to antibiotics used in cell culture required for vaccine manufacture or through residues in food. Alternatively, cats may have experienced anaphylaxis due to pre-formed IgE directed at an antigen similar to the inciting antigen. Finally, although there is not a universally agreed definition of anaphylaxis, 4 in a recent review 20 ‘true’ anaphylaxis was defined as a type-I (immediate) hypersensitivity with a requirement for pre-formed immunoglobulin E antibodies, whereas non-immunologic anaphylaxis does not require preformed mediators. It is possible that some cases we report represent examples of non-immunologic anaphylaxis.
Although the diverse signalment of affected cats in this study fails to support any age, breed, or gender predilection, clinical signs during the anaphylactic event, treatment and clinical outcome were more consistent. This suggests that prediction of susceptible cats is impossible, but that early recognition and successful treatment of anaphylaxis in affected cats might be possible. In the present study, all but five affected cats showed signs within 30 min of receiving the ophthalmic antibiotic; and over half within 10 min. These data suggest that, although not always practical, consideration should be given to administering the first dose of an ophthalmic antibiotic to a cat while they are under veterinary supervision and, where possible, within a veterinary clinic or hospital where appropriate emergency therapy can be rapidly instituted. In the current study, long-term survival without noted sequelae was achieved with supportive care in 48/58 (83%) cats where outcome was described. In this study, cats typically were treated with a combination of intravenous fluids, oxygen supplementation, corticosteroids or antihistamines. Epinephrine was administered less frequently, and yet it is the treatment of choice for anaphylaxis in humans. 21 In particular, it is interesting to note that all cats which died did not have their eyes flushed, whereas all 27 cats that did have their eyes flushed survived. Although our study does not permit assessment of efficacy of therapies offered, it seems reasonable to recommend eye flushing as a benign and potentially helpful therapy for cats experiencing anaphylaxis following application of an ophthalmic preparation.
Recognition that antibiotic-containing ophthalmic preparations may not be predictably benign in all cats, and may be fatal in some, necessitates that their use is reserved for cases in which they are indicated. In 50 (86%) of cats in which an indication was provided in the present study, the ophthalmic antibiotic believed to have caused the anaphylactic event was administered for treatment of conjunctivitis, ‘ocular infection’, or intermittent epiphora; ocular lubrication; or ‘to facilitate eye examination’. Not all of these diagnoses or indications strongly support use of an ophthalmic antibiotic. For example, 76% of cats received the antibiotic for treatment of conjunctivitis or an ocular infection. The most common ocular surface pathogens of cats are feline herpesvirus, Chlamydophila felis, and Mycoplasma species 22 ; pathogens against which neomycin, polymyxin B, bacitracin, or gramicidin singly or in any combination are not effective. Although tetracycline antibiotics are indicated for treatment of feline conjunctivitis due to Mycoplasma species or C felis, tetracycline in combination with polymyxin was also associated with anaphylaxis in the present study. In addition, cats with C felis conjunctivitis respond better and sometimes only to systemically administered drugs.23–25 Taken together, these data from the current and previous studies regarding causes of feline conjunctivitis, their susceptibilities to topically applied ophthalmic antibiotics in common use and the potential for some of these drugs to — albeit uncommonly — induce anaphylaxis suggest that ophthalmic antibiotics incriminated in this study should not be used in cats with conjunctivitis unless a susceptible organism is known or at least strongly suspected to be the cause. Use of an ophthalmic antibiotic seems particularly unwise when the desired effect is simply ocular lubrication or prevention of desiccation, especially in cats under general anesthesia, when cardiovascular implications of anaphylaxis might be more difficult to detect and treat. This seems particularly important as polymyxin has been associated with neurotoxicity in several species, including cats, and even neuromuscular blockade.26–29 Although it is also possible that inactive ingredients within vehicle or preservatives may cause anaphylaxis, use of an ointment without antimicrobial activity seems rational when only lubrication or prevention of desiccation is required.
Despite the severity of signs observed in cases reported here, including in some instances death, it is noteworthy that only approximately half were reported to a regulatory agency or pharmaceutical company. The FDA CVM relies on veterinarians to voluntarily report adverse drug events to the FDA or to drug manufacturers. Manufacturers of drugs approved by the FDA for use in animals are required to submit to the FDA CVM information on adverse events reported to them by veterinarians or animal owners. The FDA CVM pharmacovigilance program monitors and evaluates these data and works with drug manufacturers to make appropriate label and/or packaging changes, draft ‘Dear Doctor’ letters, or develop risk management programs as necessary. Significant underreporting is recognized as a limiting factor to this process and this study supports that concern. Reasons for this may include the event not being recognized as a drug safety issue or a lack of familiarity with reporting processes. Detailed instructions for reporting adverse drug events may be found on the FDA Center for Veterinary Medicine website (http://www.fda.gov/AnimalVeterinary/SafetyHealth/ReportaProblem/ucm055305.htm).
Improved awareness of the potential for anaphylaxis in cats receiving certain ophthalmic antibiotics, limited use of these drugs except in patients where the chosen antibiotic is indicated, more close observation of cats following ophthalmic antibiotic administration, and improved event reporting will likely enable more rapid and specific therapy, and better highlight drugs of concern in future.
Footnotes
Acknowledgements
The authors gratefully acknowledge the assistance of veterinarians who provided survey responses for this study
Supplementary data
Supplementary data Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.jfms.2011.06.007.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
