Abstract

Pathophysiology — and its diagnostic relevance
The pleural space is a potential space surrounding the lungs. It is formed by the visceral and parietal pleura, which are serous membranes that line the outer surface of the lungs and inner surface of the thoracic cavity, respectively. A small volume of fluid within the pleural space lubricates movement of intrathoracic structures during respiration. Pleural fluid formation and drainage is governed by Starling's forces. Any process that disrupts capillary or interstitial hydrostatic or oncotic pressures, lymphatic drainage or vessel integrity can result in net fluid accumulation, as can extension of pathological fluid accumulation from adjacent structures (eg, parapneumonic spread of infection). Because the mediastinum is fenestrated in cats (and dogs), fluid usually accumulates bilaterally unless it becomes loculated due to inflammation. 1 Ongoing fluid accumulation eventually results in pulmonary atelectasis and hypoventilation. It is stated that 30 ml/kg of pleural effusion causes subtle dyspnoea and 60 ml/kg results in obvious dyspnoea, 2 although evidence for these figures is not presented.
In human medicine, pleural effusions formed by an imbalance between oncotic and hydrostatic forces are defined as transudates and those resulting from increased capillary permeability as exudates. There is a good correlation between biochemical characteristics of the fluid and serum, principally protein and lactate dehydrogenase (LDH) measurements, and pathophysiology, such that these values can be relied on to predict fairly accurately whether fluid has accumulated due to a transudative or an exudative process. 3
MULTIMEDIA
Four video recordings to aid recognition of pleural effusion (ie, showing respiratory patterns in cats with and without pleural effusion and jugular pulsation) are included in the online version of this article at
This is not the case in veterinary medicine where controlled studies correlating fluid characteristics with aetiology are limited. 4–9 Effusions in animals are typically classified according to their cell count and protein content as transudates (low protein, low cell count), exudates (high protein, high cell count) or modified transudates (intermediate) (Table 1). 10 The relationship between fluid characteristics and the underlying pathophysiological process is less well defined and there is often overlap between fluid type and aetiology (Table 2). Chylous pleural effusions (chylothorax) provide a good example. They result from leakage of chyle from the lymphatics and can occur with several underlying disease processes including congestive heart failure (CHF), neoplasia and trauma; in cats, however, they are commonly idiopathic. 11 Using total protein (TP) and total nucleated cell count (TNCC), chylous effusions may be classified as modified transudates or as exudates. 12
Classification of pleural fluid based on total protein (TP) concentration and total nucleated cell count (TNCC) 10
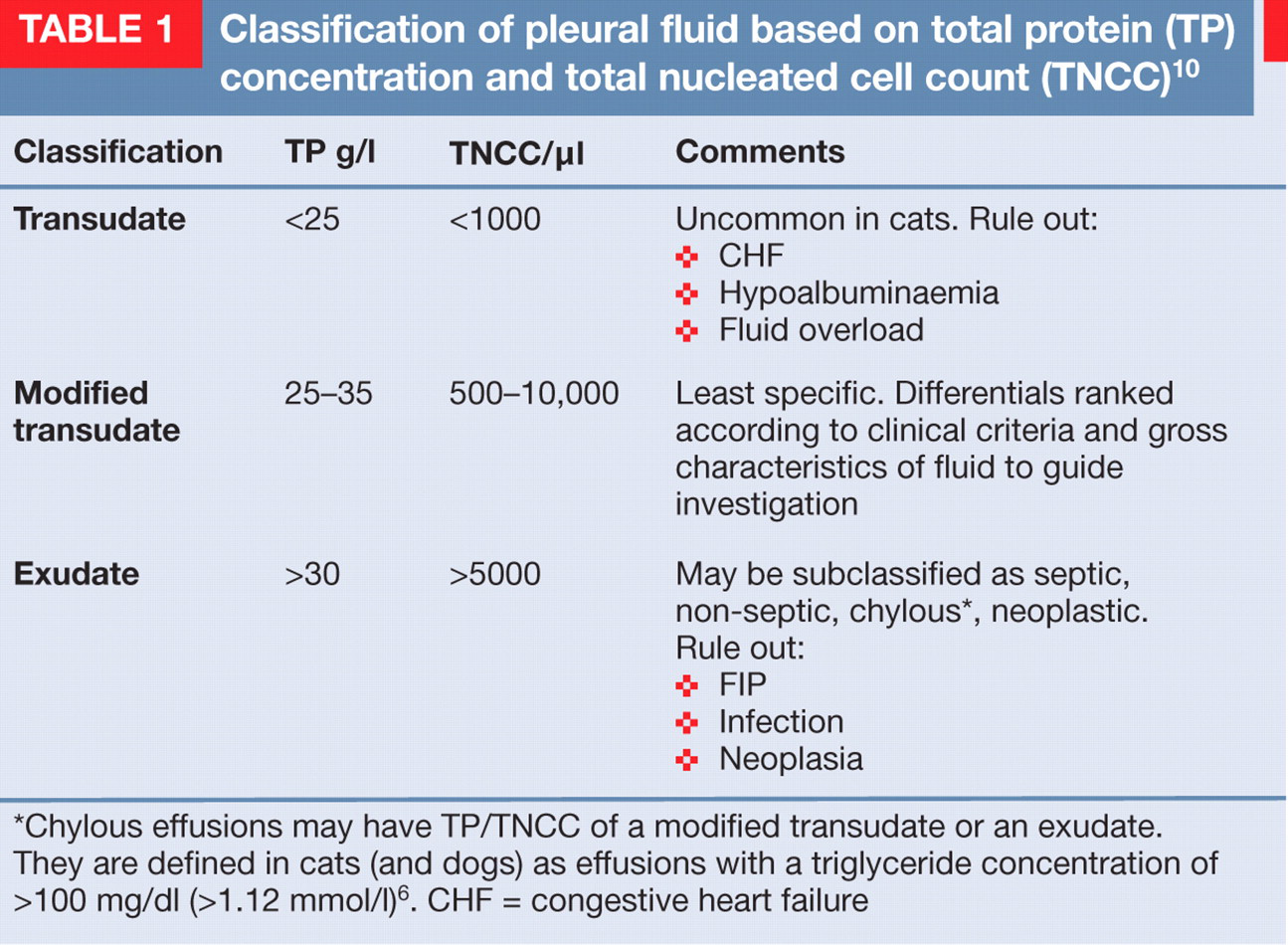
Chylous effusions may have TP/TNCC of a modified transudate or an exudate. They are defined in cats (and dogs) as effusions with a triglyceride concentration of >100 mg/dl (>1.12 mmol/l) 6 . CHF = congestive heart failure
Clinical features of common causes of pleural effusion in cats

FIP = feline infectious peritonitis, TP = total protein, TNCC = total nucleated cell count
More useful clinically are the gross characteristics, triglyceride and cholesterol concentrations of fluid and serum, and the presence of chylomicrons. On cytology small lymphocytes predominate initially but neutrophil and monocyte counts increase over time. 11 The results of fluid analysis are useful only in as much as they elucidate the underlying disease process. It is important to appreciate their value and limitations (discussed later). In cases of pyothorax, neoplasia and feline infectious peritonitis (FIP), fluid characteristics may be diagnostic, whereas determining that the fluid is a modified transudate is non-specific and must be interpreted in the light of other clinical data.
