Abstract
This study describes radiographic changes associated with mycobacterial infection in 33 domestic cats confirmed by culture or interferon-gamma testing. Infection was seen most frequently in adult (average age 5.7 years; range 1.5–12 years), non-pedigree (87%; 27/31), neutered male cats (69%; 22/32). The most common infections were Mycobacterium microti (60%; 18/30) and Mycobacterium bovis (37%; 11/30); Mycobacterium avium and Mycobacterium malmoense were infrequently cultured (3% of each; 1/30). Radiographs were available for the thorax (24 cats), abdomen (eight), appendicular skeleton (11) and head (three). Radiographic changes affected the thorax most commonly, consisting of bronchial (46%; 11/24), alveolar (38%; 9/24), nodular unstructured interstitial (38%; 9/24) or unstructured interstitial (25%; 6/24) lung patterns, which were often mixed. Perihilar or sternal lymphadenopathy were common (42%; 10/24), particularly perihilar lymphadenopathy (25%; 6/24). Skeletal changes were found in the distal antebrachium (three), pes (two), maxilla, scapula, spine, manus, femur, and tarsus (one each). Changes were typically osteolytic (73%; 8/11), often permeative osteolytic (64%; 7/11).
Osteoproliferative changes were seen in three cats and soft tissue swelling in five cats, which were adjacent to the bony abnormality in four cats. Other changes included submandibular soft tissue swelling, marked aortic, aortic root and brachiocephalic trunk calcification, and soft tissue swelling with calcification in the distal antebranchium which was not involving bone. Abdominal changes were uncommon (seen in 2/8 cats) and consisted of hepatomegaly and hepatosplenomegaly. In summary, radiographic changes were varied, no lesion was pathognomic for mycobacterial infection, and pathology was seen most commonly in the thorax.
Several species of mycobacteria can cause disease in cats. These include primary pathogens (tuberculous group mycobacteria such as Mycobacterium bovis or Mycobacterium microti)1–3 and saprophytes that can become pathogenic under certain circumstances (non-tuberculous mycobacteria [NTM] that are usually found in soil, water and decaying vegetation). 4 Depending on the route of infection, affected cats may present with clinical signs related to respiratory or alimentary tract disease, or localised lesions affecting the skin, lymph nodes or joints (which may later spread to cause pulmonary or systemic disease).1,5,6
Historically, over 95% of confirmed cases of tuberculosis in cats were caused by M bovis infection3,5,7 with only a few cases caused by Mycobacterium tuberculosis 8 and occasional cases caused by M microti.9–11 Occasional cases of systemic Mycobacterium avium infection were also reported.12–15 Most infections were believed to result from ingestion of milk from cattle with M bovis infection: the cats presented with alimentary lesions. With the reduction of tuberculosis in the national herd and pasteurisation of milk, this is no longer how mycobacteriosis usually presents in cats. Currently, in Great Britain, three quarters of cats with mycobacterial disease present with single or multiple cutaneous lesions and half of the cats have lymph node involvement, typically affecting the submandibular nodes. Systemic or pulmonary signs are seen less frequently. 16 Cutaneous lesions typically affect the ‘fight and bite sites’; such as the head and legs, and are believed to result most commonly from being bitten when playing with infected prey. In Great Britain, data from the Veterinary Laboratory Agency (VLA) shows that between 2005 and 2008 M microti was cultured from 19% of feline mycobacteriosis cases, M bovis from 15%, M avium from 7%, and non-M avium NTM from 6%; mycobacteria could not be cultured from over half of the cases (despite the samples having histopathological changes indicative of mycobacterial infection) (Tim Crawshaw, personal communication 2010). Unfortunately, many mycobacteria are very difficult to grow in culture media.
All members of the tuberculosis complex pose potential zoonotic risks. However, there have been no recently reported cases of cats passing tuberculosis onto humans. In contrast, M tuberculosis and M bovis can both cause reverse zoonoses and there have been a small number of cases where humans have infected their cats with M bovis. 16
Clinical signs of feline mycobacteriosis vary depending on the route of infection. Cutaneous infections can spread haematogenously to the lungs or extend locally to affect deeper tissues, bones or joints. When the infection spreads to the lungs, or where it is acquired through inhalation, lesions may arise in the lungs and/or hilar lymph nodes and affected animals can present with anorexia, dyspnoea, and a cough, which is typically soft and wheezy.3,5,9,10,17 Occasional cases develop pneumothorax or pleural fluid, and pericardial effusions have also been seen.2,17
The first radiographic report of feline mycobacteriosis was published in 1930, describing a case of pulmonary tuberculosis. 18 Since then radiographic changes have been described infrequently, and they have never been the focus of a scientific paper. The radiographic features of feline mycobacteriosis are not well known in the veterinary community and, therefore, it may not be considered as a differential diagnosis in cases with pertinent radiographic findings. The aim of this paper was to review the radiographs of a large number of cats with confirmed mycobacteriosis and to describe the range of radiographic changes that can occur.
