Abstract
The clinical follow-up of four cats presumptively diagnosed with pseudomembranous cystitis is described. All presented with acute urine outflow obstruction and acute renal failure. Urethral catheterisation was performed without difficulty but ultrasonography and contrast radiography consistently revealed abnormal bladder wall and content. One cat was euthanased, the remaining three underwent an exploratory cystotomy. Abundant inflammatory and necrotic tissue covering an ulcerated bladder mucosa was removed. All cats recovered uneventfully. No definitive cause was identified but the clinical course of the disease was not typical of idiopathic cystitis.
In human medicine, pseudomembranous cystitis is defined by the presence of intravesical shaggy layers of necrotic material covering a haemorrhagic and ulcerated bladder mucosa. 1 This condition is not well-described in the veterinary literature. The clinical reports of four cats presumptively diagnosed with feline pseudomembranous cystitis are outlined here.
Case reports
All four cats presented with lethargy, anorexia, pollakiuria and dysuria of several days duration. See Table 1 for detailed information about each individual case. This was the first known episode of feline lower urinary tract (FLUT) disease for two cats whereas recurrent bouts were described for the two others. Three cats had initially been taken to their attending veterinarian but worsened despite treatment. On admission to the Veterinary Teaching Hospital (VTH) of the National Veterinary School of Toulouse, all cats exhibited a firm and painful bladder along with severe clinical depression and dehydration. Plasma biochemistry revealed azotaemia and profound hyperkalaemia. Correction of fluid and electrolytes disturbances was undertaken immediately. Cystocentesis consistently failed to decompress the bladder or, in two cats, to allow urine collection. Urinalysis, when performed, revealed non-specific abnormalities and the urocultures remained sterile. Plain radiographs were unremarkable except for bladder overdistension. Conversely, abdominal ultrasonography revealed bladder wall thickening and unusual hyperechoic septa dividing the lumen into compartments (Fig 1). These findings were confirmed by double contrast cystography in one cat (Fig 2).

