Abstract
A 4-month-old intact male domestic shorthair cat was evaluated for urinary outflow obstruction after several weeks of medical management for traumatic urethral rupture. Positive-contrast retrograde urethrography and anterograde cystoscopy performed 4 weeks after the initial urethral injury confirmed a stricture approximately 1 cm distal to the bladder trigone at the site of the initial urethral tear. A self-expanding metallic urethral stent (SEMS) was placed under fluoroscopic guidance to relieve the urethral stricture and re-establish luminal patency. After stent placement, the cat was able to void urine normally with minimal urinary incontinence noted. This resolved several months post-stent placement. No known clinical complications persisted other than mild intermittent hematuria.
Urethral stricture is an uncommonly reported cause of urethral obstruction in the cat and may result from chronic urethritis or trauma, including catheter or urethrolith-induced injury, or as a congenital anomaly. 1,2 Invasive and non-invasive management of a urethral stricture may be challenging, particularly in small feline patients. Traditional treatment options include surgical urinary diversion (ie, transpelvic or perineal urethrostomy) or urethral resection and anastomosis. This case report describes the first time a urethral stent was used to treat a cat with urinary tract obstruction secondary to traumatic urethral stricture.
A 1.89 kg, 4-month-old male intact domestic shorthair cat was referred for evaluation of stranguria and ischuria that developed after approximately 4 weeks of routine medical management for a traumatic urethral tear. The cat was initially evaluated at the referring veterinarian's hospital immediately after incurring vehicular trauma. Lower urinary tract injury was suspected, and a polypropylene urethral catheter was placed transurethrally into the urinary bladder in a routine fashion under general anesthesia. Moderate resistance to placement was noted. The catheter was removed approximately 12 h later. Shortly thereafter, a urethral obstruction developed with an associated uroabdomen. An exploratory laparotomy was performed, and a proximal urethral tear was identified. Medical management was attempted via maintenance of an in-dwelling 3.5 Fr red rubber urinary catheter over a 2.5-week period. Multiple attempts at urinary catheter removal (both intentional and inadvertent) resulted in persistent re-obstruction. Culture of the urine obtained via cystocentesis 25 days after the initial urethral injury, prior to referral, yielded an Escherichia coli infection.
At this time the patient was referred to our hospital. On physical examination, a small and thickened urinary bladder was palpated. An in-dwelling 3.5 Fr red rubber urinary catheter was in place. Hematological analysis revealed leukocytosis (white blood cell count 28.9×103/μl, reference interval (RI) 4.2–15.6 103/μl) characterized by neutrophilia (18,785/μl, RI 2500–12,500/μl), lymphocytosis (7225/μl, RI 1500–7000/μl), monocytosis (867/μl, RI 0–850/μl) and eosinophilia (2023/μl, RI 0–1500/μl). Serum biochemical abnormalities included elevated alkaline phosphatase (ALP 90 U/l, RI 0–62 U/l), hyperphosphatemia (phosphorous 8.5 mg/dl, RI 3.0–7.0 mg/dl), mild hyperkalemia (K 5.8 mEq/l, RI 3.9–5.3 mEq/l), and creatine kinase elevation (CK 556 U/l, RI 64–440 U/l). The cat's urine specific gravity was 1.033. The cat was seronegative for feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV).
Abdominal ultrasonography revealed an irregular, diffusely thickened urinary bladder wall (0.83 cm in thickness [normal 0.13–0.17 cm]). 3 There was no evidence of pyelectasia or hydroureter. The cat's mesenteric lymph nodes were prominent (0.6 cm), and there was a minimal amount of peritoneal effusion that was an insufficient volume to sample. Thoracic radiographs were within normal limits.
Positive-contrast retrograde urethrography was performed under general anesthesia using digital radiography to determine the cause of the urinary obstruction and to allow planning for the placement of a self-expanding metallic urethral stent (SEMS), if necessary. The in-dwelling 3.5 Fr red rubber urethral catheter was replaced with a 5 Fr red rubber catheter. This was met with moderate resistance in two regions of the proximal urethra. The urinary bladder was distended with 1:1 mixture of sterile saline (0.9%) and iohexol (240 mg/ml). The patient was positioned in lateral recumbency, and a measurement catheter (Marker Diagnostic Catheter, Infiniti Medical, Malibu, CA) was placed in the colon in the same horizontal plane as the urinary catheter to account for radiographic magnification and allow for accurate measurement. Subsequent forceful injection of the contrast solution into the urethra revealed two areas of narrowing starting 1.7 cm caudal to the trigone (see Fig 1). There was no contrast leakage from any segment of the urethra or the urinary bladder. The 5 Fr red rubber urinary catheter was reinserted after the radiographic study and attached to a closed system to enable ongoing drainage of urine from the urinary bladder. The cat was recovered from anesthesia while an appropriate sized SEMS was ordered and obtained. The cat was hospitalized and treated with Normosol-R (4 ml/kg/h IV) and enrofloxacin (5 mg/kg IV) based on antimicrobial susceptibility testing.

