Abstract
A 17-month-old cat presented for frequent swallowing, occasional cough and stertor. Radiographic evaluation revealed a soft tissue mass at the level of the soft palate. Oral examination revealed a soft tissue mass within the soft palate. Surgical resection was performed. Histopathology was consistent with a large developmental cyst. Following surgical management the cat has remained clinically free of signs for 2 months.
The soft palate forms the floor of the nasopharynx and the roof of the oropharynx. It is a mobile, valve-like structure that separates and protects the proximal airway during swallowing as well as closing off the oral cavity during nose breathing. The importance of this separation is highlighted by the fact that the mucous membrane of the dorsal surface of the soft palate is of respiratory type, while digestive type is present on the ventral surface. 1 Because of this structures important role in protecting the airway, any disease process that causes inflammation, swelling or damage to the linings may impede not only swallowing but breathing as well. Although pharyngeal masses with similar clinical signs have previously been described in cats, to the authors’ knowledge there have been no published reports of a soft palate cyst in a cat.
A 17-month-old indoor only female spayed domestic shorthair cat was presented for evaluation of frequent swallowing and sonorous breathing with occasional cough. The owner reported that the stertorous sounds were worse when the cat was sleeping and had been present and stable since the time of adoption 6 weeks prior. No sneezing or nasal discharge was reported and the cat exhibited a good appetite at home. There was no report of exercise intolerance or respiratory distress. The cat had been previously treated for presumed pneumonia by the referring veterinarian with amoxicillin, marbofloxacin, prednisolone and lysine, without improvement in clinical signs.
The cat was bright and alert upon presentation. Initial physical examination was within normal limits except for a grade 2/6 focal systolic murmur and a soft upper respiratory noise on inhalation. A limited non-sedated oral examination was performed and was unremarkable. Non-sedated orthogonal thoracic radiographs were obtained and revealed a mild to moderate bronchial pattern. The cardiac silhouette was considered to be normal. A single lateral cervical radiograph was obtained and revealed an abnormal soft tissue opacity in the caudal pharyngeal region that appeared to be associated with the caudal aspect of the soft palate (Fig 1). Differential diagnoses for this soft tissue opacity included soft palate mass, nasopharyngeal polyp, foreign body or neoplasia. An echocardiogram was performed due to the presence of the murmur and revealed no structural heart disease. A complete blood count, chemistry panel, thyroid panel and urinalysis were performed and were within normal limits. A feline leukemia and feline immunodeficiency antibody test were both negative.