Aetiology
Fortunately, the list of common causes of a moderate to large volume pleural effusion in the cat is relatively short. While incidence data are not available, FIP, CHF, pyothorax and neoplasia together accounted for 88–100% of 265 cases with a confirmed aetiology (Table 3). 4–9 In two studies, 9/19 cats (12.5% of total) and 3/7 cats (15% of total) were defined as having idiopathic chylothorax where an aetiology could not be determined despite investigation. 6,7
Summary of published case series of feline pleural effusion demonstrating the major underlying aetiologies and disease processes

FIP = feline infectious peritonitis, CHF = congestive heart failure
CHF
Pleural effusion can result from left and/or right CHF causing a transudate, a modified transudate or a chylous effusion. Increased ventricular diastolic pressure results in increased capillary hydrostatic pressure in the systemic and/or pulmonary circulation. The visceral pleura drain into the pulmonary veins in both cats and dogs but pleural effusion from left CHF (LHF) seems to be more common in cats than dogs. 1,13 As parietal pleural veins drain into the systemic venous circuit, right CHF (RHF) may also cause pleural effusion. RHF is a fairly common cause of chylothorax in the cat. 6,14,15 Chylothorax in this setting presumably results from increased pressure in the major lymphatics as the thoracic duct terminates in the left external jugular veins or jugulosubclavian angle.
Diverse underlying cardiac problems can result in heart failure. In cats heart failure is often due to diastolic dysfunction. 13 Common cardiomyopathies (hypertrophic, restrictive, unclassified, thyrotoxic) should be ruled out first. While cardiac signs are common in hyperthyroid cats (eg, tachycardia, murmur, gallop), earlier diagnosis has resulted in a decrease in the frequency of CHF in cats with thyrotoxicosis. 16,17 One study reported a significant decline in the prevalence of CHF in hyperthyroid cats from 12% in 1983 to 2% in 1993. 16
Neoplasia
Mediastinal, bronchopulmonary or primary pleural neoplasia may cause pleural effusion with the characteristics of a modified transudate, an exudate or a chylous effusion. In published case series, 26–61% of feline pleural effusions were associated with neoplasia (Table 3). Mediastinal lymphoma accounted for the majority of neoplasia-associated pleural effusions (60–79%) reported in cats between 1972 and 1995. 4,5,7,8 Although the prevalence of feline leukaemia virus (FeLV) associated mediastinal lymphoma has declined with that of FeLV, an increased risk of developing mediastinal lymphoma has been documented in young cats of the Siamese breed group, independent of their FeLV status (Table 2). 18 Other neoplastic and non-neoplastic mediastinal masses such as thymoma, thymolipoma, thymic hyperplasia and developmental anomalies should be considered where mediastinal lesions are identified. 19–21
Definitive diagnosis of mediastinal lesions is essential to determine prognosis and guide treatment options. In addition to fluid production, mediastinal lesions may cause dyspnoea due to a space-occupying mass effect. Regardless, they are included here because they often occur concurrently, presentation and diagnostics are similar and it can be difficult to differentiate a mass from fluid using radiology alone. Primary and, less commonly, secondary bronchopulmonary carcinoma makes up the bulk of the other neoplasms. Primary pleural neoplasia is rarely reported in the cat. 22
Infectious pleuritis
Infectious pleuritis is caused by obligate and facultative anaerobes of oropharyngeal origin in more than 80% of cases, resulting in accumulation of a purulent exudate (pyothorax). Parapneumonic spread of infection following colonisation of lung tissue by oropharyngeal flora seems to be the most frequent cause of feline pyothorax. 23
FIP
The effusive form of FIP is associated with widespread vasculitis. Subsequent exudation of high protein effusion can occur in peritoneal, pleural or pericardial cavities.
