Abstract
Pneumonectomy is the resection of all lung lobes from one side of the thorax. The clinical findings, treatment and outcome of four cases of feline chronic pyothorax managed with exploratory thoracotomy and pneumonectomy are reported. All cases were initially medically managed with thoracic drain placement and antibiosis. However, resolution was not achieved with medical therapy and diagnostic imaging findings consistent with an area of abscessation or marked lung lobe consolidation were identified, supporting a decision for surgical management. Surgical exploration was performed via median sternotomy and, on the basis of gross inspection, non-functional lung was removed. A left-sided pneumonectomy was performed in three cats and a right-sided pneumonectomy in one. All cases survived to discharge and an excellent quality of life was reported on long-term follow-up. Pneumonectomy appears to be well tolerated in the cat.
Pyothorax is an infection of the pleural space characterised by the accumulation of a purulent exudate. It is most commonly encountered in young cats, with a mean age of 4–6 years, and no breed or gender predisposition.1–3 The suspected major routes of infection include direct inoculation, aspiration of normal oropharyngeal flora and extension from an adjacent site of infection.1,4–8 It is well recognised that aggressive medical management, including broad spectrum antibiosis, pleural cavity drainage and lavage via indwelling thoracic drains and supportive care, is the first line approach to feline pyothorax and full clinical resolution can be achieved in the majority of cases.3,6,7,9
Surgical exploration of the thorax is indicated where diagnostic imaging has identified an area of marked consolidation or an intrathoracic abscess, and in cases where medical management has failed to achieve clinical resolution.3,9 The aims of the exploratory thoracotomy are to remove any primary cause, excise areas of necrotic tissue and pleural adhesions, lavage the entire pleural cavity, and ensure accurate positioning of thoracostomy tubes.5,9,10 The prognosis for resolving infection by thoracotomy is reported to be good to excellent; however, previous reports do not provide details of the surgical techniques performed.3,9
Pneumonectomy is the resection of all lobes in either the left or right lung field. Experimental studies in normal dogs have documented that right or left pneumonectomy is well tolerated in this species.11–19 However, to the authors' knowledge, there is only one previous case report in the veterinary literature of a cat undergoing pneumonectomy. 19 The pneumonectomy was performed for resection of a pulmonary squamous cell carcinoma and the cat was reported to make an uneventful recovery from surgery but died 89 days postoperatively, due to progressive heart failure.
This report describes four cases of feline chronic pyothorax which have achieved long-term resolution and a return to a good quality of life following exploratory thoracotomy, debridement and pneumonectomy after a failed response to aggressive medical management. The report documents that pneumonectomy of the left or right lung appears well tolerated in the cat and is compatible with a good long-term outcome.
Materials and methods
Records at the Queen Mother Hospital for Animals, Royal Veterinary College, London, from August 2003 to March 2009, were searched for cats that underwent pneumonectomy during management of chronic pyothorax. Data retrieved included: history, physical examination findings, diagnostic imaging findings, surgical procedures, perioperative complications, histopathology results, duration of hospitalisation. Long-term outcome was assessed by telephone questionnaire with the owner.
Results
Four cats were identified that had undergone pneumonectomy for management of chronic pyothorax (Table 1). All had undergone a period of medical management (range 8–37 days, median 27 days), including pleural cavity drainage via a thoracostomy tube or tubes and systemic antibiotic administration. On presentation, all cats were noted to be tachypnoeic with a restrictive respiratory pattern and dull or absent lung sounds on examination of one hemithorax. Case 4 had a thoracic wall abscess at the site of previous thoracostomy tube placement. Haematology and serum biochemistry analysis were performed in all cats preoperatively and consistently demonstrated a neutrophilia (median 16.9 × 109/l; range 15–26.5 × 109/l, reference interval (RI) 2.5–12.5 × 109/l) and decreased packed cell volume (median 20%; range 12–26%; RI 24–45%). Other parameters were unremarkable. Pleural fluid cytology revealed degenerate neutrophils with intracellular bacteria in all cats. Microbiology cultured no bacterial growth from pleural fluid in cases 1, 2 and 3 and Pasteurella species plus a profuse growth of mixed anaerobes were isolated in case 4.
Details of four cats that had undergone pneumonectomy for management of chronic pyothorax.

MN/FN = male/female neuter, DSH = domestic shorthair.
In all cats the decision to pursue surgical exploration was based upon diagnostic imaging findings identifying severely thickened pleura and lung lobe consolidation and/or a mass lesion within the pleural space (Figs 1—5), in addition to failure of prior medical treatment (Table 1). Surgical exploration was performed via a median sternotomy in all cats. Consolidated lung lobes and associated debris were removed, pleural lavage was performed and a thoracostomy tube was placed on the affected side (Portex, 14–16 Fr; Table 1).