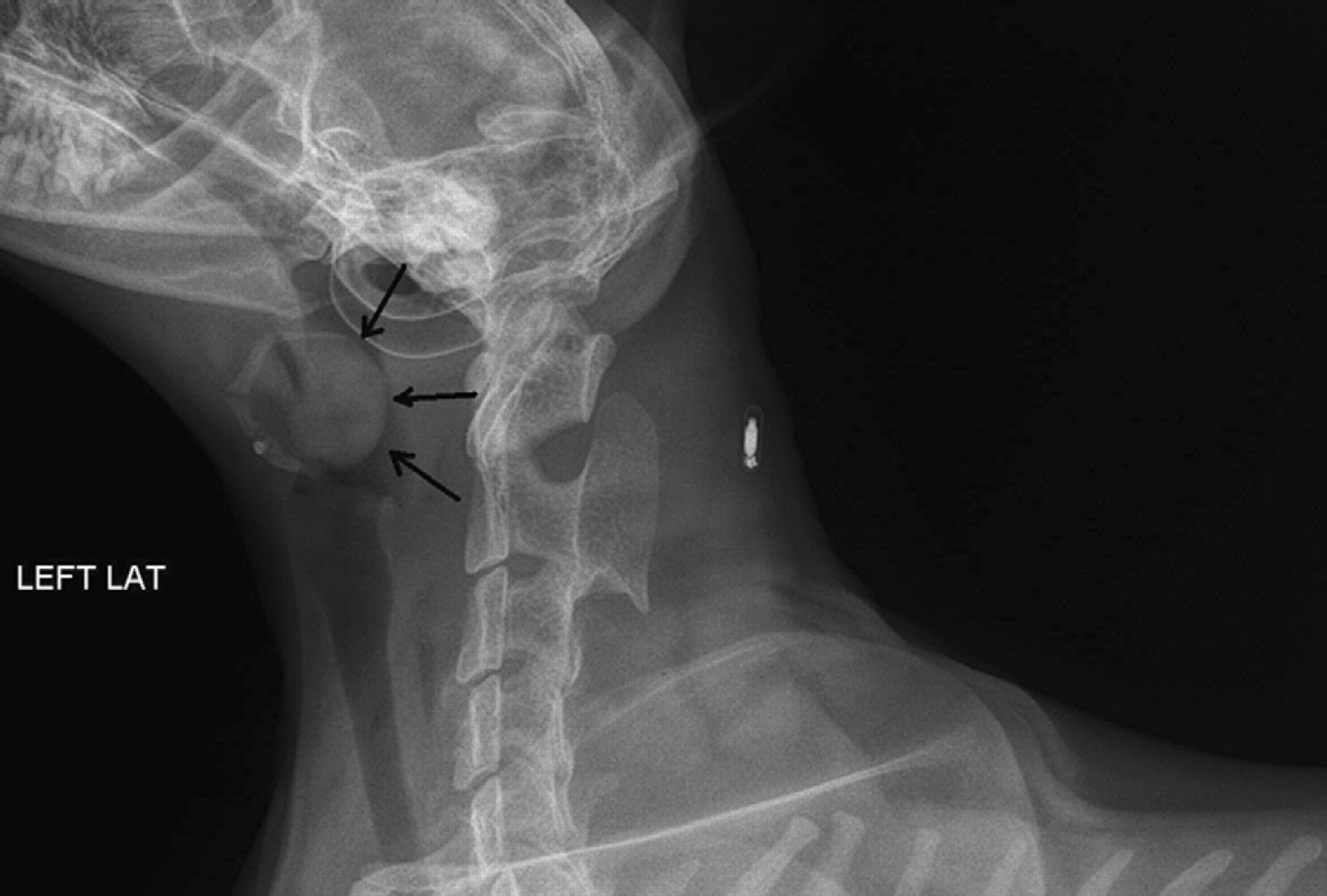
Lateral cervical radiograph reveals a soft tissue opacity associated with the caudal aspect of the soft palate (arrows).
The following day the cat was anesthetized for a complete oral examination with propofol (5 mg/kg IV) and maintained on 1.5–2% isoflurane in oxygen, intravenous fluid support was provided at a rate of 40 ml/h (10 ml/kg/h). The examination revealed a firm mass approximately 2 cm×1 cm within the dorsal and ventral mucosal surfaces of the soft palate. The ventral mucosa of the soft palate had no evidence of ulceration, damage, erythema, or mass lesions. No other abnormalities were found in the oropharyngeal region except for a mildly enlarged left pharyngeal tonsil. Biopsy or excision of the mass was recommended. The cat recovered uneventfully from this procedure and surgery was scheduled for the following day. The following day the patient was premedicated with buprenorphine (0.015 mg/kg IM), and was induced with diazepam 0.5 mg/kg IV and propofol 4 mg/kg IV. Anesthesia was maintained with 1–2% isoflurane in oxygen and intravenous fluid support was provided at a rate of 40 ml/h (10 m/kg). The patient was placed in sternal recumbency, a mouth gag was placed and tape was placed under both upper canines to aid in visualization of the soft palate. The caudal pharynx was packed off with moistened gauze and a stay suture of 3-0 Monocryl was placed at the ventral aspect of the soft palate for retraction. The ventral mucosa of the soft palate was transected with metzenbaum scissors which revealed a well circumscribed green, encapsulated mass. The mass was bluntly and sharply dissected from the soft palate in its entirety (Fig 2). The visible dorsal aspect of the soft palate appeared edematous and was removed along with the mass. The mucosal defect was closed in one layer with 4-0 Monocryl in a simple continuous pattern. The remaining soft palate reached the mid tonsillar crypts bilaterally. The mass and soft palate mucosa were submitted for histopathologic review. Recovery from anesthesia was unremarkable. Dexamethasone SP (0.1 mg/kg IV) was given to help prevent postoperative swelling. There was an immediate improvement in stertor. Post-surgically the patient was treated with buprenorphine (0.01 mg/kg IV q 8 h). The patient was discharged the following day with tramadol (3 mg/kg PO q 8 h as needed). At recheck examination 14 days later, the owner reported complete resolution of clinical signs at home. A non-sedated oral exam was performed and no obvious abnormalities with the soft palate incision were seen.