Other
Other documented causes of pleural fluid accumulation in the cat include trauma, coagulopathy, peritoneopericardial diaphragmatic hernia, uraemia, pulmonary thromboembolism, lung lobe torsion, possible extension from a perinephric pseudocyst, pancreatitis, glomerulonephropathies and Aelurostongylus abstrusus infection. 2,7,8,24–29 Trauma, which can result in effusion secondarily to haemorrhage, diaphragmatic hernia, thoracic duct rupture or, rarely, urinothorax, 7 was the underlying cause in 8% of cases in one study. 30 Although heartworm disease is often listed as a possible aetiology in the cat, evidence that pleural effusion occurs as a consequence of natural infection in this species is limited. 31,32
Undetermined
In some cases the aetiology may remain elusive because the mechanism is recognised as being idiopathic (for chylothorax) or because of lack of access to advanced diagnostic techniques. Pulmonary thromboembolism is a common cause of pleural effusion in humans but has been documented infrequently as a cause in the cat where there is limited availability of computed tomography (CT) angiography. 2 Even so, 15–20% of pleural effusions in humans remain undiagnosed even after pleural biopsy. 33
A practical classification to direct the clinical investigation
A clinically useful classification for pleural effusion in the cat is presented in Table 2. Based on this, the quality of data collection from a minimally invasive examination can be maximised and differential diagnoses ranked for the patient. This directs the clinical investigation and facilitates expedient identification of the underlying aetiology so that owners can be informed regarding management options and the prognosis for their pet.
Recognising pleural space disease
Observation
Observation of the respiratory pattern in a dyspnoeic patient is the first step in localising the problem. The respiratory rate and depth, the phase of respiration that is laboured (inspiratory, expiratory or both), the presence or absence of audible respiratory noise (stridor, stertor, wheeze) and the presence or absence of respiratory noises on auscultation (referred large airway sounds, breath sounds, wheezes, crackles) should be noted (Table 4).
Assessment of respiratory patterns in dyspnoeic cats

Cats with significant pleural space disease adopt a sternal position with abducted elbows. A restrictive (rapid, shallow) respiratory pattern with increased inspiratory effort is typical (see video 1,
Upper respiratory tract (URT) obstruction is the other major cause of inspiratory dyspnoea. In this case respiration is usually slow and deep (obstructive) and accompanied by other URT signs such as stertor or stridor (see video 2,
Auscultation
Auscultation helps to distinguish pleural space disease from pulmonary parenchymal disease when there is a restrictive pattern. Breath sounds are decreased or absent with pleural space disease. Differential diagnoses then include pleural effusion, pneumothorax, intrathoracic mass or diaphragmatic hernia. Where there is effusion, the reduction in breath sounds is often more pronounced ventrally and a fluid line may be appreciated on auscultation or percussion. Concurrent pulmonary oedema may contribute to the dyspnoea in LHF and result in auscultable crackles dorsally. Pleural effusion or pericardial effusion can cause muffled heart sounds. An intrathoracic mass or focal accumulation of fluid can displace the cardiac apex beat. With diaphragmatic hernia, borborygmi may be auscultated in the thorax.
In contrast, pulmonary parenchymal diseases severe enough to cause restrictive respiration are characterised by increased lung sounds, crackles or wheezes on auscultation.
Auscultation over the trachea and larynx should be performed routinely to differentiate sounds referable to URT obstruction from sounds emanating from the LRT.
Percussion
Thoracic percussion is more difficult to perform in cats than dogs because of their small stature. If tolerated, it can be a useful diagnostic tool. Percussion is performed by firmly tapping one or two fingers placed against an intercostal space and comparing the resonance of the sound produced at several different locations over the thorax bilaterally. A more resonant sound dorsally (‘drum-like’) provides evidence for pneumothorax. A less resonant or dull sound ventrally provides evidence for loss of pulmonary aeration.