Lateral thoracic radiograph of case 2 showing loss of definition of the cardiac silhouette and the ventral aspect of the diaphragm, consistent with a pleural effusion. There is a convex soft tissue opacity caudal to the cardiac silhouette (as indicated by arrows). The thoracostomy tube is visible.
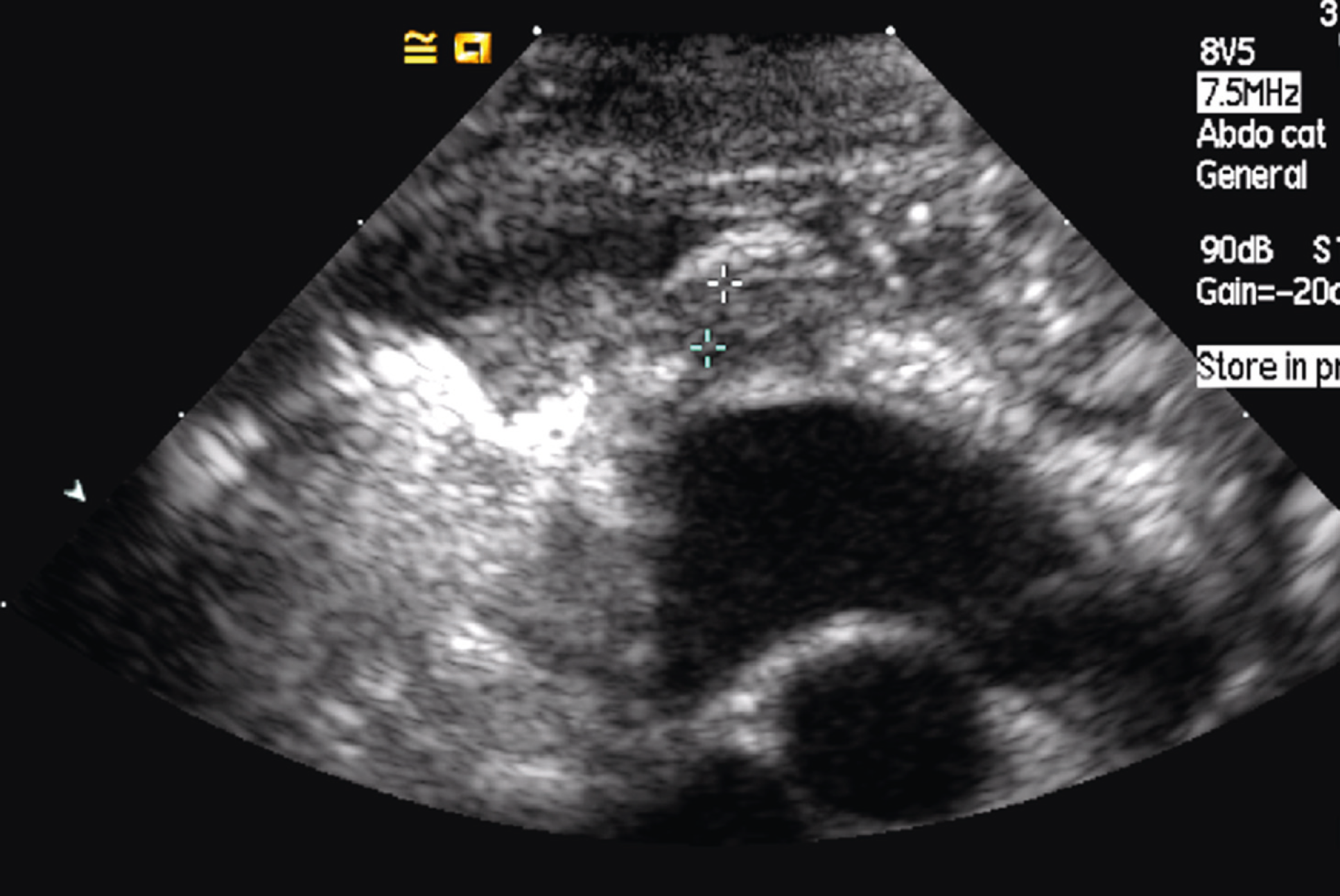
Thoracic ultrasonography of case 2; left parasternal view showing thickened pleural membrane (outlined by markers).