Positive-contrast retrograde urethrography. A severe urethral luminal narrowing is observed 1.7 cm caudal to the trigone followed distally by a 1.3 cm length of irregular dilation of the urethra and a second 7 mm length of luminal narrowing 3.1 cm caudal to the trigone. A measurement catheter with 1 cm radiodense marks was placed in the colon to account for radiographic magnification and allow for accurate measurement.
Twenty-four hours after antimicrobial treatment was initiated, the cat was re-anesthetized and positioned in dorsal recumbency for SEMS placement. A limited (1.5 cm) ventral median celiotomy was performed to expose the urinary bladder. An endoscopic port (Ternamian EndoTip, Karl Storz Endoscopy, Goleta, CA) was inserted into the urinary bladder through an approximately 4 mm stab incision. 4
Anterograde cystourethroscopy was performed with a 1.9 mm rigid cystoscope (Telescope 30° 1.9 mm, Karl Storz Endoscopy, Goleta, CA) that was introduced into the urinary bladder through the port, revealing hyperemic and thickened bladder mucosa. Approximately 1 cm caudal to the bladder trigone, the urethra was severely narrowed, consistent with the proximal urethral stricture observed on the contrast study, preventing further advancement of the cystoscope (Fig 2).

Anterograde cystourethroscopy. Severe urethral narrowing 1 cm caudal to the bladder trigone, consistent with a urethral stricture.
A 0.035 inch diameter, angled, floppy-tip hydrophilic polymer coated nitinol guidewire (Weasel Wire, Infiniti Medical, Malibu, CA) was advanced in a retrograde direction through the urethral catheter and into the port, resulting in through-and-through guidewire access. The urinary catheter was removed over the wire, and a 4 Fr vascular access sheath was advanced retrograde over the wire and into the urinary bladder. With fluoroscopic guidance, a retrograde contrast cystourethrogram was repeated as previously described to confirm the length of the urethral obstruction (35 mm) and the diameter of the urethra adjacent to the stricture site (5.5 mm). A measurement catheter placed adjacent to the cat in the same horizontal plane as the urethra was again used to confirm previous measurements.
A 6 mm×40 mm self-expanding nickel–titanium alloy (nitinol) urethral stent (Vet Stent-Urethra, Infiniti Medical, Malibu, CA) was selected. The stent delivery system was advanced through the bladder access port in an anterograde direction over the wire and positioned across the urethral lesion using fluoroscopic guidance. The stent was then appropriately deployed (Fig 3).

Postoperative lateral radiograph revealing successful urethral stent deployment and placement. Slight compression of the stent is noted caudally, in the region of the most distal stricture.
Anterograde cystourethroscopy was repeated after withdrawal of the stent delivery system. The proximal end of the stent was observed immediately caudal to the bladder trigone (Fig 4). Manual expression of the cat's bladder yielded no resistance immediately after urethral stenting. Voluntary urination without assistance occurred within 3 h of surgical recovery. The cat was discharged on enrofloxacin (5.9 mg/kg [1/2 of 22.7 mg tablet] PO q 24 h for 2 weeks) and meloxicam (0.005 mg/kg PO q 24 h for 2 weeks).
