Abstract
In this retrospective study medical records of 11 cats with gastrointestinal lymphoma were evaluated to determine the efficacy of radiation therapy when used in a rescue therapy setting. All cats had relapsed or resistant lymphoma. Two fractions of radiation were delivered over 2 days for a total of 800 cGy. Acute effects of radiation were not noted, except one cat that had a self-limiting loss of appetite. Response was noted in 10/11 cats. Median survival post-radiation therapy was 214 days and the overall median survival in this study was 355 days. This study suggests that abdominal irradiation for feline gastrointestinal lymphoma was well tolerated and may contribute to a positive clinical response.
Historically, the first line therapy for the various forms of feline lymphoma has been chemotherapy. In general, cats respond well to chemotherapy protocols with response rates ranging between 50% and 75% depending on protocol, grade, and location of the lymphoma. 1–11 The use of radiation therapy in feline lymphoma has primarily used in nasal, extranodal, or single node lymphoma.
Rescue therapies for cats with progressive or resistant lymphoma have received very little attention in the literature. A single publication describes the use of doxorubicin for progressive feline lymphoma. 12 The paucity of published rescue protocols for cats is in contrast to greater than a decade of literature focusing on rescue protocols for canine lymphoma, which has generated more than 11 rescue protocols. 13–23
In 1902 radiation therapy was discovered to have efficacy against human lymphoma. 24 In the following decades it became universally accepted that malignant lymphocytes are radiosensitive. 25 During the 1900s, radiation therapy developed into a mainstay for a majority of therapy protocols for lymphoma (Hodgkin's and non-Hodgkin's). Chemotherapy inclusion in lymphoma protocols did not occur until the 1950s. 25 Even with this long and extensive history, the amount of radiation required in lymphoma therapy (whether palliative or as part of a multimodality protocol) remains undecided. 26–28 The majority of human protocols for the treatment of lymphoma are chemotherapy based, but radiation continues to play a role in certain types and stages of lymphoma. 24–29
Over the last two decades, veterinary oncology has begun to explore the use of radiation therapy for canine lymphoma. It has been included in induction and consolidation protocols as well as in the rescue setting. 23,30–34 In recent literature, radiation therapy increased remission and survival times when administered in combination with chemotherapy. 30,34 In cats, while radiation therapy has been shown to be an effective part of a multimodality approach for nasal and extranodal lymphoma, 35–37 efficacy in other forms of feline lymphoma has not been established. The purpose of this retrospective study was to evaluate the efficacy and toxicity of abdominal radiation when used in the rescue setting for cats with alimentary lymphoma.
Please cite this article in press as: Dorothy L Parshley et al., Abdominal irradiation as a rescue therapy for feline gastrointestinal lymphoma: a retrospective study of 11 cats (2001–2008), J Feline Med Surg (2011), doi:10.1016/j.jfms.2010.07.017
Materials and methods
Records of cats treated between 2001 and 2008 by the Oncology Department at Veterinary Clinics of America All Care Animal Referral Center (VCA/ACARC) were retrospectively reviewed. Eleven records were selected for inclusion in this study. All cats had a cytologic or histopathologic diagnosis of alimentary lymphoma and all were rescued with abdominal radiation. Critical for selection of these cats was the presence of follow-up data after radiation therapy. The minimum follow-up was 17 days, with a majority of cats (nine cats) having more than 25 days of follow-up data.