Confirming pleural space disease
The presence of pleural effusion can be rapidly and noninvasively confirmed with either a single dorsoventral (DV) radiographic view or thoracic ultrasonography. Maintaining the patient in sternal recumbency avoids positional atelectasis and requires minimal restraint. A single DV view will confirm pleural space disease and differentiate between pleural effusion, pneumothorax or diaphragmatic hernia. The increased oxygen demand associated with additional restraint for symmetric positioning is not usually justified prior to thoracocentesis as significant pleural space disease will be apparent, even from slightly rotated views. Horizontal beam (lateral) views with the cat in a standing position similarly require minimal restraint.
The radiographic signs of pleural effusion are listed below. 34 Unilateral effusion should raise the index of suspicion for pyothorax or chylothorax. 11,23
Thoracic ultrasonography is similarly useful for confirming a moderate to large volume pleural effusion, diaphragmatic hernia and, in the hands of a skilled operator, pneumothorax. 35
Interlobar fissure lines
Rounding of the lung margins at the costophrenic angles
Retraction of the lobar borders from the thoracic wall
Widening of the mediastinum
Scalloping of the lung margins at the sternal border
Effacing of cardiac silhouette (silhouette sign)
Dorsal displacement of the trachea
A dorsoventral radiographic view rapidly confirms moderate to large volume pleural effusion and requires minimal restraint

Approach to the dyspnoeic cat with pleural effusion
The priorities for the clinician managing a cat with pleural effusion are discussed in the following sections beginning, first and foremost, with stabilisation of the patient. Thereafter, data obtained from signalment, history and physical examination is used to rank the list of common differentials. This ranking is further informed by the gross characteristics of fluid obtained at thoracocentesis.
Stabilisation
It is not unusual for dyspnoea to be subtle or absent prior to arrival at the clinic. As pleural fluid accumulates the haemoglobin saturation falls gradually (Fig 1). Combined with reduced activity, this allows the cat to compensate initially. When the arterial partial pressure of oxygen (PaO2) falls below 60 mmHg, haemoglobin saturtion falls precipitously. 36 This will be reached gradually as underlying disease progresses — however, an acute increase in oxygen demand, such as that associated with handling, can precipitate decompensation. By the time patients with pleural effusion show signs of respiratory distress (open-mouth breathing, agitation, vocalisation, extension of the neck, failure to maintain sternal positioning) little reserve remains.
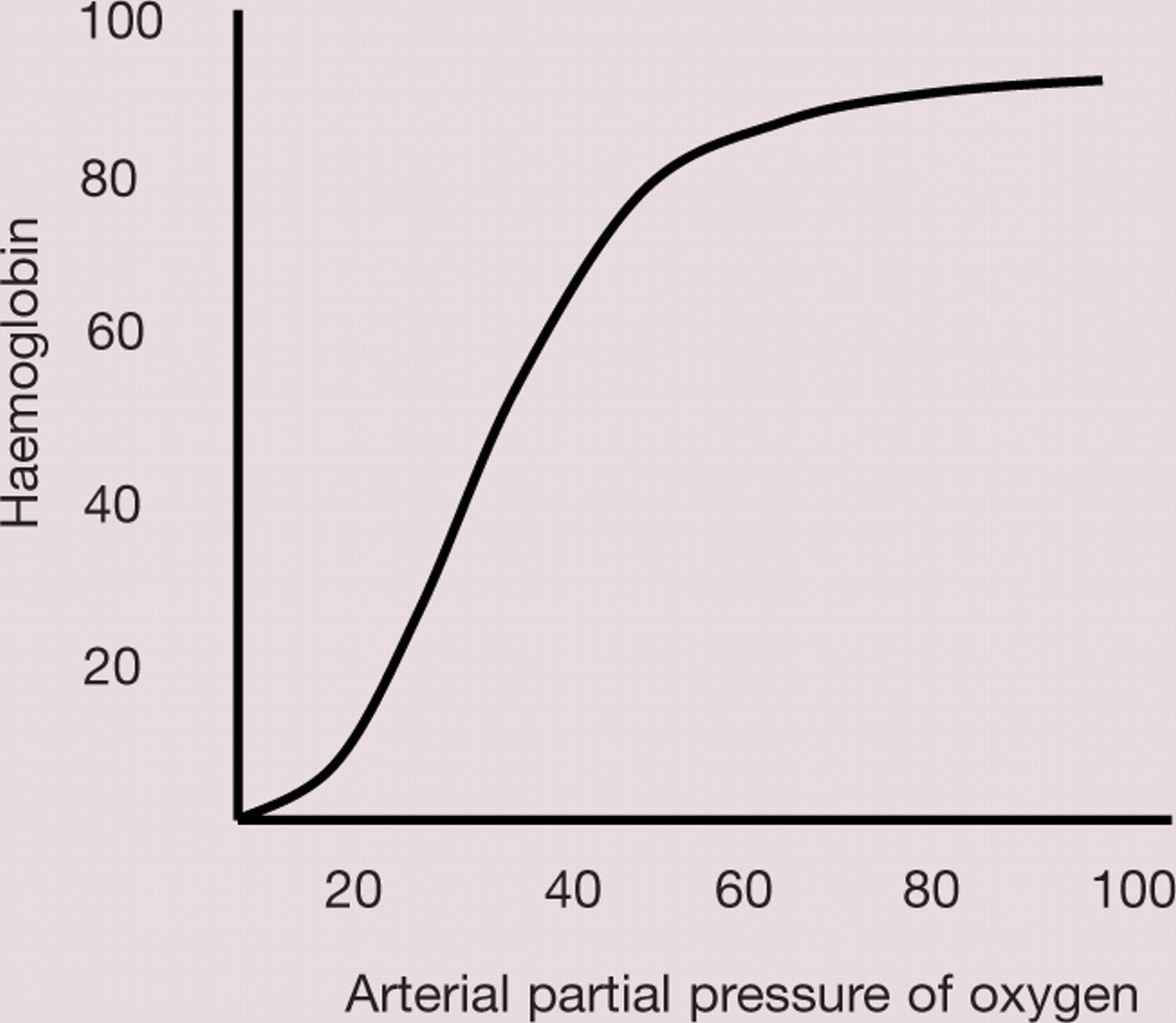
Oxygen dissociation curve (see text for explanation) 36
Stabilisation techniques
Oxygen chamber

Mask delivery

Flow-by

Thus cats showing clinical signs of pleural space disease have significantly compromised respiration and are at risk of respiratory failure. This reinforces the importance of triage at reception. All cats should be observed on arrival. Where respiratory distress is noted or suspected, immediate stabilisation is indicated. The choice of the various techniques (see box on page 698), and order in which they are carried out, depends on assessment of the individual patient.