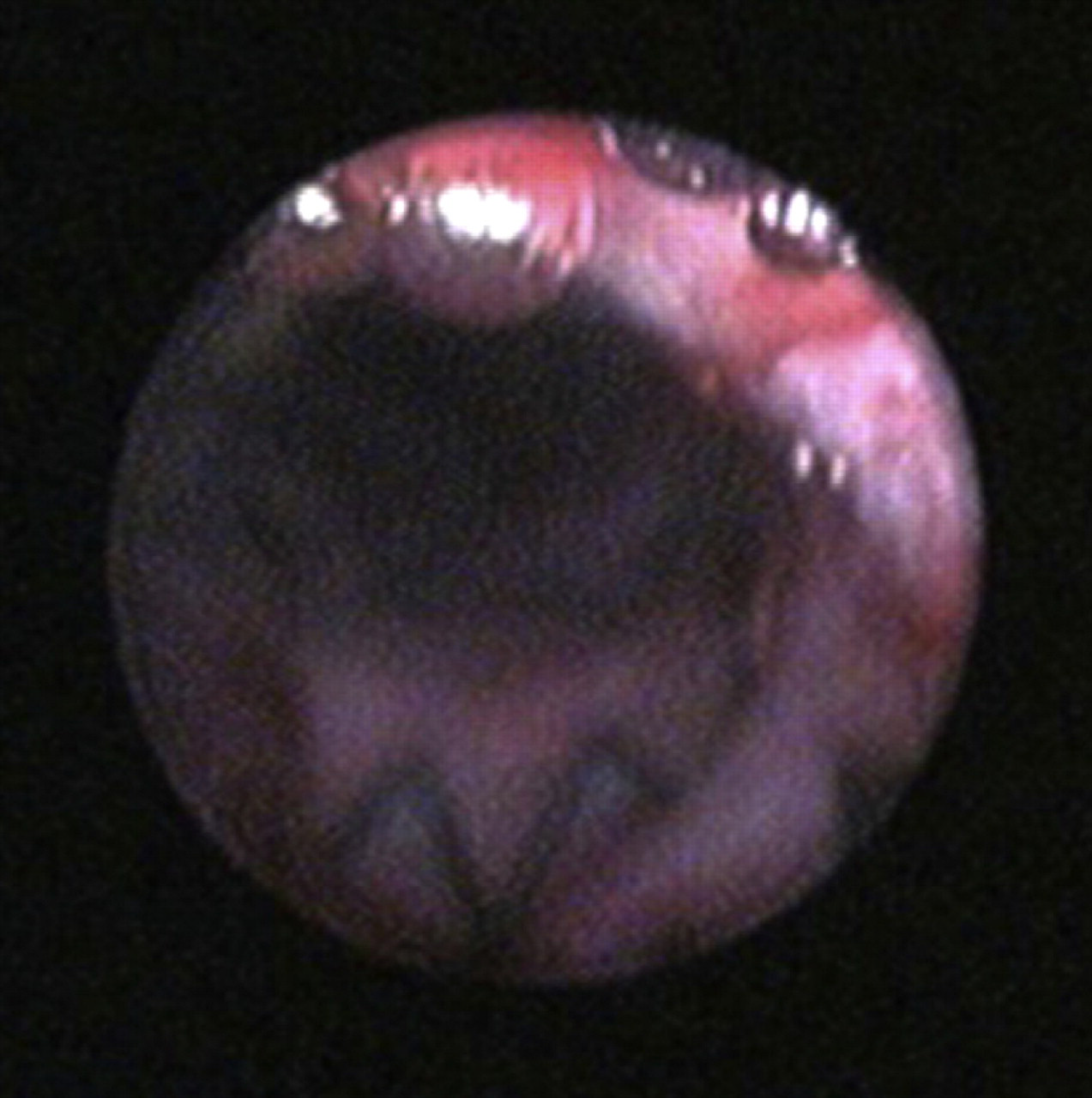
Anterograde cystourethroscopy post-SEMS placement. Luminal patency was immediately re-established post-urethral stent placement.
To date, 6 months after stent placement, the cat remains active with normal urinary flow and mild, intermittent hematuria. Mild urinary incontinence was reported 2 months post-stent placement, manifested as normal urine voiding with a good stream, but with intermittent episodes of small-volume urine dribbling. However, complete urinary continence was reported and observed at re-evaluation 6 months postoperatively. Follow-up radiographs obtained at 2, 3 and 6 months post-stent placement revealed no evidence of stent migration or fracture (Fig 5). Multiple urine cultures as late as 6 months post-stent placement were negative for bacterial growth.
