Abstract
A 3-year-old Ragdoll cat was referred for investigation of polyuria, polydipsia, vomiting, weight loss and hypercalcaemia. Serum biochemical abnormalities included total and ionised hypercalcaemia and hypophosphataemia. Following clinical investigations a diagnosis of idiopathic hypercalcaemia was made. Because of the severity of the hypercalcaemia and the associated clinical signs, treatment for hypercalcaemia was commenced with pamidronate. Major electrolyte abnormalities were detected but, remarkably, were accompanied by minimal clinical signs. The cat was subsequently treated with oral alendronate and is clinically normal 15 months later. Reports of the use of bisphosphonates in cats are limited and close monitoring of patients is recommended.
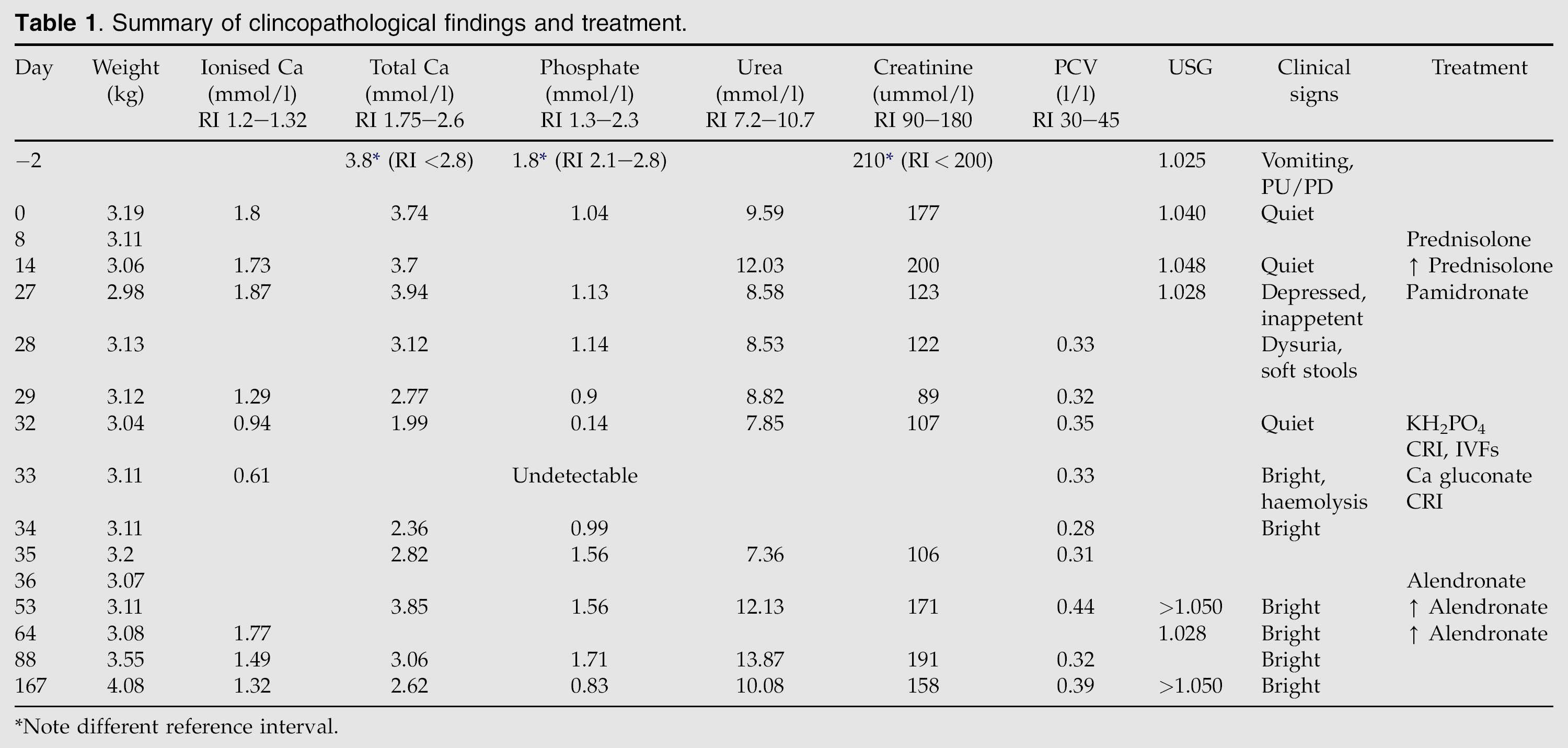
A 3-year-old spayed female Ragdoll was presented to the referring veterinarian with a 3-day history of polyuria, polydipsia and vomiting. The cat had been losing weight over a 3-week period. The diet was a mixture of tinned and dry commercial foods. Physical examination by the referring veterinarian was unremarkable. Serum biochemistry (day −2) revealed total hypercalcaemia, mild hypophosphataemia and mild renal azotaemia (Table 1). The cat was seropositive for feline immunodeficiency (FIV) antibody and seronegative for feline leukaemia virus (FeLV) antigen (Snap FIV FeLV Combo, Idexx Laboratories). The cat had been vaccinated against FIV (Fel-O-Vax FIV, Boehringer Ingelheim).