Dependent on the underlying disease process, other considerations for patient stabilisation include identification and correction of hypothermia, hypotension, hypoglycaemia, and fluid and electrolyte imbalances.
History
Historical abnormalities may relate to the presence of the pleural effusion and/or the underlying disease. Non-specific signs such as reduced appetite, weight loss or lethargy are common. 7,11,23 Dyspnoea is noted in 60–80% of cases and coughing in up to 30% of cats with chylothorax and pyothorax. 7,11,47 Coughing may be associated with the presence of effusion or it may be related to the aetiology (eg, concurrent bronchopulmonary infection or neoplasia, or airway compression from a cranial mediastinal mass). Coughing due to cardiac disease is not as commonly recognised in cats as dogs, but it can occur. 48 It is the authors' experience that owners may not recognise coughing in cats, but rather attribute this sign to ‘hairballs’ or dry retching. Careful questioning is required.
Signalment — an aid to ranking common differentials
Signalment data cannot be used to exclude an aetiology but may assist in ranking the common differentials (Table 2).
In young cats, effusive FIP, mediastinal lymphoma and pyothorax are likely, whereas the median age for developing bronchopulmonary neoplasia, thymoma and decompensated thyrotoxic cardiomyopathy is 11–13 years. 39–41 Approximately 70% of FIP cases occur in cats less than 1 year of age. 42
An increased risk of developing FIP has been demonstrated in purebred and in entire cats and there is evidence of heritability in some lines. 42,43 However, FIP is often diagnosed in domestic crossbreeds, and older cats may sometimes be affected.
Cats of the Siamese breed are overrepresented for both mediastinal lymphoma (young cats) and chylothorax. 11,18,44
Familial cardiomyopathies have been identified in many breeds, including the Maine Coon, Ragdoll, British and American Shorthair, Bengal, Sphynx, Norwegian Forest and Siberian. 45 Less information is available regarding breed predispositions for other types of cardiac disease. The Chartreux breed has been identified as being overrepresented for tricuspid dysplasia. 46
While congenital aetiologies are more likely in young cats, cats with congenital, heritable and acquired cardiac disease can develop heart failure at any age.
Some acquired cardiac problems are age-related (eg, thyrotoxic cardiomyopathy).
A previous history of upper respiratory tract infection in a young cat would raise suspicion for pyothorax. 23 Recent history of a multicat environment and/or a stressor, such as neutering or rehoming, in a young cat would increase suspicion for FIP. 42 Owners of cats >8 years old should be questioned about signs related to hyperthyroidism such as polyphagia, weight loss, polyuria, polydipsia and gastrointestinal signs. Oesophageal signs including dysphagia, regurgitation and ptyalism may reflect compression of the oesophagus from an intrathoracic mass. Haemothorax would be less likely in cats without outdoor access or exposure to anticoagulant rodenticides. There is conflicting data regarding whether pyothorax is more common in outdoor or indoor cats. 7,49
Clinical examination
The importance of gentle handling of cats in respiratory distress cannot be overemphasised. Struggling must be avoided. Some tests, such as rectal temperature, may need to wait until the patient is stable. Nonetheless, useful information can be gained non-invasively to help rank the common aetiologies.