Ultrasonographic aspect of the bladder of cat 3 at admission. Note the echoic septa dividing the bladder lumen into compartments.
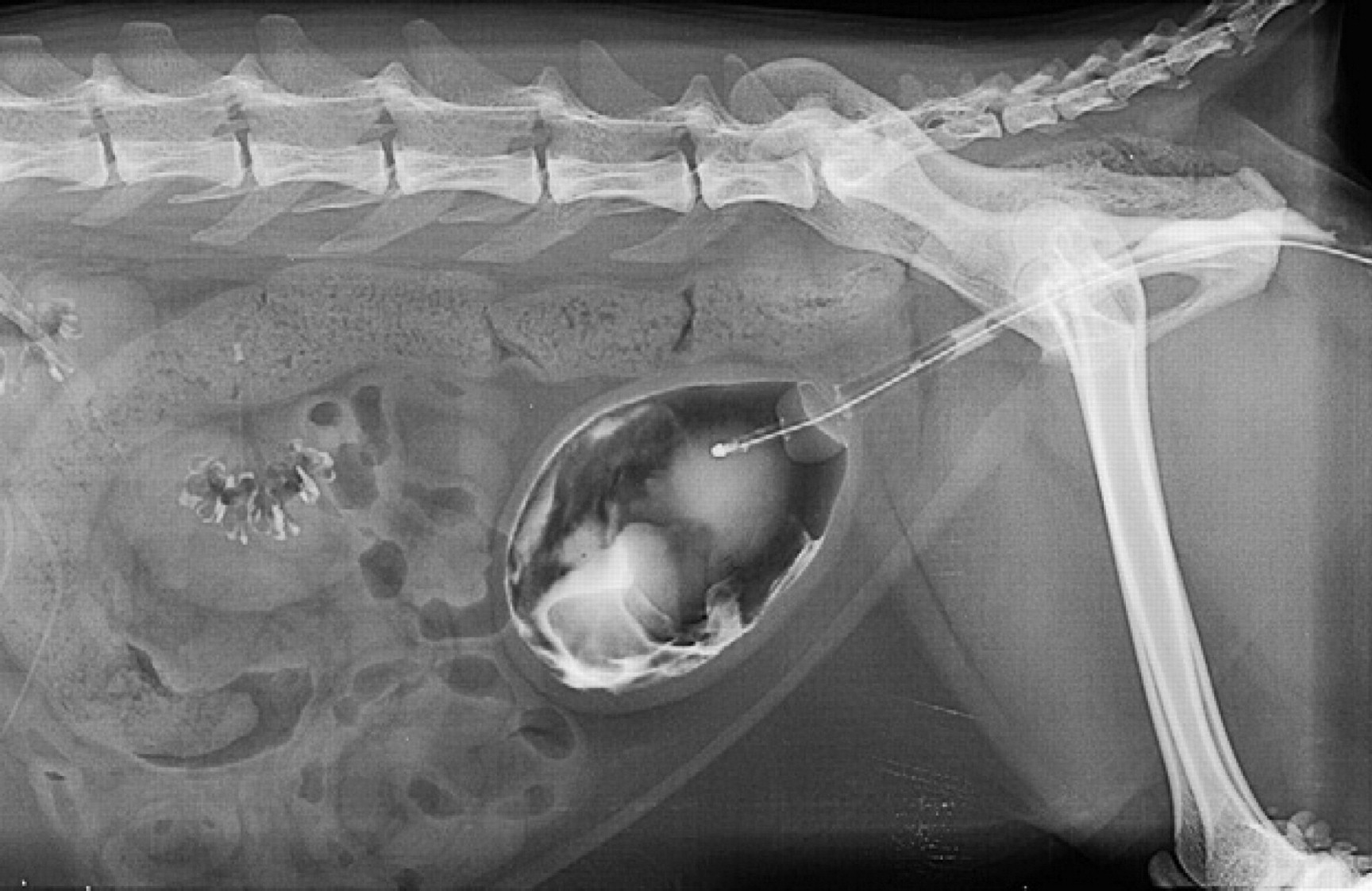
Double contrast cystography performed on the same cat 3. Note the abundant intravesical material in continuity with the mucosal surface of the bladder wall which appears thicker than normal.
Findings from four cats with pseudomembranous cystitis.


M = male; FLUTD = feline lower urinary tract disease; NM = neutered male; NP = not performed; PNN = polymorphonuclear neutrophils; RI = reference interval; USG = urine specific gravity; DSH = domestic shorthair.
Indwelling urethral catheterisation was performed under sedation or anaesthesia in all the cats without difficulty. The urethral catheter failed to collect urine from one cat although its correct placement and patency were checked by radiography and repeated flushing, respectively. The owners declined cystotomy, requested euthanasia and post-mortem examination was performed subsequently. An exploratory cystotomy was performed in the three other cats. The appearance of the bladder, whether per operative or at necropsy, was similar in all cats and consisted of a thickened wall and large pieces of fibrino-haemorrhagic tissue adhering to the mucosal surface (Fig 3). The resected material and bladder wall biopsies were examined microscopically. The bladder contents consisted of a fibrinous exudate with multiple blood clots, extensive necrotic remodelling, suppurated foci (Fig 4a) and occasional focal mineralisation. The bladder mucosa presented with severe and diffuse ulceration, necrosis and haemorrhage (Fig 4b). These changes were considered evocative of a feline counterpart of human pseudomembranous cystitis.
