Abstract
Introduction
A number of different Mycobacterium species can infect pet cats. 1 Dermal nodules and nonhealing draining ulcers are reported in cats with infections involving the Mycobacterium tuberculosis complex (MTC; Mycobacterium bovis, M tuberculosis or M microti), M avium—M intracellulare complex (MAC), M lepraemurium and an increasing number of non-tuberculous mycobacteria (NTM). 2,3 Identification of the causative agent is important because of potential zoonotic risks, as well as for prognosis and for optimal treatment selection. The clinical signs and histological features may help to suggest one or other mycobacterial species, but clinical signs can overlap widely and, for a definitive diagnosis, culture and/or molecular analysis is needed. 1
Clinical report
A stray, approximately 6-year-old, female domestic shorthair cat (4.6 kg body weight) was presented in July 2006 for investigation of significantly enlarged mandibular lymph nodes (1.2 and 1.6 cm in diameter) and a firm, indolent mass (3 × 3.6 cm in diameter) on the left mandible that had been noticed by the owner 1–2 weeks earlier. The cat had been adopted in May 2006 by the owner, and its exact age and its history prior to adoption were unknown. The mass was infiltrating the soft tissue of the external side of the left mandible and extending into the oral cavity, where it had multiple fistulous tracts in the area of the caudal mandible. The cat was otherwise in good general condition, with a normal appetite, and no other abnormalities could be detected.
Haematology, blood biochemistry and electrolytes revealed only an increase in serum globulin concentration (64 g/l; reference interval 28–51 g/l). Feline leukaemia virus (FeLV) and feline immunodeficiency virus (FIV) tests were negative. Skull radiographs showed a soft tissue swelling surrounding the mandibular body without infiltration of the bone. The mass on the chin was biopsied and histopathological evaluation revealed pyogranulomatous inflammation within which rare acid-fast bacilli were identified (using Ziehl-Neelsen [ZN] stain), which suggested a mycobacterial infection. Thoracic radiographs (laterolateral and ventrodorsal) showed no abnormalities.