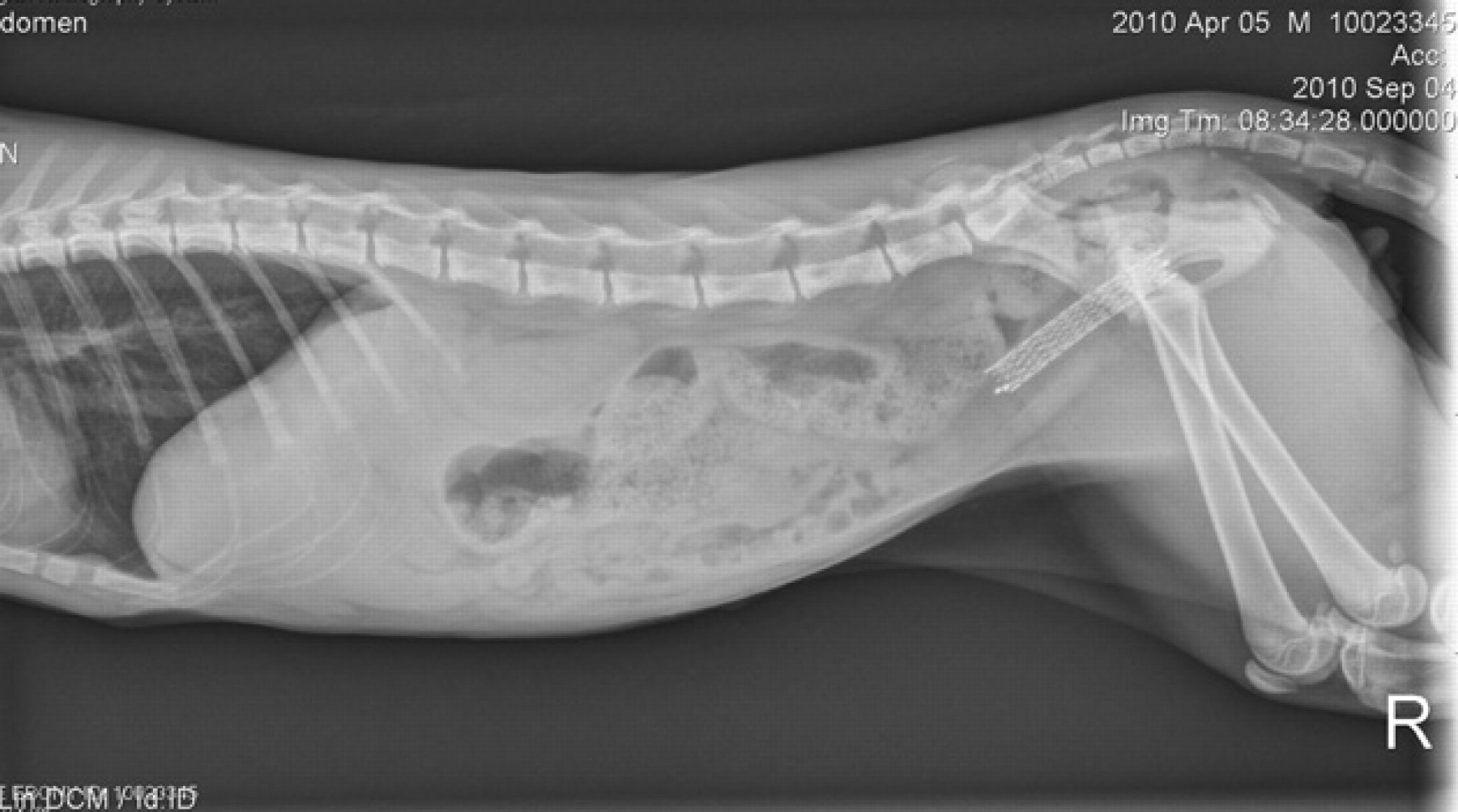
Follow-up lateral radiograph 2 months postoperatively. There is no evidence of stent migration or deformation. The previously noted slight compression at the caudal aspect of the stent has resolved.
With appropriate urinary diversion, the risk of clinical stricture formation at the site of traumatic urethral injury is reported to be low as long as a longitudinal strip of urothelium remains intact across the injury site. 5–10 Temporary urinary diversion may be achieved via urethral catheterization or tube cystotomy. If there is incomplete diversion of urine from the lesion and/or disruption in mucosal continuity, intraluminal retraction of the mucosa into a cuff of periurethral tissue at the injury site may result in fibrosis, scarring and contraction with subsequent urethral obstruction. 5,9,11–14 Urethral dilation proximal to the stricture site is a possible sequela to stricture formation and may promote further inflammation and fibrosis in the affected region. 14,15 In this cat, urethral dilation was observed proximal to the more distal stricture. The clinical manifestations of urethral stricture are typically not present until 4–6 weeks after urethral injury and may include ischuria, stranguria, and/or pollakiuria. 5,13,14
This is the first case reported, to the authors’ knowledge, describing the use of a urethral stent for the treatment of trauma-induced urethral stricture. While stent placement under fluoroscopic guidance to treat malignant urethral obstruction in dogs is becoming increasingly more common, there are very limited reports of the use of urethral stents in cats. 16–18 Compared to primary urethral repair (ie, urethral anastomosis), the use of a stent is less invasive and more rapidly performed, and as such, may be associated with a lower risk of peri-operative morbidity and/or mortality, including surgical dehiscence, urine leakage or stricture. In circumstances where the etiology of a urethral stricture is unknown, urethral resection and anastomosis may provide the advantage of gross and histological examination of the resected tissue. However, in cases, such as the cat in this report, where the clinical history is compatible with a benign etiology, there may be no added benefit to traditional surgical repair.