Materials and methods
This radiographic study is a descriptive, retrospective case series. Radiographs of cats with confirmed mycobacterial infection were submitted to one of the authors (DGM) between 1992 and 2010. Inclusion criteria consisted of: (i) confirmation of mycobacterial infection and (ii) at least one radiographic image of diagnostic quality. To confirm mycobacterial involvement, aspirates and/or biopsy samples of affected tissue had been stained with Ziehl—Neelsen (ZN) and found to have changes suggestive of mycobacteriosis. 2,19 Tissue culture 20 or interferon-gamma testing had been used to identify which mycobacterial species were involved.21,22 All tissue culture was performed at the VLA, Weybridge, UK.
Radiographs were examined with knowledge of mycobacterial infection but without specific clinical information by a board-certified radiologist (TS). Image quality criteria were evaluated first to ensure sufficient diagnostic quality. Radiographs were reviewed for the following diagnostic criteria: osteoproliferative changes, osteolytic changes and soft tissue swelling for skeletal structures; abnormalities in the mediastinum, pleural space, pulmonary vasculature, cardiac silhouette, pulmonary parenchyma and thoracic boundaries for thoracic structures and evidence of organomegaly, serosal detail and appropriate abdominal organ opacities for abdominal structures. Lung abnormalities were described with pattern diagnosis according to standard terminology.23,24 The extent of disease was characterised as focal, local or generalised and the degree was ranked as normal, moderately or markedly increased.
Results
A total of 33 cats met the inclusion criteria of confirmed mycobacterial infection and diagnostic radiographs.
Thirty cases had mycobacterial infection confirmed on culture, and three were ZN-positive with concurrent positive interferon-gamma results consistent with infection with a member of the tubercle group. The most common infection was M microti, which was cultured in 60% (18/30) of cases. M bovis was cultured in 37% (11/30) cases, and M avium and Mycobacterium malmoense were each cultured in 3% (1/30) of the cases. The gender was not known for one cat. For the remaining cats, 69% (22/32) were neutered males and 31% (10/32) were neutered females. The breed was not known in two cases. Most of the cats were non-pedigree; 84% (26/31) were domestic shorthair cats, 10% (3/31) were Siamese and 3% (1/31) were domestic longhair cats. The age was not known for six of the cats. For the remaining cats the mean age was 5.7 ± 3.4 years (range 1.5–12 years).
Areas of the body for which radiographs were available included thorax (24), abdomen (eight), appendicular skeleton (11) and head (three). Twenty four cases had at least one thoracic radiograph which met the inclusion criteria and 21 cases had thoracic abnormalities. Radiographic changes affected the thorax most commonly. The most common lung pattern was a bronchial pattern, which was seen in 46% (11/24); 38% (9/24) had an alveolar pattern, 38% (9/24) had a nodular interstitial pattern and 25% (6/24) had an unstructured interstitial pattern (Fig 1). Four cats had an alveolar pattern as the only lung pattern, one had an unstructured interstitial pattern alone, four had a bronchial pattern alone and two had a nodular interstitial pattern alone. One cat had a bronchointerstitial pattern, two had a broncho-interstitial pattern and one had a nodular interstitial pattern. Five cats had alveolar, bronchial and nodular interstitial patterns and one had a nodular interstitial and bronchial pattern. Of the 24 cats with thoracic radiographs 42% (10/24) had either sternal or perihilar lymphadenomegaly. The most common finding was marked perihilar lymphandenomegaly (Fig 2), which was present in 25% (6/24) of cases.