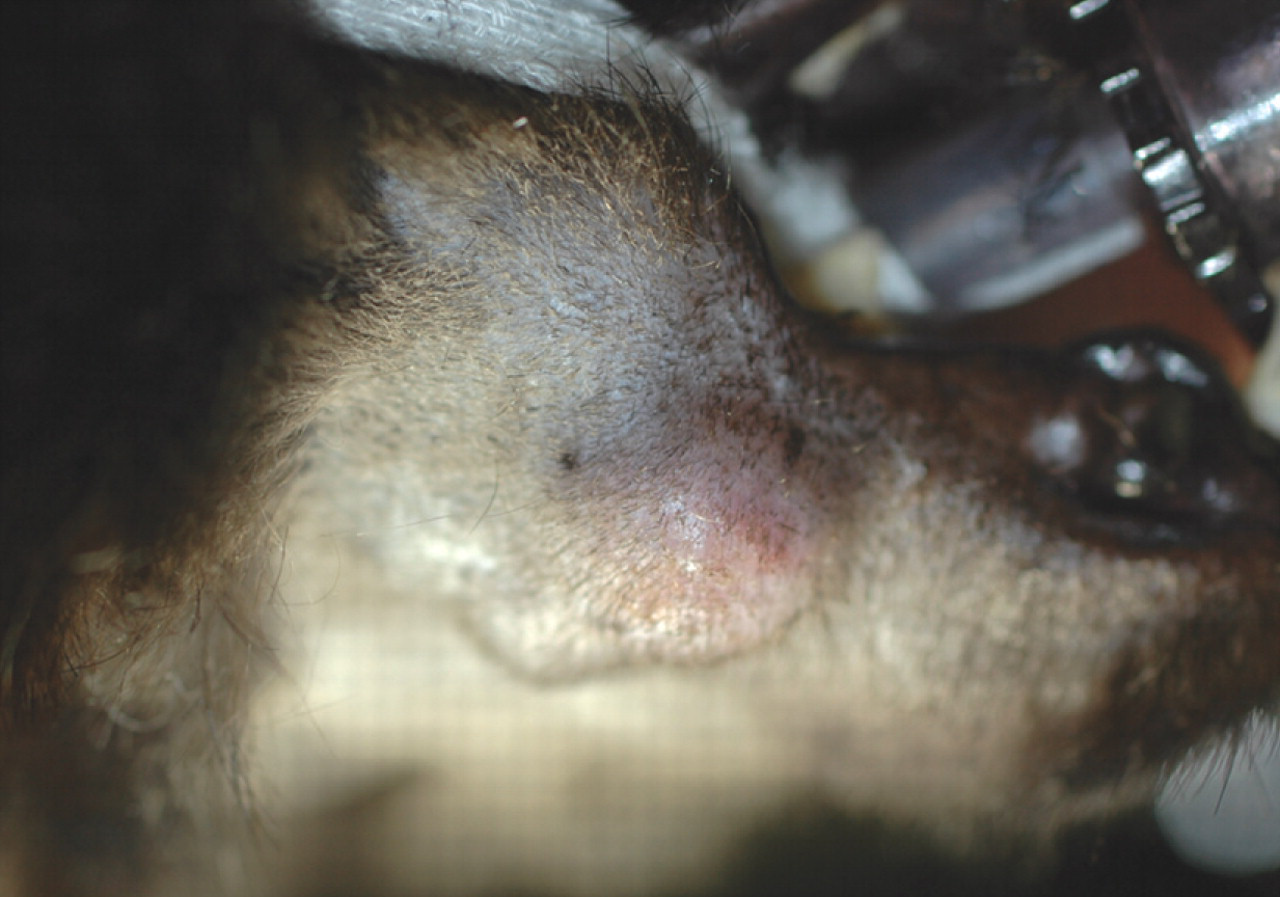
Mass on the left mandible (area is shaved), at the time of biopsy in July 2008

Deep dermal and subcutaneous granulomas. Haematoxylin & eosin stain, bar = 400 μm
Therapy with doxycycline (Doxycat; Biokema, Switzerland, 20 mg orally [PO] q24h) and enrofloxacin (Baytril; Bayer AG, Germany, 50 mg PO q24h) resulted in complete remission of the mass and the mandibular lymph node enlargement within 4 months of treatment. Therapy with both antibiotics was continued for a further month following clinical resolution.
In May 2007, when the cat was presented for annual vaccination, the lymph nodes and the mandibular region were normal on palpation.
Six weeks later, however, a subcutaneous mass in the area of the left mandible was detected and subsequently completely removed by surgery. Histology again revealed granulomatous panniculitis, but this time no acid-fast bacilli could be detected. Bacterial culture was not performed and no antimicrobial treatment was given.
The right mandibular lymph node became enlarged in October 2007, and three or four firm confluent swellings appeared in the November in the left mandibular region. In February 2008, doxycycline (20 mg PO q12h) and enrofloxacin (25 mg PO q24h) were re-introduced and again given for 5 months, but failed to produce any improvement. The mandibular mass (now 0.7 cm in diameter) and the enlarged lymph node were biopsied a third time in July 2008 after the antibiotics had been stopped for 13 days (Fig 1). Serum biochemistry and electrolytes were reassessed as a pre-anaesthetic check and found to be unremarkable. Thoracic radiographs again revealed no abnormalities.

Epithelioid macrophages admixed with scattered neutrophils, lymphocytes and plasma cells. Haematoxylin & eosin stain, bar = 100 μm
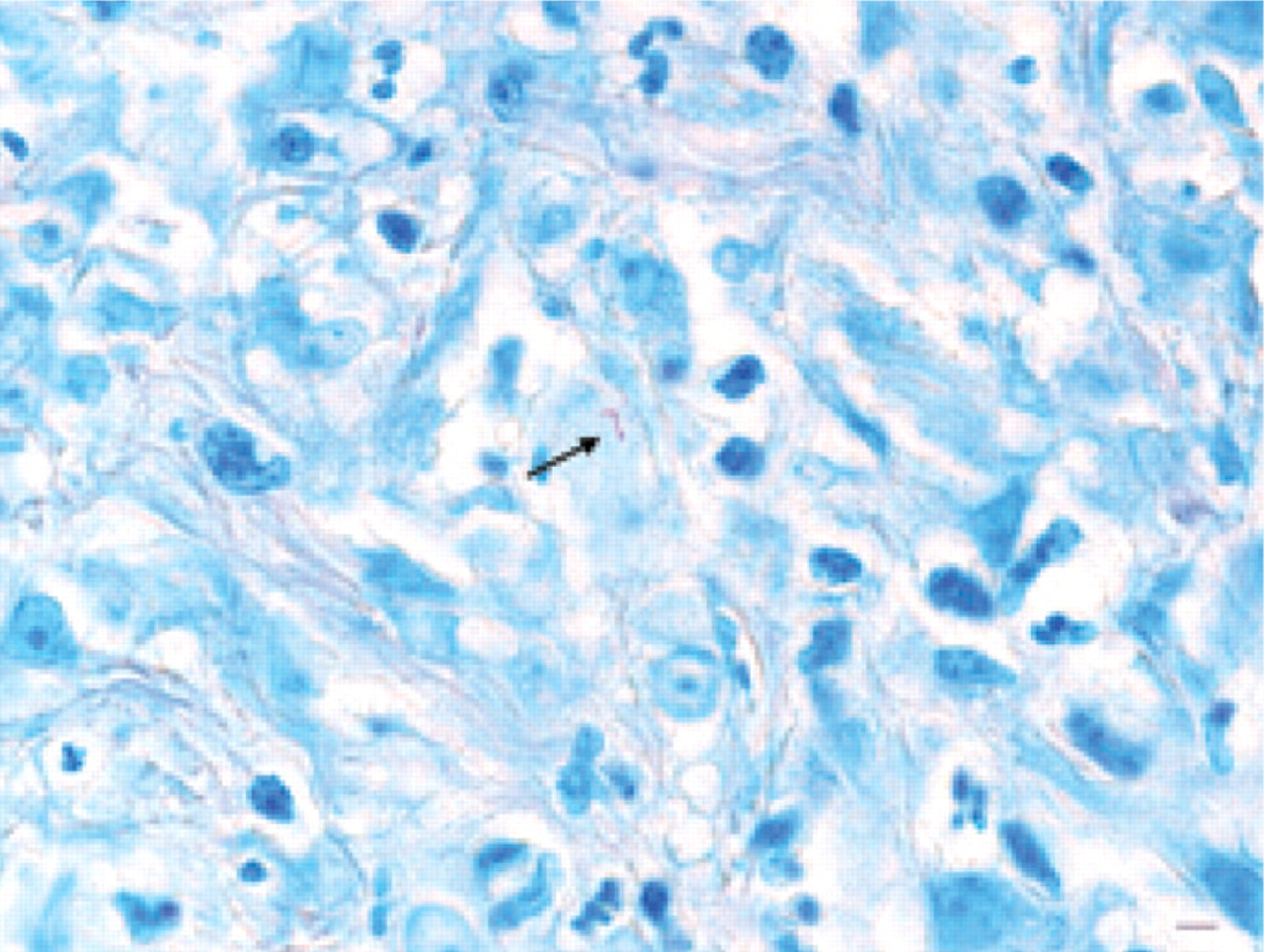