Summary of clincopathological findings and treatment.
Note different reference interval.
On physical examination at referral (day 0) the cat weighed 3.19 kg with a body condition score of 2/5. Repeat serum biochemistry demonstrated persistent total hypercalcaemia and hypophosphataemia and ionised calcium was elevated. Ionised calcium was measured using an i-Stat point-of-care analyser (Abbott Laboratories) on blood collected in pre-filled heparinised syringes. Urine specific gravity was 1.040 with normal urea and creatinine (Table 1). Mild elevations in alanine aminotransferase and total bilirubin were noted. A commercial real-time polymerase chain reaction (PCR) targeting two regions of the GAG gene of FIV returned a negative result (Gribbles Veterinary Pathology, Victoria Australia). On microscopic examination of the urine, occasional calcium oxalate crystals were identified. Idiopathic hypercalcaemia or humoral hypercalcaemia of malignancy was considered to be the most likely differential diagnosis. Other causes of hypercalcaemia including primary hyperparathyroidism, granulomatous disease, intoxication with cholecalciferol or its analogues, bony lesions and hypoadrenocorticism were considered less likely based on the signalment, history and physical examination findings. The cat was admitted for further investigation.
Thoracic radiographs and abdominal ultrasound examination were unremarkable. Parathyroid glands could not be identified on cervical ultrasound examination. Serum intact parathyroid hormone (iPTH) was consistent with a parathyroid-independent process (iPTH<12 pg/ml; reference interval (RI) 22.0–122.0 pg/ml) using a commercially available assay (Vetnostics, Australia). Briefly, a solid-phase, two-site, chemiluminescent immunoassay directed against human iPTH (1-84), (Immulite PTH assay, Siemens Medical Solutions Diagnostics, Vic, Australia, previously DPC, Los Angeles, CA, USA) was modified to measure feline iPTH. Styrene beads (8 mm) were coated with goat polyclonal PTH (39-84) and samples were assayed using the Immulite 2000 analyser. 1 Dilutions of crude feline parathyroid adenoma extract (1:100, 1:200 and 1:500), obtained after surgical removal, showed parallelism with the standard curve. Eight serum samples from cats with renal secondary hyperparathyroidism were serially diluted and showed parallelism to the standard curve. Assayable PTH fell by >50% from basal levels at 30 min post-removal of parathyroid adenoma in three cats. The intra-assay coefficient of variation (CV) at 60 and 250 pg/ml was 6.9% and 7.0%, respectively, and the inter-assay CV was 7.2% was 8.6%, respectively. The sensitivity of the assay was 12 pg/ml. Parathyroid hormone-related peptide (PTHrP), assayed using an immunoradiometric assay validated for use in cats (DiaSorin, Stillwater, MN, USA), was <1.3 pmol/l (RI<1.3 pmol/l). 2 25-Hydroxycholecalciferol, assayed using a radioimmunoassay validated for use in cats (DiaSorin, Stillwater, MN, USA), was within the reference interval (74 nmol/l; RI<145 nmol/l). 3 Idiopathic hypercalcaemia was diagnosed and the cat was discharged on prednisolone (Pred-X 5; Apex Laboratories) 5 mg q 24 h orally (PO).