Data recorded included signalment, date of diagnosis, weight at initial diagnosis and immediately preceding radiation therapy, location of disease, method of diagnosis, cell size, diagnostic imaging methods, presenting clinical signs, duration of the clinical signs, feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV) status, laboratory test results, type of chemotherapy protocol(s), response to chemotherapy, radiation therapy protocol, response to radiation therapy, and date of death if known. Laboratory results recorded included complete blood counts (CBC) and serum chemistries prior to chemotherapy, prior to radiation therapy, and 2 weeks after completion of radiation therapy. A cat was considered anemic if a packed cell volume was 28% or lower. Anatomic location of the lymphoma was determined using diagnostic reports from abdominal ultrasound, endoscopy, and radiographs. Whether the diagnosis was made via cytology or histopathology was recorded. Chemotherapy protocols included CHOP, COPLA, VCAA, leukeran and prednisone, 39 as well as single agent protocols used for induction or due to progressive disease. Lomustine was used exclusively in the rescue setting. Four cats received rescue chemotherapy prior to radiation therapy, including lomustine, mitoxantrone, L-asparaginase, and COPLA protocol. After radiation therapy three cats received chemotherapy in the adjunctive setting, which included use of mitoxantrone, lomustine, and doxorubicin. The chemotherapy agents employed included doxorubicin (Adriamycin; Teva Pharmaceuticals Industries: 1 mg/kg diluted in 30 ml 0.9% NaCl administered IV over 20 min), mitoxantrone (Novatrone; Hospira: 5.5 mg/m2 diluted in 0.9% NaCl administered IV over 20 min), vincristine (Oncovin; Eli Lilly: 0.6 mg/m2 administered IV bolus), cyclophosphamide (Cytoxan; Baxter: 200 mg/m2 administered IV bolus), lomustine (CCNU/CeeNU; Bristol-Myers Squibb: 40–50 mg/m2 orally once every 3–4 weeks), chlorambucil (Leukeran; GlaxoSmithKline: 2 mg orally q 72 h), cytosine arabinoside (Cytarabine; Bedford Laboratories: 100 mg/m2 divided into q 6 h or q 8 h doses SC over 2 days), and L-asparaginase (Elspar; Ovation Pharmaceuticals 400 IU/kg SC at induction or for rescue).
Radiation therapy was administered using an isocentrically mounted Picker Co-60 Model C8, SN 170. The radiation field included the abdominal cavity and spared the spine. Rear limbs were retracted caudally to minimize the volume of femur irradiated. Radiation was administered via parallel, opposed portals using SAD geometry. No beam modification or additional blocks were used during radiation administration. Dose was manually calculated at midplane.
Cats were maintained under general anesthesia during radiation therapy. Anesthesia was induced using propofol (Diprivan; AstraZeneca, 3–5 mg/kg); the cats were intubated and maintained using an isoflurane (Hospira) oxygen admixture. Two cats received a second course of radiation therapy when progressive disease was noted 1 year and 2 years later. Response to therapy for these two cats was solely recorded for the first dose of radiation therapy and not the subsequent doses.
Survival was defined as the time between diagnosis or radiation therapy and the date of death or euthanasia. Cats were censored when they were lost to follow-up.
The Kaplan–Meier product limit method was used to estimate survival and response. Categorical data was tested by log rank analysis. A value of P<0.05 was considered significant. Statistical analyses were performed using computer software (GraphPad Prism 4 for Windows).
Results
Eleven cats met the requirements for entry into this retrospective study. All cats had either cytologic or histopathologic diagnosis of lymphoma, were treated with various chemotherapy protocols, and were rescued with radiation therapy. Median follow-up period was 214 days. All cats had either partial remission (6/11) or complete clinical remission (5/11) after chemotherapy induction protocol.
The median age of the cats was 12 years with 72% (8/11) over 11 years old. Eight of the 11 cats were male, and all were neutered. Domestic shorthair was the predominant breed (7/11). All 11 cats were negative for both FeLV and FIV.
The most common sign prior to diagnosis of lymphoma was weight loss (7/11). The cats that weighed 3.0 kg or less at the time of diagnoses also weighed 3.0 kg or less at the time of radiation therapy. Other signs included vomiting (5/11), anorexia (3/11), and diarrhea (2/11). Two cats had a previous diagnosis of inflammatory bowel disease. Duration of clinical signs prior to the diagnosis ranged from 1 week to over a year. Seven cats (63%) had clinical signs for 4 months or less prior to a diagnosis. Median weight of the cats was 3.4 kg and weights ranged between 2.7 kg and 5.14 kg.
Diagnosis of lymphoma was obtained via histopathology in a majority of cats, through surgical (6/11) or endoscopic (1/11) biopsy. Four cats were diagnosed by cytology via ultrasound guided fine needle aspiration. Six cats had small cell lymphoma, four cats had large cell or lymphoblastic lymphoma, and one cat had large granular lymphocyte lymphoma. None of the cats had a cellular immunophenotype determined.