Rare acid-fast bacilli (AFB) within epithelioid macrophages. The bacteria (arrow) appear as slender rods of approximately 5 × 2 μm with a hooked end. Ziehl-Neelsen stain, bar = 5 μm
Histopathology of the lesions characterised them as deep dermal and subcutaneous confluent granulomas composed of large numbers of closely packed epithelioid macrophages admixed with variable numbers of neutrophils, lymphocytes and plasma cells (Figs 2 and 3). Ziehl-Neelsen staining revealed rare acid-fast bacilli within the epithelioid macrophages (Fig 4). Routine bacterial cultures and direct microscopic evaluation of an imprint of the biopsy following ZN staining were negative.
In August 2008, once histopathological findings were known, the cat was anaesthetised again, and biopsies were collected from the chin nodule and the enlarged lymph node and sent to the Institute for Infectious Diseases, Bern, Switzerland, for specialist mycobacterial culture and identification.
On direct microscopic evaluation of macerated, centrifuged and auramine-stained biopsy material, low and moderate numbers of acid-fast bacilli were detected in the lymph node material and chin nodule, respectively. After 1 week, while the microbiology results were pending, the other (left) mandibular lymph node started to enlarge, so treatment with doxycycline and enrofloxacin was recommenced, at the same dosages as previously. No improvement was seen after 6 weeks of treatment, at which point clarithromycin was added (Klacid; Abbott AG, Switzerland, 62.5 mg PO q12h). One week after introducing clarithromycin (October 2008), M microti was identified (see box below for details).
In the light of the mycobacterial identification, doxycycline and enrofloxacin were stopped, clarithromycin was continued at the described dosage, and marbofloxacin (Marbocyl; Vétoquinol, France, 10 mg PO q24h) and rifampicin (Rimactan; Sandoz, Switzerland, 75 mg PO q24h) were added. For logistical reasons (capsules containing 75 mg of Rimactan had to be manufactured by a local pharmacy) rifampicin was started 3 days after the marbofloxacin.