At re-examination on day 14 the cat was reported to be lethargic and had lost a further 130 g (body weight 3.06 kg). Persistent total hypercalcaemia, ionised hypercalcaemia and mild prerenal azotaemia were detected (Table 1). The frequency of prednisolone therapy was increased to 5 mg q 12 h PO. On day 27 the owners reported ongoing lethargy and the cat had become anorectic. Physical examination revealed a further decrease in body weight (body condition score 1.5/5). Serum total hypercalcaemia, ionised hypercalcaemia and hypophosphataemia persisted. The cat was admitted for symptomatic treatment of severe hypercalcaemia. Rehydration was commenced with 0.9% NaCl+15 mmol/l KCl intravenously (IV) at 15 m/h. Twenty-four hours later the cat's demeanour had improved and she was eating well. Four milligrams of pamidronate (Aredia, Novartis) was diluted in 36 ml of 0.9% NaCl and administered as an intravenous infusion over 4 h. Twenty-four hours after the commencement of pamidronate therapy the cat developed stranguria, pollakiuria and pyrexia (body temperature 39.4°C; RI 37.8–39.2°C). Ultrasound examination of the urinary bladder was carried out to look for evidence of cystic calculi. No abnormalities were detected. Urine was collected by cystocentesis for culture. Antibiotic therapy was commenced with amoxicillin–clavulanic acid (Clavulox; Pfizer) 52.5 mg subcutaneously (SC) q 12 h. Analgesia was achieved using buprenorphine (Temgesic; Reckitt Benckiser) 0.03 mg q 8 h IV. Forty-eight hours after the administration of pamidronate (day 29), ionised calcium had normalised but hypophosphataemia persisted. The cat was discharged from hospital on oral amoxicillin–clavulanic acid pending urine culture results. A heavy, growth of non-haemolytic Escherichia coli which was sensitive in vitro to amoxicillin–clavulanic acid and marbofloxacin, was cultured.
At recheck on day 32 the cat was bright and responsive but had been lethargic and inappetent at home. Serum biochemistry was performed and revealed normal total serum calcium, moderate ionised hypocalcaemia and marked hypophosphataemia with a normal packed cell volume (PCV) (Table 1). The cat was hospitalised for treatment of hypophosphataemia and monitoring of hypocalcaemia. Intravenous 0.9% NaCl+20 mmol/l KCl was commenced at 6 ml/h. Potassium dihydrogen phosphate was administered at 0.132 mmol/h for 15 h and then increased to 0.16 mmol/h for a further 60 h when phosphate became undetectable in serum on day 33 (Table 1). Ten percent calcium gluconate was diluted 1:3 in 0.9% NaCl and 6 ml was administered subcutaneously 24 h after presentation when ionised hypocalcaemia worsened. Assisted feeding of a high-density caloric diet (Hill's Prescription Diet a/d) supplemented with a low-lactose milk replacer (Di-Vetalact; Sharpe) was initiated. During treatment (days 32–34) the cat was intermittently pyrexic (body temperature 39.6°C) and had small intestinal diarrhoea. Ionised calcium and serum phosphate normalised on day 35 and the cat was discharged on marbofloxacin (Zeniquin; Pfizer) 12.5 mg q 24 h PO and alendronate (Fosamax; Merck Sharp & Dohme) 5 mg q 7 d PO.