Dorsoventral radiograph of case 3 showing a marked soft tissue opacity of the left hemithorax, consistent with a pleural effusion. The left caudal lung lobe is positioned cranially and the left cranial lobe is not visible, suggesting a degree of left cranial lung lobe collapse. There is a right-sided mediastinal shift which may be a result of the left-sided pleural effusion. A 5 cm × 1 cm soft tissue opacity can be seen within the subcutaneous tissue lateral to the left seventh to 10th ribs which represents an abscess at the site of the previous thoracostomy tube.

Right lateral thoracic radiograph of case 3 shows a rounded soft tissue opacity caudal to the cardiac silhouette, which may represent an accumulation of fluid, an abscess or an area of lung consolidation (indicated by arrows).

Thoracic ultrasound image in case 3 demonstrating a suspected pleural abscess (as indicated by arrows).
The protocol for general anaesthesia included premedication with methadone (0.2 mg/kg IV) and induction of anaesthesia with alphaxalone (2 mg/kg IV, Alfaxan; Vetoquinol) or propofol (4 mg/kg IV, Vetofol; Norbrook), based upon clinician preference. Anaesthesia was maintained using intermittent positive pressure ventilation with isoflurane in oxygen. In all cats, continuous pulse oximetry demonstrated that the oxygen saturation remained at, or above, 94% throughout the procedure. Perioperative analgesia included the use of a continuous rate infusion of fentanyl (0.2–0.5 mg/kg/ min IV, Sublimaze; Janssen-Cilag) and a ropivacaine intercostal nerve block (1 mg/kg, Naropin; AstraZenica). Postoperative analgesia was provided with methadone (0.2 mg/kg IV q 4 h), followed by buprenorphine (0.01–0.02 mg/kg IV q 6–8 h, Vetergesic; Alstoe). Interpleural ropivicaine was delivered while the thoracostomy tube remained in place (1 mg/kg q 6 h). Perioperative antibiosis was administered at induction and q 2 h during surgery (cefuroxime 20 mg/kg, Zinacef; GlaxoSmithKline, or amoxicillin clavulanate 20 mg/kg IV, Augmentin; GlaxoSmithKline).
All cats received a typed whole blood transfusion; cases 1 and 3 received a transfusion perioperatively for management of intraoperative haemorrhage and cases 2 and 4 received a transfusion postoperatively (2 days and 4 days) in response to a fall in packed cell volume (nadir of 16% and 14%; RI 24–45%). Case 1 developed a pneumothorax 3 h postoperatively which was drained via the thoracostomy tube and did not reoccur. The thoracostomy tube was removed at 1–6 days postoperatively (median 4 days).
Histopathological analysis of excised tissue revealed diffusely collapsed lung tissue with pleural thickening and chronic active pyogranulomatous pleuritis in all cats. Microbiological analysis of excised tissue samples cultured no bacterial growth in cases 1 and 3, Pasteurella species and Pseudomonas species in case 2, and Pasteurella species in case 4.
Postoperative antimicrobial therapy was based upon culture and sensitivity profiles when bacterial isolates were yielded (enrofloxacin 5 mg/kg q 24 h or potentiated amoxicillin 20 mg/kg q 12 h). In cases 1 and 3 where no bacterial isolates were yielded, an empirical antibiotic regimen was chosen based upon clinician preference (clindamycin 5 mg/kg q 12 h, Antirobe; Pfizer or cefuroxime 20 mg/kg q 12 h and metronidazole 10 mg/kg q 12 h). Where available, intravenous preparations were used for the first 2–4 days postoperatively, followed by oral administration for an additional 14–20 days postoperatively.
Oesophagostomy tubes were placed in cases 1 and 3 at the time of surgery. All cats were inappetent for 2–4 days postoperatively, but regained a moderate appetite by the time of discharge. Duration of hospitalisation ranged from 7–10 days (median 10 days). All cats were noted to have a normal respiratory rate and effort at the time of discharge. On follow-up questioning of the clients, all cats were considered to have made an excellent recovery. All cats returned to ‘normal’ activity levels and enjoyed an indoor/outdoor lifestyle. No abnormalities were noted with respect to breathing pattern or exercise tolerance. At the time of follow-up, cases 2, 3 and 4 were alive; 776, 437 and 295 days post-surgery, respectively. Case 1 was euthanased 762 days postoperatively following treatment for a traumatic diaphragmatic rupture.