Mycobacterial identification and susceptibility testing
For mycobacterial identification, DNA was extracted from the biopsies of the lymph node and the nodule on the chin. 4,5 All polymerase chain reaction (PCR) amplifications were performed on a GeneAmp PCR system 9700 (Applied Biosystems, Foster City, CA, USA). For the amplification of the internal transcribed spacer (ITS), a nested PCR was performed 6 using the primers listed in Table 1. The ITS PCR products were sequenced using a BigDye Terminator Sequencing Kit v1.1 (Applied Biosystems) following the manufacturer's instructions. Capillary electrophoresis of sequencing products was performed on an ABI 310 Genetic Analyzer (Applied Biosystems). DNA sequences were assembled and analysed with SeqMan and MegAlign computer programs (DNASTAR, Madison, USA). Comparison of DNA sequences and their corresponding amino acid sequences in the GenBank database was performed with BLASTN search. 9 Spoligotyping was performed according to the method of Kamerbeek et al. 10 To identify the obtained spoligotyping pattern and the corresponding spoligotype international type (SIT) number, data were compiled in Microsoft Excel and analysed with the MIRU-VNTRplus online analysis tool (http://www.miru-vntrplus.org). 11
Oligonucleotide primers for amplification of the internal transcribed spacer (ITS) region

F = forward, R = reverse
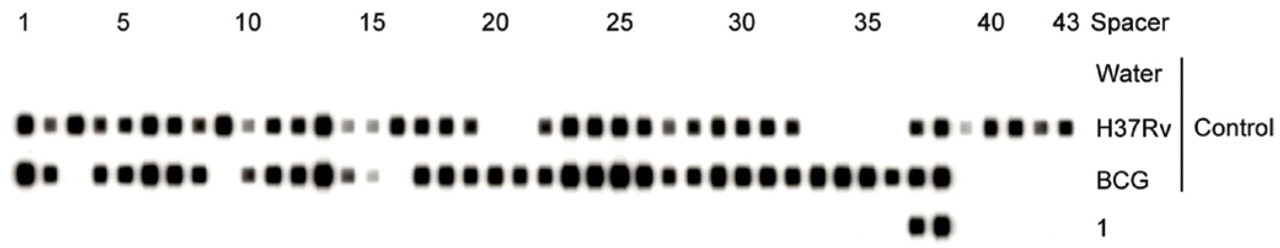
Spoligotyping patterns of amplified DNAs are shown. The numbers above each pattern represent the spacers. Water, M tuberculosis H37Rv and M bovis BCG were used as controls; 1 = DNA of the chin nodule
Two hours after the first dosage of rifampicin, the normally very calm and relaxed cat was suddenly nervous, and pruritic over its entire body. Its skin was erythematous, especially on the abdomen and around the nipples. The rest of the clinical examination was unremarkable. An adverse drug reaction to rifampicin was suspected and the drug was withdrawn. The cat's behaviour normalised within 6–7 h and this reaction did not recur. The mandibular nodules started to decrease in size after 2 weeks of treatment with clarithromycin (and 1 week after initiating marbo-floxacin). In October 2008, after the cat had been treated for 6 weeks with clarithromycin, the mandibular nodules and the mandibular lymph nodes were completely normal. The antibiotics were continued for a further 4.5 months and then stopped in April 2009 (Fig 6).
At the time of writing, the cat had been off medication for 13 months and no recurrence had been seen.