Diagnostic imaging studies of the cats included abdominal ultrasound (10/11) and radiographs (4/11). Serum chemistry results were available for all cats at the time of diagnosis. Only one cat had significant chemistry abnormalities (elevated alanine transferase and alkaline phosphatase). On ultrasonographic evaluation this cat was suspected to have lymphoma infiltration of the liver. Seven cats had a CBC prior to and 2 weeks after radiation therapy. Anemia was the most common abnormality detected (3/7) and those cats that were anemic at diagnosis were also anemic just prior to and after radiation therapy. No platelet or white blood cell abnormalities were noted at any time point.
All cats had intestinal involvement including diffuse intestinal involvement (5/11), duodenal (3/11), ileal (2/11), jejunal, and gastric disease. Mesenteric lymph node involvement was present in three cats. Four cats had either splenic or liver involvement. Based on limited staging information available, all cats were either stage III (7/11) or stage IV (4/11) at time of diagnosis. All cats had failed previous chemotherapy protocols or had progressive disease after achieving a remission (partial or complete). In one cat radiation therapy was initiated during induction chemotherapy due to incomplete clinical response to therapy.
Ten cats received a total abdominal radiation dose of 8 Gy, and one cat received 4 Gy. Two cats received two additional fractions of 4 Gy to the mandibular lymph nodes and mediastinum for a total of 8 Gy to each location. Two cats received further radiation therapy when progressive disease was diagnosed 1 year and 2 years after initial abdominal radiation.
One cat died the day after the last dose of radiation therapy. This cat was critically ill prior to the administration of radiation therapy due to significant liver infiltration with lymphoma.
Acute effects associated with radiation therapy were minimal; one client reported a decreased appetite for a few days post-therapy. All cats that survived beyond radiation therapy (10) had a period of clinical and symptomatic resolution of their lymphoma.
Median overall survival duration was 355 days (range of 88–998 days) (Fig 1). Median survival duration after radiation therapy was 214 days (range of 1–768 days) (Fig 2). Four of the 11 cats were lost to follow-up and were censored. Various factors were evaluated for potential influence on median survival duration and median PFI after radiation therapy. Factors evaluated were anemia, duration of the clinical signs, weight of animals at the time of radiation therapy, cell type (large cell versus small cell), and use of chemotherapy after radiation. Cats that were anemic did have lower overall survival and survival after radiation therapy but due to the low number of cats with CBC results statistics were not performed. Only weight significantly impacted survival outcome (Fig 3). Cats weighing less 3.4 kg (median weight) had significantly decreased overall survival and survival after radiation therapy (P<0.014 and P<0.01).