Different radiographic lung patterns in different cats with mycobacteriosis. (A) Diffusely calcified bronchial walls and concretions (broncholithiasis) resulting in a bronchial pattern. Radiograph courtesy of Pam Meldrum. (B) Homogenous caudoventral lung opacification with air bronchogram (arrow) and cardiac border obliteration consistent with alveolar pattern. (C) Diffuse accumulation of multiple very small soft tissue nodules consistent with a miliary nodular pattern. Radiograph courtesy of Barbara Gallagher, University College Dublin. (D) Large pulmonary nodule (long arrow) and numerous smaller lung nodules, consistent with a nodular interstitial pattern. There is also marked perihilar (between short arrows) and sternal lymph node (arrowheads) enlargement. Radiograph courtesy of Paul Hanlon.

Marked perihilar lymphadenopathy in cat with mycobacteriosis. (A) Lateral thoracic radiograph depicting the deviated trachea (T) and mainstem bronchus (arrow) by the enlarged lymph node (Ln). (B) Corresponding post-mortem photographdepicting the enlarged caseous lymph node (Ln) cut free from the trachea (T). Lu = lung.
Eleven cats had radiographs available for assessment of musculoskeletal changes. Skeletal abnormalities were found at the following locations: distal antebrachium (three), pes (two), maxilla, scapula, ventral spine, manus, femur, and tarsus (one each). The most common changes were osteolytic lesions which were noted in 73% (8/11) of cases, which was most typically a permeative osteolytic lesion (64%; 7/11) (Fig 3). Osteoproliferative changes were noted in three cats and were characterised as smooth, irregular, or ‘sunburst- like’ (one each). Soft tissue swelling was present in five cats, which was adjacent to the bony abnormality in four of them. In one cat, there was only submandibular soft tissue swelling. One cat had marked aortic, aortic root and brachiocephalic trunk calcification (Fig 4), with concurrent marked soft tissue swelling with calcification in the distal antebranchium which was not involving the bone.
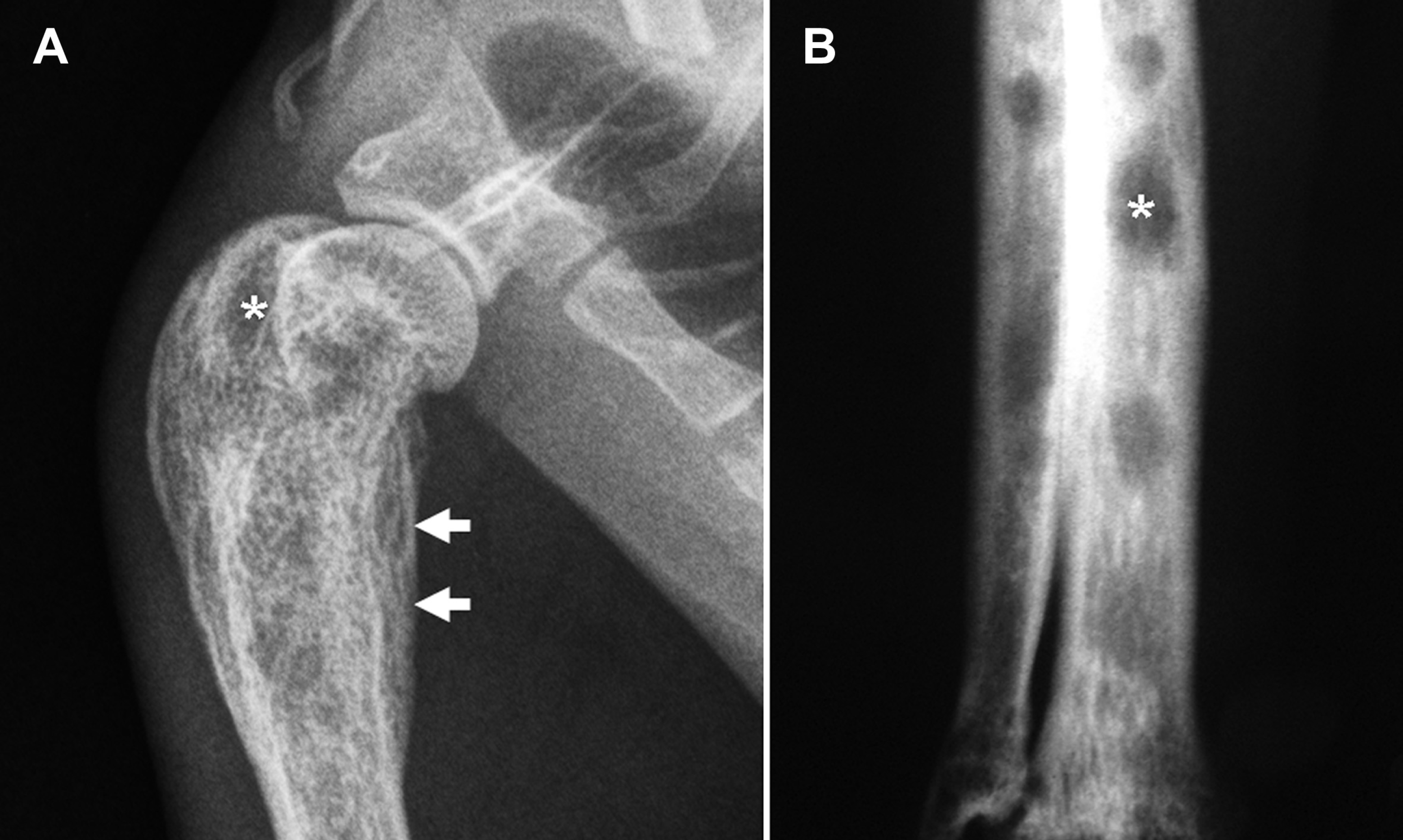
(A) Lateral radiograph of the proximal humerus of a cat with mycobacteriosis with poorly demarcated areas of osteolysis (*) consistent with permeative osteolysis and smooth periosteal reaction (arrows). Radiograph courtesy of Kelly Bowlt, Bristol University. (B) Relatively demarcated areas of osteolysis (*) in the distal antebrachium on a craniocaudal radiograph in a different cat.

Lateral thoracic radiograph depicting wall calcification of the aortic root (arrowhead), descending aorta (Ao) and brachiocephalic trunk (arrow) in a hypercalcaemic cat with mycobacteriosis.
Abnormalities on abdominal radiography were uncommon. Of the eight cases for which abdominal radiographs were available, abnormalities were noted in only two cases. One cat had hepatosplenomegaly and one had hepatomegaly alone.
Discussion
Infection was most commonly seen in adult (average age 5.7 years), non-pedigree (87%), neutered male cats (69%), and most cases were caused by either M microti (60%) or M bovis (37%). Radiographic changes were found most commonly affecting the thorax, consisting of bronchial (52%), alveolar (43%) or nodular interstitial (43%) lung patterns, that were frequently mixed. Perihilar and/or sternal lymphadenopathy were common (42%). Skeletal changes were less common and were typically osteolytic (73%). Occasional cases showed soft tissue calcification, hepatomegaly or hepatosplenomegaly.