Review of the literature
Aetiopathogenesis
In cats and dogs tuberculosis can be caused by a number of closely related bacteria, including M tuberculosis, M bovis (the ‘bovine bacillus') and M microti (the ‘vole bacillus'). In the developed world, tuberculosis in cats and dogs is now recognised infrequently. 12 When it is diagnosed it is usually caused by M bovis or, in the case of cats, M microti. Of the recent cases of tuberculosis in cats in the UK (approximately 115 cases identified in the past 4 years by the Veterinary Laboratories Agency [VLA], UK), 55% were caused by M microti and 45% by M bovis (VLA, personal communication 2009). 13 Over the past 14 years, the VLA has identified over 100 cases of feline tuberculosis caused by M microti, showing that this infection is far from rare. 14 By contrast, infection of cats with M tuberculosis is exceedingly rare, 15 probably because they are naturally resistant to it. 16
Only five cases of tuberculosis in dogs in the UK have been confirmed by the VLA in the past 20 years, all of which were due to M bovis. 17,18 There have also been a handful of published case reports concerning tuberculosis in dogs, most of which involved M bovis, 15,17,19–22 although four cases resulted from M tuberculosis infection; 15,23–25 only one (from France) was caused by M microti. 26

The cat, pictured in April 2009, when the antibiotics were stopped
Interestingly, the profile of causes of tuberculosis in cats and dogs is quite different from the picture seen in humans. At least in industrialised countries, over 90% of cases in people result from infection with M tuberculosis, only approximately 1% are caused by M bovis, 27 and disease due to M microti is very rare. 28
M microti was first recognised as a cause of tuberculosis in voles in 1946. 29 Since then it has been isolated from many mammalian species (although it is very slow to grow) 14 and molecular diagnostics have improved its recognition. 30 It has been found in many countries, including the UK, mainland Europe (Germany, the Netherlands, Switzerland), South Africa and South America, and been seen in humans, cats, voles, wood mice, shrews, South American camelids, pigs, wild and pet ferrets, squirrel monkeys, rock hyrax, a dog, a badger and a bull. 1,14,26,28,31–45 Experimental infection has been achieved in guinea pigs, ferrets, a rabbit and a calf. 32 Natural infection is very common in some species, such as field voles. A necropsy study of field voles in the UK found 21% to be infected, 30 with the prevalence varying from 0–50% depending on the geographical location, the season (being highest in spring) and the presence of clinical signs (in advanced stages the disease is often cutaneous). 29,46 Some domestic species, including cats and llamas, appear to be particularly susceptible to M microti infection. 28
In cats, M microti infection was previously, rather confusingly, termed M microti-like, as it was unclear at the time that it was actually the same organism. 12,47,48 In addition, a number of reports have discussed cases of infection that were attributed to M tuberculosis 49 or M tuberculosis var bovis, 1,50 which on further review and/or investigation were almost certainly due to M microti.
M microti was initially believed to be nonpathogenic to humans. Historically, it was used as a vaccine against tuberculosis, being injected into thousands of healthy people and apparently causing no signs of ill health. 28,51,52 However, a small number of clinical cases have now been reported, with affected people typically presenting with pneumonia (and occasional encephalitis or peritonitis), some of whom have been immunocompromised and some of whom have not. 28,36,37,53–55
Analysis of the isolates has led to the recognition of two types of M microti based on their growth characteristics and their spoligotypes: the vole type and the llama type. 37 The llama type has so far been recognised in humans, llamas, a cat and a dog, 26,28,37,55 while the vole type has been recognised in voles, cats and a badger. 28,37
Epidemiology
In cats and dogs M microti infection is believed to be transmitted via direct contact with wild rodents and other small mammals. In cats, most cases present with cutaneous lesions that typically affect the ‘fight and bite sites’ — ie, the face and legs, which are the areas most likely to be bitten when playing with prey — and known risk factors include being keen hunters and regularly catching small rodents. 1,14 Studies have shown that, in the UK, wild field voles (Microtus agrestis), bank voles (Clethrionomys glareolus) and wood mice (Apodemus sylvaticus) can all be naturally infected with M microti. 30
The authors believe that the most likely mechanism by which cats become infected is by hunting infected small wild rodents, which is supported by the finding of spatial clustering of cats and voles with M microti infection within certain areas of the UK. 14
All members of the tuberculosis complex pose potential zoonotic risks. However, there are no reports of cats or dogs passing M microti on to humans.