At recheck, 1 month later (day 53), the cat was reported to be bright and eating well. No abnormalities were found on physical examination and she had gained weight. Serum biochemistry showed a mild elevation in urea with normal creatinine and concentrated urine. Total hypercalcaemia was identified with normal serum phosphate (Table 1). The dose of alendronate was increased to 10 mg q 7 d PO. Frequency of administration was increased to 10 mg q 3 d PO on day 64 because of persistent hypercalcaemia (Table 1). Antibiotic therapy was discontinued following negative urine culture. At assessment, 5 months after initial (day 167) presentation, the cat was bright with a good appetite and had gained 1.1 kg. No abnormalities were detected on physical examination, serum biochemistry or urinalysis (Table 1). The dose frequency of alendronate has been gradually reduced. At the time of writing, 18 months after initiating bisphosphonates, the cat is clinically and biochemically normal. The current dose of alendronate is 10 mg q 7 d.
Idiopathic hypercalcaemia was diagnosed in this case on the basis of the stepwise elimination of other recognised causes of hypercalcaemia. It is unusual for cats with idiopathic hypercalcaemia to develop clinical signs referable to hypercalcaemia with only 4% of affected cats presenting with vomiting and only 1% with anorexia. 4 Long-term or marked hypercalcaemia can cause non-specific but severe clinical significant signs in dogs and humans. These include anorexia, nausea, vomiting, weakness, abdominal pain, constipation, polyuria, polydipsia, dehydration or obtundation. 5 The vomiting, obtundation and inappetence noted in this case were attributed to the hypercalcaemia based on the degree of elevation of the ionised calcium and the absence of another identifiable cause despite extensive investigations. Because of the severity of these signs symptomatic treatment for hypercalcaemia was initiated.
Hypophosphataemia was identified in the cat in this report on a number of occasions prior to treatment. This is not consistent with other reports in which the serum phosphate levels in cats with idiopathic hypercalcaemia have been within reference range. 6 Hypophosphataemia in association with hypercalcaemia is usually associated with increased circulating PTH and PTHrP as is seen in hyperparathyroidism and neoplasia. However, in this case the PTH concentration was consistent with a parathyroid-independent process, cervical ultrasound was unremarkable and PTHrP was not elevated. Although neoplasia remains a possible differential diagnosis in this case, 18 months after treatment the patient is clinically well and no evidence of a neoplastic process has been identified.
Elevated serum calcium can result in decreased urine concentrating ability due to reduced tubular sodium resorption and the impaired action of antidiuretic hormone. The mild renal azotaemia noted on day −2 was considered likely to be secondary to the ionised hypercalcaemia in this case.
There is little information available on the symptomatic treatment of hypercalcaemia in cats. The management of hypercalcaemia in humans and dogs comprises rehydration, promoting calciuresis, treatment of any underlying disease process and inhibition of accelerated bone resorption. Glucocorticoids have been used in the treatment of feline idiopathic hypercalcaemia. 2 Glucocorticoids act by reducing bone resorption, increasing renal excretion of, and decreasing intestinal absorption of calcium. 7 The response to treatment of idiopathic hypercalcaemia with prednisolone has been variable. In this case there was no response to prednisolone therapy despite using high doses.
Other options for treating hypercalcaemia include dietary modification, frusemide and calcitonin. Increasing dietary fibre has been reported in the treatment of idiopathic hypercalcaemia in cats. 8 However, many high-fibre diets are also calcium-rich to counter-act the increased gut transit time. In this case a high-fibre diet was considered to be inappropriate due to the patient's poor body condition. Frusemide administration, usually in combination with intravenous fluid therapy, is often used in rescue protocols for the treatment of acute ionised hypercalcaemia to promote calciuresis. 9 It was elected not to use frusemide in this patient due the previously documented azotaemia. Calcitonin administration has also been used in the treatment of hypercalcaemia secondary to toxicosis or increased bone resorption. Data regarding its use in cats and in dogs suggests that the response may be short-lived and accompanied by side effects such as gastrointestinal signs and allergic reactions. 10 Instead, due to the severity of the biochemical changes and their clinical significance, bisphosphonate treatment was initiated. The bisphosphonates are a group of drugs that inhibit bone resorption and are the standard therapy for malignant humoral hypercalcaemia in humans. 11,12 They act by inhibiting osteoclast apoptosis and sites of active bone turnover. 9,13 They have been used in veterinary patients for the treatment of primary and secondary bone cancer, cholecalciferol intoxication, and humoral hypercalcaemia of malignancy. 9,14,15 There is a single reported case of the treatment of a cat with concurrent idiopathic hypercalcaemia and chronic kidney disease with pamidronate. 16