Consistent with the cats in the current study, most cats with mycobacterial infection in Great Britain are known to be adult, neutered, male and non-pedigree. 25 It is interesting that 10% of the cats in the current study were Siamese as this breed has previously been suggested to be predisposed to mycobacterial infections, particularly M avium.12,13,15 However, from the current study it is not possible to confirm whether Siamese cats are predisposed to mycobacterial infections or whether this was coincidence.
For mycobacterial infections to cause radiographic changes there must be substantial local or systemic pathology. In cats, systemic infection is most commonly caused by M bovis or M microti infections,5,6,11,25–27 but may also been seen with M avium infection.12,28–30 NTM other than M avium (such as Mycobacterium fortuitum, 32 Mycobacterium thermoresistible, 31 Mycobacterium visibilis and Mycobacterium simiae 3 ) only rarely cause systemic disease.31–33 Reported radiographic features of these NTM include severe diffuse to patchy alveolar consolidation, which in one case was reported to be associated with prior lipid aspiration, 32 while dystrophic calcification was seen in another cat following a year of therapy. 31 Mycobacterium xenopi has been found causing mesenteric lymphadenomegaly in one cat 35 and a tracheal granuloma in another cat. 33
Radiography has previously been described as a potentially useful tool in the diagnosis of mycobacterial infection in cats. Thoracic changes with M bovis, M microti or M avium infections have been described as very variable, with changes including unstructured interstitial, miliary, nodular interstitial, bronchial or mixed-pattern lung infiltration, localised or generalised lung consolidation, tracheobronchial lymphadenopathy, or pleural effusion.2,11,16,25–27,29,30 Pulmonary and/or lymph node calcification has been reported only rarely, as has involvement of axillary lymph nodes. 12,25 While clinicians may be expecting to see large cavitating lesions as seen in humans or dogs with postprimary tuberculosis,34,36 these are rarely seen in cats. In fact, the only paper describing cavitating tubercles in cats was published in 1949. 5 Why these changes have not been seen since then is unclear. However, it probably reflects changing routes of infection; infection by inhalation may have been more likely to lead to this type of lesion and this route of infection is now believed to be exceedingly rare in cats. 6
Radiographic changes with M bovis, M microti or M avium infections have been reported less frequently in areas of the body other than the thorax. Nonthoracic soft tissue changes may include dystrophic or metastatic calcification, and enlarged retropharyngeal or popliteal lymph nodes.12,25 Abdominal changes may include hepatosplenomegaly, abdominal masses, calcified mesenteric lymph nodes, or ascites.25,28,29 Bone lesions tend to consist of areas of lysis and sclerosis, osteoarthritis, discospondylitis, vertebral osteomyelitis, or periostitis. Thoracic involvement may be associated with hypertrophic pulmonary osteopathy.11,25
The radiographic findings in the current study were similar to those described previously in case reports, small case series and reviews. However, while the broncho-interstitial lung pattern was described more frequently in the literature,2,11,25–27,29,31 we found rather more distinct bronchial, alveolar and nodular interstitial lung patterns. Factors that may be contributing to these differences may include differences in radiographic techniques, different classification systems of lung patterns or different strains of mycobacteria causing different patterns of lung pathology.