Discussion
Pneumonectomy, or removal of all lung tissue from one side of the thoracic cavity, requires isolation, ligation and transection of the vasculature and main bronchi to the affected lobes. 20 In this report a standard pneumonectomy technique was used via a median sternotomy incision, and a thoracoabdominal stapling device or manual sutures were used to achieve ligation of the hilar vessels and bronchi. In the veterinary literature interrupted horizontal and continuous suture patterns have been used for closure of the bronchial stump. Additionally, the tip of the bronchial stump is often oversewn for added security against air leakage.21,22 Stapling devices are used by approximating the edges of the bronchial stump and the associated vessels, and applying the staples transversely, thus allowing simultaneous vessel and bronchus ligation and, hence, significantly reducing surgical time. Stapling devices are widely reported to be safe and effective, and are associated with minimal perioperative complications.23–28 Both ligation and surgical staples were used effectively in this report and both techniques had an excellent long-term outcome. Individual ligation of the pulmonary artery, vein and bronchi was performed in case 2 due to space limitations within the thoracic cavity resulting in problems positioning the head of the thoracoabdominal stapling device.
Pneumonectomy in humans is associated with relatively high perioperative morbidity and mortality. Complications of the procedure include air leakage and haemorrhage, which require re-operation if severe. 29,30 In this case series, one cat suffered from a pneumothorax following surgery but this resolved promptly with drainage via the indwelling thoracostomy tube. All four cats received a whole blood transfusion, two on recovery following significant intraoperative haemorrhage, and two in response to a fall in the haematocrit postoperatively. All four cats were suffering from a normocytic, normochromic anaemia at the time of referral which may reflect a nonregenerative anaemia of chronic disease. In addition, significant intraoperative haemorrhage occurred in all cats during disruption of extensive pleural adhesions. Following blood transfusion, all cats showed a stable haematocrit and no further intervention was required.
Other commonly reported complications following pneumonectomy in humans include respiratory insufficiency, bronchopleural fistulae, pneumonia, pulmonary oedema, congestive heart failure due to exacerbation of pre-existing cardiac disease, and oesophageal dysmotility and dilation secondary to mediastinal shift.29–31 No such complications were recognised in this case series. Case 1 represented with a diaphragmatic rupture more than 2 years following discharge. During the 2 years the cat had regained pre-surgery exercise levels and had an apparently normal respiratory rate and effort.
Following the surgery each case remained within the intensive care unit for between 3 and 8 days. Careful monitoring, oxygen supplementation, analgesia, fresh whole blood transfusion and chest drain management were all key to the successful postoperative recovery of each cat.
Acute loss of up to 50% of the lung is well tolerated and following pneumonectomy compensatory changes in the contralateral lung occur via two separate mechanisms; recruitment of existing alveolar-capillary reserves and remodelling of the remaining lung structure with new alveolar growth.13–19,32 Presumably with a prolonged disease course the compensatory mechanisms are already taking effect. Surgery to remove non-functional lung tissue and restrictive adhesions should facilitate effective expansion of the normal lung. Additionally the diseased lung may have been acting as a shunt, which, when removed, allows a greater proportion of the cardiac output to reach functional lung tissue so improving oxygen saturation.18,19
Experimental research in the dog has demonstrated that the left and right lungs account for 42 and 58% of the lung volume, respectively. 14 According to the relative contribution to total lung volume, there have been reservations regarding right-sided pneumonectomy; previously right-sided pneumonectomy was not recommended due to the likelihood of fatal pulmonary hypertension following resection of more than 50% of the lung volume. 20 However, experimental trials have shown that despite the increased pulmonary vascular resistance following pneumonectomy, the pulmonary arterial pressure remains within the RI. 12 Additionally, it has been demonstrated that animals undergoing a right-sided pneumonectomy show a greater amount of compensation in the form of regenerative alveolar growth, which is largely unseen in left-sided pneumonectomies.15,33 Case 2 reported here underwent a right-sided pneumonectomy and made a good recovery. Such success has been demonstrated in other case reports and experimental trials, but an increased risk of postoperative complications has been associated with right-sided pneumonectomy compared with left.12,13,19
Limitations of this case series include the low case numbers and the extended time period over which the cases were managed, with case management varying at the discretion of the attending clinician. Arterial blood gas analysis would ideally have been performed pre- and postoperatively and at the time of follow-up. This would have allowed a more accurate assessment of respiratory function and also of the development of compensatory mechanisms over time. This was, however, not performed perioperatively due to technical challenges related to patient size and could not be justified long term as there were no clinical indications for further intervention.
Conclusion
Surgical management is appropriate in cases of feline chronic pyothorax when diagnostic imaging modalities document extensive pulmonary parenchymal change despite pleural drainage. With extensive thoracic pathology necessitating right or left pneumonectomy a satisfactory postoperative and long-term outcome can be achieved. A high level of postoperative care is required but an excellent level of respiratory function and return to pre-surgery exercise levels is possible. Right- or left-sided pneumonectomy is tolerated in the cat.