Examination of the cardiovascular system
Mucous membranes should be examined for colour and capillary refill time. Jugular veins can be evaluated for distension and pulsation, with the cat sitting or standing. The neck should be moderately extended and the overlying hair wetted or clipped. The jugular veins are not distended in healthy cats. Distension occurs due to increased systemic venous pressure or venous occlusion between the jugular vein and the right heart.
Normal jugular venous pulsation does not ascend higher than a third of the way up the neck in healthy cats. Jugular pulses must be differentiated from carotid artery pulsations, which may be transmitted through adjacent soft tissues in thin or agitated cats. A true jugular pulse will disappear if the jugular vein is occluded below the level of the visible pulse; pulsation will continue if it is being transmitted from the carotid artery (see video 3,
Changes in cardiac rate, rhythm and heart sounds should be assessed carefully. In one study, the majority of cats with dyspnoea from cardiac disease had an abnormality on cardiac auscultation. 53 Increased heart rate and the presence of a murmur or gallop sound can be useful indicators of cardiogenic causes of pleural effusion. However, the possibility that the cat has structural cardiac disease but is not in heart failure — that is, the pleural effusion is non-cardiogenic — should also be considered. It is important to note that the absence of a murmur does not rule out cardiac disease. In one study, the sensitivity and specificity of the presence of a heart murmur for diagnosing cardiomyopathy were 31% and 87%, respectively. 54
Clinical examination findings that make cardiogenic causes of pleural effusion more likely include a murmur, gallop, arrhythmia, crackles dorsally, jugular distension and jugular pulsation.
Examination of other body systems
Poor body condition score is a non-specific indicator of underlying debilitating disease in cases that are not trauma or toxin-associated. Pyrexia may be a feature of FIP, pyothorax or neoplasia. Hypothermia may indicate sepsis in cases of pyothorax, especially if there is also bradycardia and hypoglycaemia. Hypothermia in conjunction with tachycardia and pale mucous membranes may indicate hypovolaemia. The cat should be examined for signs of trauma and haemorrhage at other sites. The thyroid lobes should be palpated routinely. Cranial mediastinal mass lesions may cause reduced compressibility of the anterior rib cage and may be palpable at the thoracic inlet. 52,55 Paraneoplastic syndromes associated with intrathoracic mass lesions include myasthenia gravis, exfoliative dermatitis and polymyositis. 19 Pulmonary carcinoma in cats has a propensity to metastasise widely to unusual locations including the eyes, digits and skeletal muscles. 56 Cats with intrathoracic lesions may present with lameness due to digital metastases or hypertrophic osteopathy. In young cats, FIP is a major differential and signs potentially referable to this problem should be noted including abdominal effusion, ocular changes (eg, uveitis, keratic precipitates) and neurological signs.
Gentle abdominal palpation and assessment for a fluid wave may be possible. Simultaneous peritoneal and pleural effusion can occur with systemic disease processes or following extension from one cavity to the other. In the cat, double effusion increases suspicion for FIP, RHF, neoplasia, haemorrhage, systemic inflammation or infection, nephrotic syndrome and ruptured diaphragm. Of cats with effusive FIP, 62% have peritoneal effusion, 17% have pleural effusion and 21% have double effusions. 42 Extension of pyothorax into the abdominal cavity is rare. 57 Glomerulonephropathies are a rare cause of pleural effusion and affected cats typically show other signs of nephrotic syndrome including ascites and subcutaneous oedema.
Thoracocentesis
Gross characteristics of pleural effusion
Pyothorax fluid before (left) and after (right) centrifugation

Chylous pleural effusion

Serosanguineous effusion

Transudate

FIP effusion. (inset) Note the viscosity

Fluid analysis — value and limitations
Fluid analysis is variably useful in determining aetiology and the results must be interpreted in the light of clinical data. Samples collected in EDTA and plain tubes, and direct smears should be submitted for TNCC, TP, differential cell count and cytology. Additional testing will be directed by clinical data and gross evaluation of the fluid.