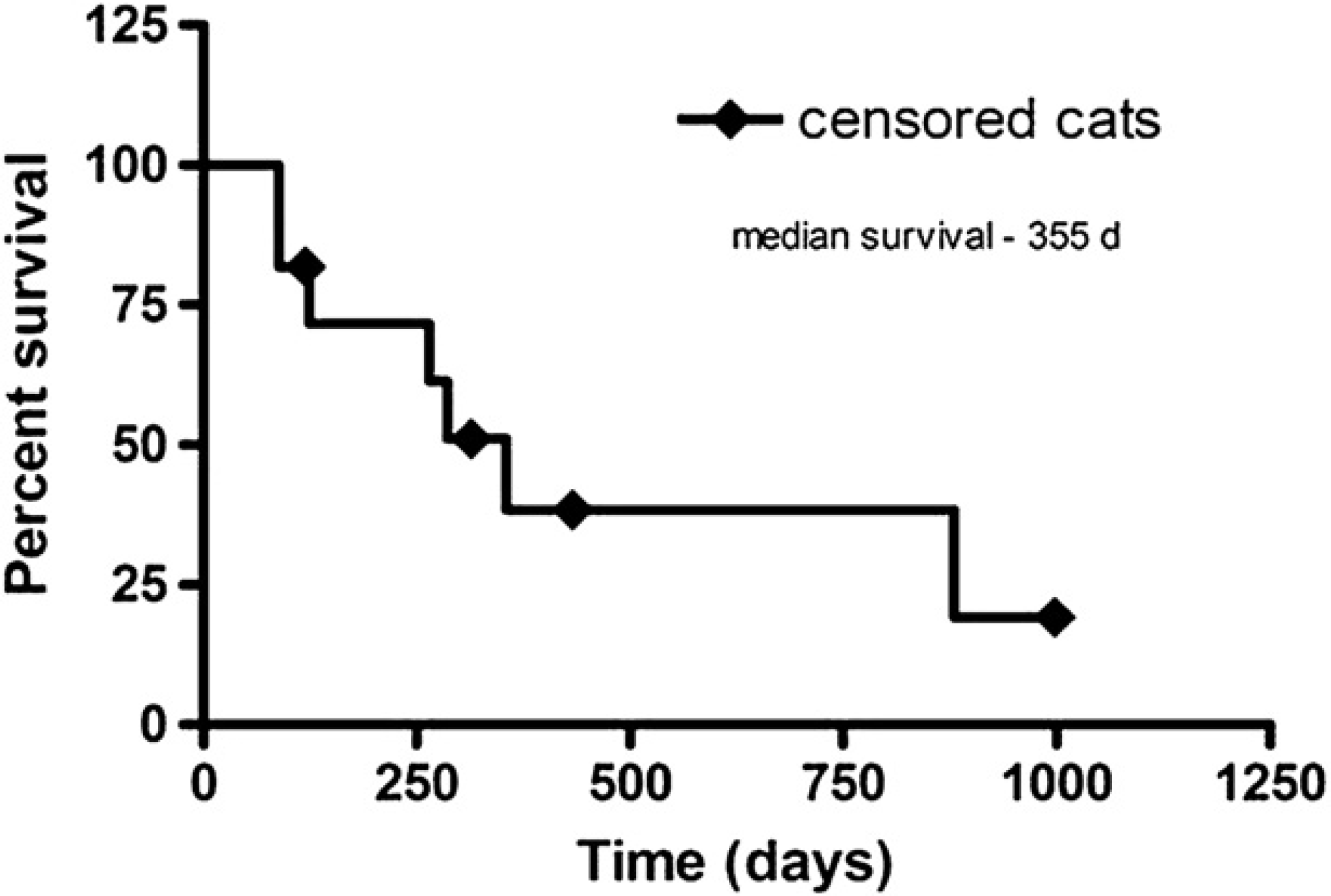
Kaplan–Meier product limit graph for overall survival.

Survival after radiation therapy.

Kaplan–Meier product limit graphs with log rank analysis results correlating overall survival (A) and survival after radiation therapy (B) to weight at time of radiation therapy. Censored cats are represented by ticks.
Discussion
The median age of the cats in this study is consistent with other reports on primary alimentary lymphoma from the ‘post-FeLV era’, when the main infectious causation of lymphoma in cats has become less prevalent. 2,3,7,42 Population distribution between males and females was a ratio of 2.3:1 (males to females), with all animals neutered. The most common sign noted prior to diagnosis was weight loss, which is also consistent with published data. 5,10,11,40,41,43
The predominant form of feline lymphoma reported is intermediate or high grade, although alimentary lymphomas may have an over-representation of low grade or indolent forms. 44 The size of lymphocyte has been in most cases related to the grade of the lymphoma. This is a loose association, as indicated in humans and in one larger study of cats; not all small cell lymphomas are low grade or of indolent behavior. 44 Thus, care must be taken in making an association between size of a lymphoma cell and grade. In our study, the number of small cell lymphomas was slightly higher (54%) than large cell lymphomas. Thus we potentially observed an equal distribution of higher grade lymphomas with low-grade lymphomas. Histopathologic evaluation confirming this grade distribution was impossible because some cases no longer had archival tissue samples and four cats had cytologic diagnosis.
Median overall survival for the cats in this study was 355 days (11.8 months). Median survival durations reported in the literature are in general less than 9.6 months. 2,3,5–8,38,40,45 It should be noted that a majority of these reports only described outcomes of induction protocols. The exception is a 2008 paper by Kiselow et al that reported a 704-day median survival. Kiselow's paper evaluated low-grade lymphoma response to therapy. 10 Previously it was predicted that low-grade or indolent lymphomas would have a longer survival than higher grade lymphomas. 4,44 As our study likely had a mixture of low and higher grade lymphomas, we anticipated that our reported median survival would be shorter than that reported by Kiselow et al. 10 Comparison of our overall median survival rate to other reports is additionally made difficult because our cats were exposed to multiple chemotherapy protocols and all cats received rescue protocols. However, as the reported survival duration of cats here was longer than seen in other reports, we infer there may be benefit to cats with alimentary lymphoma treated with radiation therapy in a rescue setting. Clearly further prospective trials are necessary to definitively establish this finding.