Pamidronate administration was preceded by correction of the patient's hydration status as intravenous administration of bisphosphonates, including pamidronate, has been reported to induce renal damage in humans and dogs. 17 The dose and rate used were similar to that previously reported. 16 The patient developed marked hypophosphataemia and hypocalcaemia. Remarkably, there were no clinical signs of hypocalcaemia and hypophosphataemia was associated with only a mild reduction in the patient's haematocrit. Due to their mechanism of action on bone metabolism the bisphosphonates have been reported to cause hypocalcaemia, hypophosphataemia and hypomagnesaemia. Hypophosphataemia is the most commonly reported abnormality to occur in humans following pamidronate administration. 18 Protracted electrolyte abnormalities have not previously been reported in companion animals following bisphosphonate administration. The use of samples collected in pre-heparinised syringes may have led to an underestimation of the ionised calcium level. 19 Thus the hypercalcaemia prior to treatment may actually have been higher than the measured level and the hypocalcaemia detected following pamidronate therapy may not have been quite as severe as suggested, although there was an accompanying fall in total calcium which is not subject to this potential source of error.
Alendronate therapy was chosen for long-term control of the hypercalcaemia. To the authors’ knowledge there are no reports of the use of alendronate in cats. Care should be taken when dosing cats with oral alendronate. In humans oral alendronate administration has been associated with oesophagitis and oesophageal stricture in up to 15% of patients. 20 A direct irritant effect of alendronate on the oesophageal mucosa of dogs has been demonstrated. 21 Drug-induced oesophageal disease resulting in oesophageal stricture formation has been reported in cats receiving oral doxycycline or clindamycin. 22,23 Because of this oral medication should be accompanied by food or a water swallow. However, food substantially reduces the bioavailability of oral alendronate and it is recommended that it be administered on an empty stomach. The oral bioavailability of alendronate in the fasted state is about 0.7% in humans, and less than 2% in all species studied. 24,25 Prevention of drug-induced oesophageal disorders can be achieved by instructing owners to give solid medications with a 5–6 ml water bolus. 26 Long-term treatment with bisphosphonates has been reported to cause osteonecrosis of the jaw in humans and dogs and is believed to be associated with reduced bone remodelling rates. Up to 25% of dogs are affected after 1 year of treatment and up to 33% after 3 years. 27 Most patients develop lesions after dentoalveolar treatment, particularly, tooth extraction followed by non-surgical root canal therapy. 28 Due to concerns over the development of such lesions in our patient the dose of alendronate has been gradually reduced.
The incidence of idiopathic hypercalcaemia has increased in cats and it is now one of the most commonly diagnosed causes of hypercalcaemia. 29 It has been postulated that an acidifying diet may contribute in some cases. 30 An acidifying diet was ruled out as contributing in the case presented here. It may be that idiopathic hypercalcaemia comprises more than one disease process. Familial benign hypercalcaemia is a condition reported in humans in whom abnormally high serum calcium levels are usually identified incidentally or following identification of other affected family members. Some affected patients present for investigation of headaches, which may manifest as depression and lethargy in other species, for which no other cause is identified. The disorder is inherited autosomal dominant with full penetrance. 31 Due to the non-specific signs and the low rate of occurrence of these in both human familial benign hypercalcaemia and feline idiopathic hypercalcaemia, the incidence of the conditions may be higher than currently believed. Whether there is a genetic basis for some cases of feline idiopathic hypercalcaemia is unknown.
This is the second reported case of the use of bisphosphonates in the treatment of idiopathic hypercalcaemia in a cat and the first to report electrolyte changes following such therapy. This case demonstrates the successful treatment of an increasingly common condition and highlights that clinicians should use bisphosphonates with care and monitor patients closely.
Footnotes
Acknowledgement
The authors would like to thank Dr Steven Anich for ongoing case management.