Malodorous effusions
A foul smell is a very useful indicator of anaerobic infection, which is typical in over 80% of pyothorax cases. 23 The fluid is usually opaque and creamy but can be pink, green-tinged or sanguineous, and flocculent material is often present. Aerobic and anaerobic culture and cytology, including Gram staining, is indicated. Fluid associated with infection typically has a high protein content (>30 g/l) and TNCC (>7000/μl), with neutrophils predominating (>85% of TNCC), and is classified as an exudate. Commercial anaerobic specimen collectors are available (eg, Vacutainer Anaerobic Specimen Collector, BD Biosciences). Where frank pus is aspirated, other tests are not necessary. Lack of odour does not rule out an infectious cause; 20% of cases of infectious pleuritis, particularly in kittens, are caused by unusual bacterial, fungal or protozoal pathogens. 23 Cytology and culture will assist in identifying these causes. Systemic toxoplasmosis can be associated with a large volume pleural effusion in the cat. 58
Milky effusions
Opaque and milky or pink fluids should be centrifuged. Chylous fluids form a cream layer upon standing but do not become clear after centrifugation because the opacity is due to lipid rather than cells or debris. The TP and TNCC lead to classification as a modified transudate or an exudate. Lipid in chylous effusions artefactually increases protein estimation when determined by refractometry. In addition to small lymphocytes or neutrophils and lipid-laden macrophages, refractile lipid droplets (chylomicrons) are often seen on cytology. In chylous effusions the pleural fluid triglyceride content is greater than the serum triglyceride concentration, while the pleural fluid cholesterol level is less than or equal to serum cholesterol. If only pleural fluid is available then triglyceride >100 mg/dl (>1.12 mmol/l) confirms chylothorax. 6 The investigation should be directed to rule out known causes of chylothorax in the cat — principally CHF, neoplasia and trauma. 11 It should be expected that many cases of chylothorax in the cat will be considered idiopathic despite investigation.
Pseudochylous effusions resemble chylous effusions grossly and may not clear after centrifugation, but do not contain chyle. They are associated with chronic pleural disease and have a higher cholesterol content than serum, while the pleural fluid triglyceride level is less than or equal to the serum triglyceride. It is thought that cholesterol enters the pleural space during acute pleural inflammation and accumulates within pleural fluid over time because of a change in lipoprotein binding characteristics that impedes transfer out of the pleural space. 3 Pseudochylous effusions seem to be rare in cats. 44
Serosanguineous effusions
Where fluids appear bloody, the clinician must differentiate between frank haemorrhage into the pleural space (haemothorax), and blood contamination of an effusion with a different aetiology (haemorrhagic effusion). Fluid can appear bloody with a haematocrit of <5%, 3 whereas haemothorax is variably defined as fluid with a haematocrit of at least 25%, or 50% of the peripheral blood haematocrit. 3,59 Where haemorrhage has been present for more than 1 h no platelets will be seen on pleural fluid smears and the blood will be defibrinated so it will not clot in a plain tube. Causes of haemothorax include trauma, anticoagulant rodenticide intoxication and neoplasia. History and physical examination data may support one of these aetiologies. Cats with acute haemorrhage into the pleural space sufficient to restrict respiration should present with obvious signs of hypovolaemia (pale mucous membranes, weak pulses, tachycardia, hypothermia).
Distinguishing cardiogenic, neoplastic and atypical infectious effusions
Translucent effusions
Where the fluid is clear or yellow, a TP estimation should be obtained cage-side. The protein scale on handheld refractometers is inaccurate at <25 g/l; tables are available to convert the urine specific gravity (USG) reading for the sample to the protein concentration between 10 and 25 g/l. 12 Low protein fluids are uncommon in cats and have few differentials. 9,29 If the protein concentration falls into the transudate range (<25 g/l) then serum albumin should be measured. If serum albumin is >15 g/l then pleural effusion due to decreased plasma oncotic pressure can be ruled out. Fluid overload can easily be ruled out from the history. CHF remains the major differential diagnosis and echocardiography is the most useful test here.
For translucent, yellow effusions with a protein content >30 g/l, FIP is a major differential. The effusion associated with FIP is typically viscous (evaluated by expelling the fluid from a syringe, see page 701), froths on agitation due to its high protein content, and clots on standing. While the protein content is consistent with an exudate, the TNCC, comprising neutrophils and macrophages, is low, consistent with a modified (<5000 cells/μl) or pure (<1000/μl) transudate. 60 The single most useful test for ruling in FIP as a cause of pleural effusion is the use of immunofluorescence to detect feline coronavirus (FeCoV) antigen in macrophages. 61 This test has a positive predictive value (PPV) of 100%. It is less useful for ruling out infection, with a negative predictive value (NPV) of 57%. A negative result may occur in a cat with FIP if there are low numbers of macrophages or if epitope masking by patient antibody occurs. Rivalta's test can be used (PPV 86%, NPV 97%) where immunofluorescence is not available, or to support a negative finding on immunofluorescence. 61 This test identifies exudates based on their ability to retain their shape in a dilute acetic acid solution. Where less invasive tests are inconclusive, and a definitive diagnosis is required, immunohistochemistry of tissue biopsies can be used to confirm FIP.