Both balloon-expandable metallic stents (BEMSs) and SEMSs have been used for management of malignant urethral obstruction in dogs. 16 An SEMS was chosen for the cat in this case report since, compared to BEMSs, SEMSs have increased flexibility and elasticity and are more resistant to deformation in regions subject to external compressive forces. 16,17,19 Although some SEMSs have been reported to undergo foreshortening when deployed, the SEMS used in this cat was a laser-cut nitinol (nickel–titanium alloy) stent, which has moderate radial force and expansile strength, good flexibility, and does not undergo foreshortening, allowing for precise placement. 16,19 In male cats, unlike female cats, the placement of a SEMS requires an anterograde approach (unless a perineal urethrostomy has previously been performed) because the small diameter of the penile urethra cannot accommodate the relatively large pre-deployment diameter (0.2 cm) of the SEMS delivery system.
Determination of the appropriate stent diameter is extremely important for a successful outcome. Previous studies in dogs have suggested that urethral stents should ideally be equal to or no greater than 1.3 times the maximum luminal diameter. 16,20 If the diameter of the stent is too small and adequate mucosal apposition is not achieved, the risk of stent migration increases. Alternatively, if the stent diameter is too large, mucosal edema and inflammation may develop. 16,20 For the cat described in this report, the maximum diameter of the dilated urethral segment cranial to the stricture was 5.5 mm, and a stent that was 6 mm in diameter was used. The largest diameter urethral segment lies within the prostatic urethra. In adult cats the prostatic urethral diameter has been reported to average 4.5–5.0 mm. 21 As the cat described in this report continues to mature, stent migration could occur as the urethral diameter enlarges, though the strictured region is unlikely to significantly increase in diameter and in fact may be acting as an anchor for the stent.
Although approximately 75% of this cat's urethra was stented, including 1 cm of urethra proximal to the stricture and 8 mm distal to the stricture, only minimal postoperative urinary incontinence was observed. Previous reports have speculated that the striated muscle of the distal feline urethra is able to generate high enough urethral pressure to maintain continence in cats even if, as in this cat, pre-prostatic and/or prostatic portions of the urethra are stented. 17,18 This cat's outcome also supports recent evidence in dogs that stent length and the percentage of the urethra that is stented may not be correlated with post-stent incontinence. 22
The urethral orifices in the cat are more caudally positioned than in dogs. 23,24 Therefore, placement of a urethral stent across a proximal urethral lesion, adjacent to or extending into the trigone, does carry the potential risk of urethral obstruction. However, this complication has not been previously reported, and there was no evidence of post-stent urethral obstruction in this cat on follow-up ultrasound examination.
As multiple urine cultures as late as 6 months postoperatively were negative for bacterial growth, the mild, intermittent hematuria that was noted early after stent placement was likely secondary to persistent cystitis, urethritis, and/or stent irritation. Anterograde cystoscopy/urethroscopy could be performed to confirm this suspicion but was not considered to be indicated as the cat was otherwise urinating normally.
No major complications developed in association with the SEMS placement in the cat described in this report, and the cat appears to be continent. Our findings suggest that placement of a urethral stent may be a viable alternative to surgery in some cats with urethral strictures, particularly if their clinical history is consistent with urethral trauma and secondary stricture formation. In instances where the lesion is further distal in the urethra, the outcome may not be as favorable. Additional studies and longer-term follow-up are warranted to further evaluate the use of SEMSs for benign urethral strictures, including their use in immature cats.
Footnotes
Acknowledgements
David Feldman DVM, DACVIM for reviewing the manuscript; the hospital staff of Advanced Critical Care, Culver City, California.