Cross section of the cyst following surgical resection. Note the proteinaceous material within the cyst.
Biopsy results revealed mild mucosal hyperplasia with submucosal edema and focally extensive degenerate/necrotic cystic material. No inflammation was observed in the unaffected submucosa or mucosal tissue. No evidence of malignancy or infectious agents was observed within the specimen. The necrotic proteinaceous material found within the mass could have been associated with abscessation but because there was no evidence of inflammation more likely corresponded to a large retained developmental cyst with accumulation of secretory proteinaceous material.
Pharyngeal cysts have been previously described in dogs, horses and humans. 2–5 These cystic structures are typically detected in young patients and the etiology of the condition is suspected to be sequestration of the ectoderm during closure of the embryonic fissure. 5 Cystic structures of the soft palate specifically have only been described in two horses. 6 They are speculated to develop during fusion of the palatine plates which grow from the maxillary process to the midline to form the palate although the specific etiology is unknown. 5 There has been no similar report of soft palate cysts in feline patients.
Cystic structures of the soft palate appear to be rare, however, nasopharyngeal polyps are not uncommon and can occur in both cats and dogs and their treatment has been discussed in detail. 7,8 They usually arise from the auditory tube or middle ear and are characterized by granulation tissue covered by stratified squamous or ciliated columnar epithelial tissue. 9 Although a definitive inciting cause has not been identified, an important distinguishing characteristic of nasopharyngeal polyps is the inflammatory nature of the condition. The case described here is distinct from nasopharyngeal polyp due to lack of inflammation, location of the mass and the presence of a cystic rather than granulomatous structure.
Due to the rarity of this condition in both veterinary and human medicine, treatment recommendations have not been established. Surgical treatment for nasopharyngeal cysts in a dog has been previously described in the veterinary literature. 2 Therefore, there is a precedent for surgical management. Because of the important anatomic location of the soft palate, masses that grow within it or diseases that cause inflammation or swelling could cause difficulty breathing as well as with swallowing food, as was seen in this case. During swallowing, the soft palate works in conjunction with the epiglottis to allow a bolus of ingesta to cross the respiratory tract. An important point whenever surgical resection of the soft palate is undertaken is that a radical resection can lead to difficulty protecting the airway during swallowing leading to aspiration pneumonia. 10 Although this patient was not suffering from a life-threatening occlusion of the pharynx yet, it is possible that if the cyst had continued to grow, clinical signs could have worsened. Surgical excision was performed in this case and was a clinically curative treatment. If complete excision is achieved there should be an excellent prognosis for the patient if this was truly a congenital cyst.
To the authors’ knowledge, this is the first reported case of an oropharyngeal cyst associated with the soft palate in a cat. Although unusual, benign cysts of the soft palate should be considered among the differential diagnoses for young cats demonstrating signs of frequent swallowing with stertor. The cysts appear to be amenable to surgical excision.