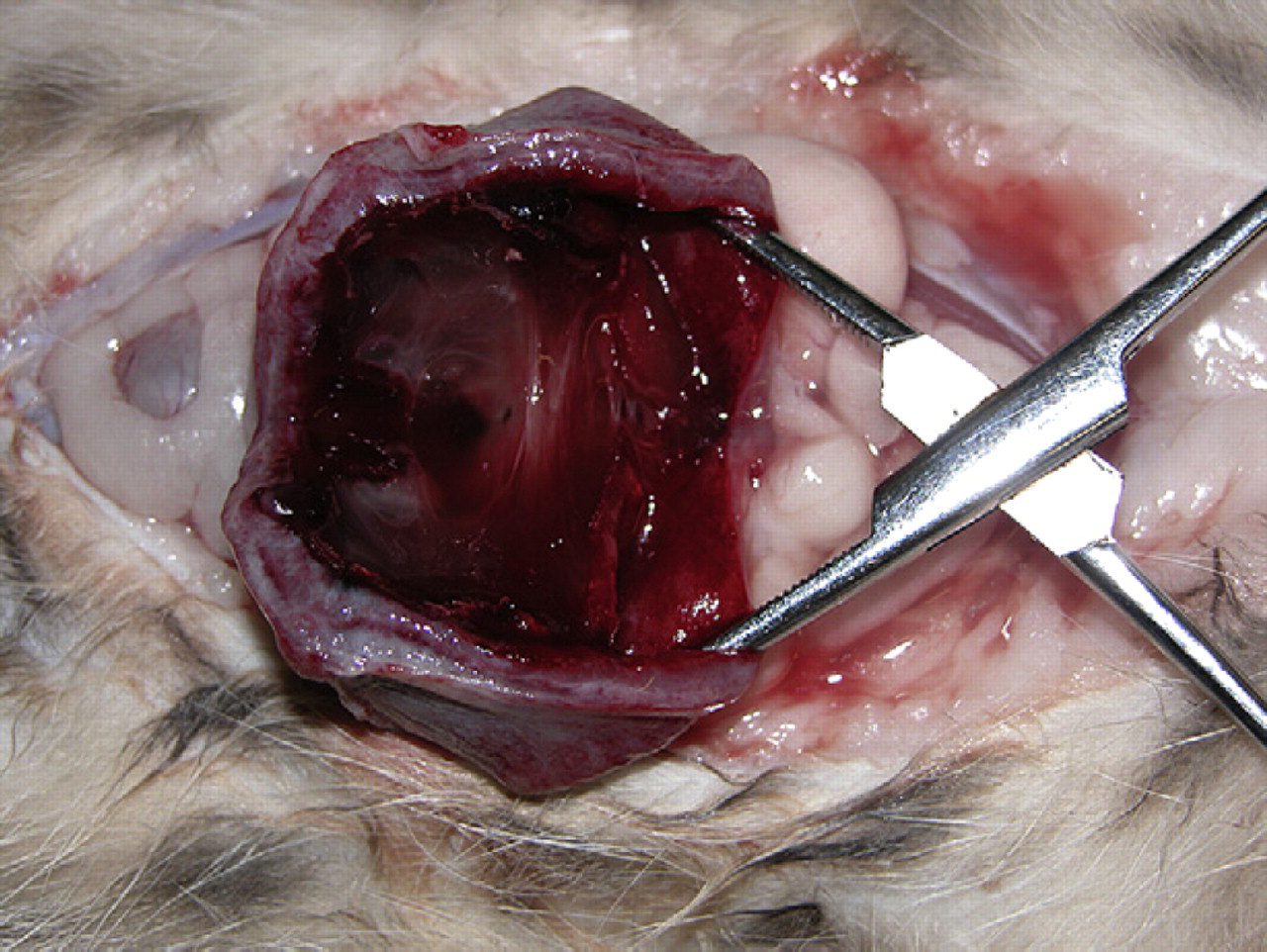
Necropsic aspect of the bladder of cat 4. The bladder has been cut along its long axis and the pseudomembranous intravesical content is apparent. Note the thick bladder wall.

Histopathological examination of intravesical material and bladder wall biopsies from cat 1. (a) Material surgically removed from the mucosal surface of the bladder wall. Haematoxylin and eosin stained ×100, fibrino-haemorrhagic clot with a few degenerated leukocytes. (b) Full-thickness bladder wall biopsy. Haematoxylin and eosin stained, ×10, the bladder mucosa is diffusely ulcerated and the lamina propria is markedly enlarged due to acute aedema and haemorrhage. The inner muscularis is focally haemorrhagic and degenerated.