Aortic and other vascular calcification is very rare in cats and has been associated with chronic renal insufficiency 37,38 However, severe peribronchiolar unstructured interstitial mineralisation has previously been noted in a case report of a cat which had pyogranulomatous cystitis associated with Toxoplasma gondii infection after renal transplantation. 39 Hypercalcaemia has previously been reported in cats with mycobacteriosis 29 which may explain the bronchial wall calcification in one cat which was known to be hypercalcaemic and the marked vascular calcification seen in another cat in this study. The latter cat had an extensive clinical work-up which did not identify any renal disease that could have accounted for the vascular mineralisation.
Most radiographic changes seen in this study represent multi-systemic disease, with pulmonary infiltration, lymphadenomegaly and organomegaly. These changes are strongly suggestive of infiltrative disease but are not specific for mycobacteriosis. Potential differential diagnoses include neoplasia (lymphoma, mastocytosis, other infiltrative neoplasia), chronic inflammation (mycobacteriosis, feline infectious peritonitis, and systemic mycosis), hyperplasia (reactive lymph nodes), hypereosinophilic syndrome, and amyloidosis. 27 Mycobacteriosis should be considered when these types of changes are identified in feline patients.
There are a number of caveats to this study. The most significant of which was that although the mycobacterial infections were confirmed ante-mortem, histopathology was only available in one case. Organ specific radio-pathological confirmation would have been ideal, however, post-mortem examinations are rarely performed in cases of confirmed mycobacterial infections as they can only be performed at special facilities with category 3 pathogen clearance. Therefore, it is not possible to confirm that the radiographic changes seen were due to mycobacteriosis. Histopathology would have been helpful to gain more knowledge about the disease process and to explain more fully the radiographic changes seen. For example, why was a bronchial pattern the most common lung pattern? The retrospective nature of this study is another limitation, in that only the areas of clinical interest were radiographed and submitted. This leads to a bias in our results, and probably underestimates subclinical disease. In addition, radiography is not the optimal imaging modality to assess all organs, such as abdominal viscera, so some disease processes may be underreported. Another limitation is the difficulty in diagnosing NTM as they are notoriously difficult to culture. Due to the stringent inclusion criteria in this study, only two cats with these infections were included. Therefore, our study population was biased towards tuberculous mycobacteria as they grow more readily in the culture system used by the VLA.
It would be interesting to undertake a prospective whole body imaging study to accurately determine the extent of clinical and subclinical disease. Similarly a study in which clinical and radiographic findings are prospectively evaluated and correlated would enhance our knowledge on feline mycobacteriosis.
Footnotes
Acknowledgements
Stephanie Lalor's residency is supported by the Feline Advisory Bureau. The authors would like to acknowledge everyone from the University of Edinburgh and the University of Glasgow who have helped with these cases and to those who provided us with radiographs, including Professor R Malik; Drs D Addie, D Foster, B, Gallagher, K Bowlt, C Gibbs, D Gould, G Hunt, S Shaw, C Sturgess; G DeBolla, C Brown, R Davison, S Gillbert, P Hanlon, R Hilton, M Hollywood, A McKay, D McMahon, P Meldrum, S Mitchell, C Morganti, N Pennefather, C Stork, D Thomas, Mvan Baarle and G Wilkinson.