Predisposition
The published reports describe M microti infection in some detail in 23 cats and one dog. 1,26,32,47,48,56 Limited detail is available for a further 101 cats. 14 All cases have been seen in adult animals (cats 2–13 years of age [mean 8 years]; the dog was 4 years old). 1,14,26,32,47,48,56 Unlike tuberculosis in general, where there is no gender bias, 57 male cats appeared to be overrepresented with M microti infection in one report. 1 In addition, most feline cases of M microti infection have come from clusters in the south-east of England and the north of England/south of Scotland. 1,14,48 Most cats that have been tested for FIV and FeLV have been negative. 1,47
Clinical signs
The most consistent clinical signs of M microti infection in cats are cutaneous lesions and/or submandibular lymphadenopathy. These are often the only signs. Skin lesions were seen in 12 of the 23 reported cats. 1,32,47,48,56 The lesions typically consist of firm raised nodules, which are frequently, but not always, freely mobile. Some of the fixed lesions extend to involve the underlying muscle and bone. Some lesions ulcerate, and draining sinuses may be seen. Lesions typically involve the face, chest wall, legs, tail base and perineum. Submandibular lymphadenopathy was reported in 11 of the 23 cats, and associated prescapular lymphadenopathy was frequently present. 1,32,47,48,56 Limited information is available for a further 61 cases, 45 of which involved the skin and superficial lymph nodes, most typically of the head, but sometimes of the limbs and tail, or neck and torso. 14 Regional lymphadenopathy, or more generalised changes affecting the mesenteric lymph nodes, have been seen in some of the cats, as has pneumonia (typically resulting in widespread small focal lesions); occasional cases have had ocular involvement or localised tuberculous arthritis. 1,14,32,47,48,56 Most of the cats were clinically well when the diagnosis was made, although disease became more generalised in some cases. Once the lungs are affected cats typically develop mild to moderate dyspnoea and a soft cough. 1,32,56
In the one reported case of M microti infection in a dog, the patient presented with acute peritonitis resulting from granulomatous abscessation of the spleen, liver and intra-abdominal lymph nodes. 26
With tuberculosis in general (ie, when caused by M bovis, M microti or even M tuberculosis), the clinical signs usually relate to the route of infection. Disease affecting the alimentary tract typically results from ingestion of infectious material; disease affecting the respiratory tract typically results from inhalation of infectious material (but can also result from haematogenous dissemination or local spread); and disease localised to the skin most commonly results from cutaneous inoculation (but can also result from local spread). 12,14,16,57
Most cases of tuberculosis in cats are now cutaneous, with respiratory and alimentary forms being seen less frequently. 12,14 The skin lesions often involve the face, extremities, tail base or perineum — the so-called ‘'fight and bite sites'. Interestingly, in cats, the primary complex is often incomplete, especially when the infection gains entry via the mouth or intestines (ie, granulomas form in the local lymph nodes, but there are no obvious lesions at the site of entry). 1,14,47,58
While there are currently very few cases of tuberculosis in dogs, most present with pulmonary and/or alimentary signs. 17–20,24,26
Diagnostic techniques
Non-specific tests
A thorough evaluation of the patient is necessary to assess the extent of local infection and the degree of systemic involvement. Changes in serum biochemistry and haematology, if present, are non-specific and vary with the severity of the disease. However, hypercalcaemia has been seen in a number of cats and dogs with tuberculosis and appears to correlate with a poor prognosis. 17,59 Radiography can be useful in the appraisal of lung involvement. However, changes are very variable and include tracheobronchial lymphadenopathy, interstitial or miliary lung infiltration, localised lung consolidation and pleural effusion (DGM, unpublished observations). 1 Abdominal radiography and ultrasound examination may reveal hepato- or splenomegaly, abdominal masses, mineralised mesenteric lymph nodes or ascites. Bone lesions tend to consist of areas of bony lysis and sclerosis, osteoarthritis, discospondylitis or periostitis (DGM, unpublished observations). 1
Specific tests
The recently developed interferon-gamma release assay is showing promise for detecting members of the tuberculosis complex in cats and may be available from specialised mycobacteriology laboratories. 60,61 Other specific tests for the diagnosis of tuberculosis (eg, serum antibody responses) have been investigated, but have generally proved unhelpful in cats and dogs. 58,62 Unlike other species, cats do not react strongly to intradermally administered tuberculin and the results from intradermal skin testing are unreliable. 58,62,63 Even in dogs, false positives and false negatives can occur (reviewed by Greene and Gunn-Moore). 2,3 Other than Rhodes et al, 60,61 none of these papers has looked at responses to M microti infection in particular.