The three cats recovered uneventfully. Urine was subjected to bacterial culture on removal of the urinary catheter and yielded bacterial growth for 2/3 cats. All were discharged a few days after cystotomy, with a course of antibiotics. A soft maintenance diet was recommended but no specific environmental changes were advised. One month after discharge, the clinical examination, urinalyses, plasma panels and ultrasonographic examination of the bladder were normal. One cat was subsequently lost to follow-up while the two others showed no further urinary signs 3 and 1.5 years after discharge, respectively.
Discussion
The four cases described here unambiguously suffered from urine outflow obstruction and post renal azotaemia but differed markedly from the usual cases seen in our hospital. Indeed, the presence of abundant organic material in the bladder lumen led to striking imaging features and required tailored management strategies. Ultrasonographic features of bladder lumen content in FLUT disease cases typically consist, at most, of echogenic urine with a swirling effect due to the presence of cells or high protein content. 2 In contrast, ultrasonographic examination consistently revealed numerous hyperechoic strips, resembling membranes, in the bladders of all the cats in this series. This, and the failure of conventional management to reliably restore urine outflow prompted the decision to perform a cystotomy for diagnostic and therapeutic purposes. Histopathological examination revealed major and unusual inflammatory changes, ie, layers of fibrinous and necrotic tissue covering a haemorrhagic and ulcerated mucosa. These changes appeared consistent with sloughing of necrotic areas of the bladder wall into the bladder lumen. 3 Despite this apparent severity, all the treated cats recovered uneventfully and remained healthy. Cystotomy may thus have contributed to the successful outcome. Surgical removal of the intravesical material, in particular, was thought to be pivotal for restoring urine outflow but it cannot be stated that these cases could not have been managed otherwise. Non-steroidal anti-inflammatory drugs and antibiotics could also have had a positive influence on the outcome of cystitis.
Although cases of feline cystitis with distinctive, unusual characteristics have already been described, the four cases presented here do not fit any of these descriptions. 4–7 The most similar condition, irrespective of species, would be human pseudomembranous cystitis. 1,8,9 This latter is indeed characterised by the presence of membranes of cell debris, fibrin, inflammatory cells and blood. Once removed, this material exposes an underlying haemorrhagic ulcerated mucosa, highly evocative of the histological lesions of the bladder wall described here. 1
In humans, pseudomembranous cystitis can be of various origins. The main one seems to be infectious (Streptococcus pyogenes, Str viridans, Staphylococcus pyogenes, Bacillus coli, B typhosus, B proteus). 8,9 In our study, the positive urocultures obtained from samples drawn after urinary catheter removal were thought to be of iatrogenic origin. Urine for microbial cultures was only available initially from 2/4 cats and proved to be negative. However, both cats had received prior antibiotherapy from their attending veterinarians. Moreover, some bacterial species do not grow easily on the culture media used for routine uroculture. Storage and transport to the laboratory, although limited to a few hours, could also have biased the results. Therefore, a bacterial aetiology cannot be fully excluded based on these negative urocultures. Moreover, all treated cats received antibiotics, irrespective of their last uroculture results, which could have cured any pre-existing infection and account for the observed absence of short term relapse.
Human pseudomembranous cystitis may also be caused by drugs, notably cyclophosphamide. 1 Various solutions are sometimes used during urethral catheterisation in cats and could have caused local chemical irritation. However, this was not the case in the VTH and was not reported by the attending veterinarians.
Interestingly, over-distension of the bladder has also been reported as a cause of pseudomembranous cystitis in humans, probably through ischaemia of the mucosa. 8,10,11 It cannot be concluded, from our observations, whether cystitis was the cause or consequence of the obstruction. As urethral catheterisation was straightforward in all cases, the obstruction could have been caused by sloughed portions of necrotic mucosa at the bladder neck. However, urethral patency was not assessed other than by unproblematic catheterisation and the possibility that pseudomembranous cystitis was a consequence of some degree of urethral obstruction with secondary bladder overdistension and pressure necrosis of the bladder wall cannot be excluded either.
Conclusion
Veterinarians should be alerted to the possible occurrence of pseudomembranous cystitis in cats as this condition poses unique problems in the management of feline urine outflow obstruction. Other reports and studies are, however, needed for a better insight into the aetiology and physiopathology of this poorly described condition.