All of the cats in this study initially responded to chemotherapy with either a partial remission or complete remission. In previous studies, cats that positively responded to chemotherapy had longer median survival times than those that failed to respond to therapy. 5,7,9,10,43 Studying a population of cats that showed initial response to chemotherapy may have biased our results in favor of longer overall survival. Additionally, three cats received chemotherapy after radiation therapy, potentially biasing our results.
Ten of the 11 cats in our study had a client reported and physical examination supported response to abdominal radiation. This represents a 90% response rate (10/11 cats). Attempts to compare these results with other similar studies are limited by the fact that there is only one other paper evaluating a rescue protocol for relapsed or resistant feline lymphoma. In the paper by Oberthaler et al, single agent doxorubicin was evaluated as a rescue protocol. This paper reported a response rate of 22% (5/23) 12 in cats that had never before received doxorubicin. It is still possible that this lower response rate may in part be due to the development of chemotherapy cross-resistance and in part to a larger population of large cell lymphoma in the cats reported in this article.
Radiation therapy administered to the abdomen was well tolerated, with only one client reporting a patient's decrease in appetite after therapy. One cat died in the hospital the day after radiation therapy finished. This cat was critically ill due to disease progression prior to the first dose of radiation. The medical record for this patient reported that death was attributable to progressive hepatic lymphoma.
Acute effects reported for dogs receiving half body radiation therapy during induction or in rescue protocols are bone marrow suppression and gastrointestinal (GI) upset. 23,30,31,33,34 In general, the magnitude of bone marrow suppression and GI upset reported was low grade and self-limiting; however, a few cases of life-threatening or fatal thrombocytopenia's were reported. 31,33 The majority of reported bone marrow suppression occurred 1 and 2 weeks after radiation therapy. 23,30,31,33,34 Six cats in our study had a CBC performed 2 weeks after radiation therapy. None of these cats had observed neutropenia or thrombocytopenia. With the exception of the single cat with decreased appetite, there was no reported GI upset in our cats. Though our cats appeared to tolerate radiation therapy better than dogs did, our radiation field was restricted to the abdominal cavity and not the entire caudal half of the body. Thus, a different type of radiation field in our protocol may explain the minimal myelosuppressive or GI adverse effects observed.
Several factors were evaluated for potential impact on response to therapy and survival. Only the weight of cats at diagnosis and radiation therapy displayed a significant correlation. The significance of this factor must be taken in light of the limited number of cats in our study. Cats that were anemic tended to have a lower overall survival and survival after radiation therapy. Implications of this trend must be taken in light of the low number of cats with CBC results. However, other studies have also commented on body weight and anemia as contributing to both response to therapy and overall survival. 8,36
Limitations of our study include potential case selection bias due to a low case number and the retrospective nature of this work. A low case number may have caused a limited or skewed representation of the population being studied. A retrospective study design means that it is virtually impossible to identify an adequate control population. Case selection bias may have also occurred due to the type and number of clients who seek rescue therapy for relapsed feline lymphoma. None of the cats in this study had post-mortem evaluation, potentially limiting the full determination of radiation adverse effects. Low case numbers also make multivariate or other correlative statistics nearly impossible. Clearly, a prospective study including a larger number of cats would improve the understanding of efficacy and safety of abdominal radiation therapy for feline alimentary lymphoma.
Clinical significance of this study, despite the small number of cats, is that abdominal radiation therapy in the rescue setting for feline GI lymphoma appears to be well tolerated at the dose of radiation used in this study. Cats may experience an apparent clinical response and potentially longer survival due to this therapy protocol. As the diagnosis of alimentary lymphoma continues to increase in the post-FeLV vaccine era, 42 having an additional therapeutic modality available may help improve outcomes for this form of feline lymphoma.