On histopathology, the cutaneous lesions typically involve the dermis and subcutis. Lesions consist of a granulomatous inflammatory reaction composed of discrete and coalescing aggregates of epithelioid macrophages with abundant eosinophilic cytoplasm. Neutrophils may be scattered among the macrophages. The granulomas may be surrounded by a more diffuse infiltrate of macrophages, plasma cells, lymphocytes and neutrophils, in a background of granulation tissue. Multinucleate Langhans giant cells are not usually present. Necrosis may be present and may vary in severity, from involving the centre of only a few granulomas, to forming large coalescing necrotic foci. Affected lymph nodes typically are largely replaced by a granulomatous inflammatory reaction closely resembling that of the cutaneous lesions, with necrosis as a consistent feature, and some develop a well-defined fibrous capsule. In other cases the perinodal tissues may also contain granulomas. The lesions seen in lungs, mesenteric lymph nodes and joints have microscopic changes resembling those of the skin and lymph nodes. 1,26 These findings are typical of those seen in classical tuberculosis. 17,49,58,62
Identification of mycobacteria
To confirm mycobacterial involvement, aspirates and/or biopsy samples should be stained with ZN stain (or another similar special stain). Unfortunately, the finding of acid-fast bacilli is rather non-specific at the genus (Nocardia species, Tsukamurella species, etc) and species level. The number of acid-fast bacilli seen may vary, depending on the species of mycobacteria, the location of the granulomas and the nature of the host's immune response. In most cases of M microti infection there are only a few acid-fast bacilli to be found within foamy macrophages, but in occasional cases they have been reported as being numerous. 1,26 The bacilli are usually long, slender and faintly beaded, with occasional ‘S’ shaped, hooked or looped forms. 1,26,32,56 (See Greene and Gunn-Moore for reviews of culture methods.) 2,3 Unfortunately, M microti often take 2–3 months to grow. 14
Correct handling of biopsy material
In practice, handling of biopsy material usually involves taking a sample from a case where mycobacterial disease is only one of a large number of possible differential diagnoses. If in-house staining facilities are available, this can be performed on aspirates or biopsy impression smears. However, in most cases, biopsy material must be sent to a specialised diagnostic laboratory. A practical approach is to collect the biopsy, cut it into four pieces, fix one in formalin for histopathological examination and, pending the results, place two in sterile containers and freeze them. Where other bacterial infections are suspected, the fourth sample should be sent unfixed for routine bacterial culture at which time ZN staining can also be requested. This way, if the sample is found to have ZN-positive organisms, one of the frozen portions can be defrosted and sent to one of the mycobacterial reference laboratories for specialist culture and/or molecular analysis. The last sample is retained in case further testing is required later.
Until the organism has been properly characterised, it should be considered a potential human pathogen. Whenever handling potentially tuberculous material it is necessary to take certain precautions. Depending on the country, there are laws and guidelines on how to deal with a suspected mycobacterial infection. People dealing with such cases should inform themselves. When a confirmed case is euthanased it is advisable to have the body cremated. Unfortunately, until the sample has been cultured (or identified by PCR) it is not possible to know if the infection is caused by M bovis, M microti or one of the other mycobacteria that can cause disease in cats and dogs.
Molecular PCR techniques are available for the identification of mycobacteria, and can be very useful, 2,3,14,15,26,49,64,65 especially where tissue for culture is not available; however, they are expensive, and have limited availability. Where these molecular diagnostics are available, these bacteria can be identified further to their specific spoligotype. 14
The case described in this report clearly demonstrates the power of molecular diagnostics for the rapid identification of mycobacterial infection. This proved to be important for the successful management of this cat. Therefore, we recommend that clinicians use a more aggressive diagnostic approach in this type of case; ie, as soon as acid-fast bacilli are detected, samples should be sent to one of the mycobacterial reference laboratories for specialist culture and/or molecular analysis. Although costs may seem to be high, this case illustrates that this is not really a valid argument for not pursuing such analysis. If molecular diagnostics had been undertaken earlier, two or three surgeries (with associated anaesthesia and biopsy procedures) could have been avoided, as would the administration of ineffective antimicrobials. Moreover, the stress and risk of side effects for the cat of receiving long-term daily medication could have been significantly reduced, as could the duration of possible zoonotic risk to the owner.
Therapy
Interim management
Pending a definitive diagnosis, interim therapy with a fluoroquinolone has previously been recommended. However, this should only be considered in cases of localised cutaneous infection. It is more sensible to recommend that double or triple therapy be initiated (Table 2). This not only gives the best chance of clinical resolution, but also decreases the potential for the mycobacteria to develop resistance to the fluoroquinolone. This is an important consideration, as generating drug resistance will not only be detrimental to the individual animal, but may also endanger human patients. Unfortunately, it is often 2–3 months before the culture results are known.
Factors to consider before undertaking treatment
Deciding to treat a case of suspected tuberculosis in a cat or dog can be contentious. Before undertaking treatment, it is important to consider the following points:
Potential zoonotic risk
All members of the affected animal's household must be involved in any decision making. Particular consideration should be given to those individuals most susceptible to the infection, such as any household members with HIV infection, or those undergoing chemotherapy or organ transplantation. The authors strongly advise against treatment where such individuals may be exposed to an infected animal. They also advise against treatment if the affected animal has generalised disease, respiratory tract involvement or extensive draining cutaneous lesions; any of these findings may increase the risk of disease transmission.
Inherent treatment difficulties and risks
Where the animal is a suitable candidate, it should be emphasised that treatment is long-term and difficult to maintain given patient non-compliance, the inherent toxicity of some of the drugs and the financial costs involved. In some cases, the drugs may at best suppress the disease and indefinite treatment may be required. Uncomplicated cutaneous disease appears to carry the most favourable prognosis. Whenever undertaking treatment, gloves should be worn for handling the animal.
Surgical excision
Surgical excision of small cutaneous lesions may be considered, but is successful in only a few cases. Debulking larger lesions risks wound dehiscence and local recurrence of infection.
Useful drugs for the treatment of M microti infection

NB Second-line treatments for tuberculosis should be reserved for resistant infections PO = per os, IM = intramuscularly, occ = occasionally
aAuthors recommend using a fluoroquinolone that is not enrofloxacin, which has been associated with retinal degeneration 67
bThese drugs are not licensed for use in pets. Drugs licensed for human use can be obtained by veterinary prescription from larger pharmacies
cCan be difficult to obtain
dGive with food or give water after the medication to avoid oesophageal injury
Because of this is it essential to counsel owners very carefully, making them aware of all of the potential risks and complications.
Enrofloxacin was given to the cat of this case report for the first 5 months, at a daily dosage of 10.9 mg/kg. This dosage exceeds the maximum dose of 5 mg/kg/day recommended by the manufacturer. The higher dose was chosen because it had previously been used successfully to treat a cat with M simiae infection. 66 However, higher doses of enrofloxacin can provoke irreversible retinal degeneration in 1 in 120,000 cats, 67 so this is not now recommended.
Other fluoroquinolones do not appear to carry the same risk of blindness when given to cats at higher doses, so their use is preferred over enrofloxacin.
Prognosis
The prognosis depends on the extent and severity of the mycobacterial infection. While many cases have responded favourably to treatment, and have achieved apparent cure or long-term remission, the prognosis should always be stated as guarded.
Treatment of choice
Ideally, antituberculosis treatment should consist of an initial and a continuation phase. The initial phase usually requires at least three drugs and lasts for 2 months, while the continuation phase requires two drugs and lasts for perhaps a further 4 months, depending on the extent of the disease. In those animals where triple therapy is not feasible, treatment should still involve at least two drugs and should be given for a minimum of 6–9 months. 12
Traditionally, the rifampicin—isoniazid—ethambutol combination has been considered the most effective regimen for the treatment of tuberculosis in animals. However, some newer and less toxic drugs are worth considering; for example, fluoroquinolones and clarithromycin, especially when given in combination with rifampicin. A potentially useful once-daily alternative to clarithromycin is azithromycin. Based on clinical experience gained over the past 15 years, one of the authors (DGM) recommends treatment consisting of an initial phase of rifampicin—fluoroquinolone—clarithromycin/azithromycin, followed by a continuation phase of any two of these drugs (Table 2). 1 Rifampicin is often poorly tolerated, and may need to be withdrawn early. If this is the case, and the lesions have resolved, the advice is to continue with the two other drugs; if they have not resolved, consider adding another drug (perhaps doxycycline) or switch to the resistance protocol (Table 2).
For ease of administration, all three once-daily medications can be given as liquids and placed in a single syringe prior to oral administration, or given as tablets with all three being given together after being placed in a single gelatine capsule. Alternatively, where oral medication proves too difficult, an oesophagostomy tube may be placed (through which the liquid medications can be given) and left in place for the duration of the treatment. In cases where resistance develops, the rifampicin—isoniazid—ethambutol combination may be considered. If necessary, ethambutol can be substituted with dihydrostreptomycin or pyrazinamide. Rifampicin and isoniazid are more effective and less toxic than ethambutol and dihydrostreptomycin, and consequently are more appropriate choices if only two drugs are required.
Footnotes
Acknowledgements
This article was presented as a poster at the annual assembly of the Swiss Society for Microbiology, University of Lausanne, June 4–5, 2009.