Additional tests
Imaging
Post-drainage chest radiographs, including lateral views, should be obtained to detect abnormalities that may have been effaced by fluid or obscured by atelectasis. The pulmonary parenchyma and vasculature, cardiac silhouette and mediastinum should be evaluated. The presence of restrictive pleural disease should be noted. The vertebral heart score should be determined. 64 Enlargement of the cardiac silhouette usually indicates cardiomegaly but pericardial effusion or rare disorders such as pericardioperitoneal diaphragmatic hernia should be considered. Echocardiographic examination will rapidly distinguish between these possibilities and is the diagnostic modality of choice to rule out decompensated cardiac disease. Ascribing significance to subtle echocardiographic changes can be challenging. The atria dilate in response to both pressure and volume overload. Although there are exceptions, if the atria are not enlarged, then CHF as a cause of pleural effusion is ruled out. Where RHF is suspected a cursory abdominal ultrasound examination is useful to detect ascites and enlargement of the caudal vena cava and hepatic veins. Where pericardial effusion is detected concurrently with pleural effusion it is likely to be a consequence of the same underlying disease process, such as CHF, FIP or neoplasia, rather than a cause of the pleural effusion, since pericardial effusion rarely causes tamponade in cats. 65
The lung fields should be inspected for evidence of underlying disease consistent with CHF, neoplasia and abscessation. Radiographic signs of pulmonary oedema in the cat are variable. They include interstitial or alveolar opacities in the perihilar area, dorsocaudal lung fields, more focally caudal to the heart or unevenly distributed throughout the lung fields. Patchy infiltrates from pulmonary oedema can be difficult to differentiate from other lung diseases including neoplasia (primary and metastatic) and infection. 66–68 Rounding of the borders of the lung lobes is consistent with pleural fibrosis seen with long-standing or irritant effusions. Fibrosing pleuritis is a cause of persistent dyspnoea after pleural fluid drainage.
Diagnosing mediastinal mass lesions, which are often accompanied by pleural effusion, can be challenging using radiography alone since mass lesions may be effaced by fluid. On a lateral view, dorsal displacement of the trachea and caudal displacement of the carina (which normally sits at intercostal space 6 in the cat) can occur with large volume effusion, cardiomegaly or a cranial mediastinal mass. On a DV or ventrodorsal (VD) view in healthy cats the width of the mediastinum is usually less than twice the width of the spine. 52 DV or VD projections are more useful than lateral views to differentiate mediastinal mass lesions from pulmonary masses. Cranial mediastinal masses displace the carina and lungs caudally and can cause lateral deviation of mediastinal structures.
Investigative approach to pleural effusion

Ultrasonographic evaluation of non-cardiac intrathoracic structures and guided fine-needle aspiration (FNA) biopsy or core biopsy are useful for identifying neoplasia, pulmonary abscessation, benign mass lesions and diaphragmatic hernia. 69 Pleural fluid provides an acoustic window, enhancing visualisation. The presence of concurrent peritoneal effusion should be investigated. Peripheral, focal, non-aerated pulmonary lesions are suitable for ultrasound-guided FNA biopsy. In one study of 56 feline and canine patients in which this procedure was performed no complications were noted and the sensitivity of this technique for diagnosis was 91%. 69 Although more limited in availability, thoracic CT is an excellent modality to detect, characterise and evaluate the extent of intrathoracic nodules or masses. 70 Similarly, CT-guided FNA biopsy or core biopsy, thoracoscopic biopsy or exploratory thoracotomy may be indicated. 71 The most common complications associated with CT-guided or ultrasound-guided core biopsy are pneumothorax and haemorrhage.
Serum testing
Routine haematology, biochemistry, urinalysis and retrovirus testing are likely to be relatively low yield in determining the underlying disease process compared with analysis of the effusion, but form part of the minimum database to guide patient management and should be carried out once the cat is stable. Other tests, including total serum T4 and Dirofilaria immitis antigen and antibody detection, may be indicated depending on the case. An exciting recent development is the validation of circulating natriuretic peptide levels to differentiate between cardiac and non-cardiac causes of respiratory distress in cats. N-terminal probrain natriuretic peptide (NT-proBNP) has been shown to have sensitivity and specificity of around 90% for detecting myocardial disease in this setting. 72,73
Case notes
Thoracic ultrasonographic screening was planned after stabilisation with supplemental oxygen. The cat was placed in an oxygen chamber on admission and monitoring of respiratory rate and depth at 5 min intervals was commenced. Pleural effusion was confirmed on thoracic ultrasound.
Thoracocentesis was performed and yielded 150 ml of non-odorous, moderately cloudy, straw-coloured fluid. Respiratory rate and effort were reduced, but the patient remained dyspnoeic (see video 4,
Fluid analysis should be carried out including cell count and differential, protein content and cytological analysis. Other parameters that would be useful to measure to differentiate CHF from neoplasia include LDH, pH and NT-proBNP.
A complete blood count, feline immunodeficiency (FIV)/FeLV tests, serum biochemistry profile and urinalysis are also indicated to look for markers of systemic disease.
These findings are most supportive of a neoplastic aetiology. Differential diagnoses include primary or metastatic pulmonary neoplasia, although the size of the pulmonary nodules is more suggestive of primary pulmonary neoplasia. An inflammatory process (eg, mycotic granuloma, FIP, toxoplasmosis, atypical bacteria) is possible, but less likely.
The findings are consistent with a neoplastic effusion of epithelial origin. Exfoliating carcinoma cells appear to be present, the main differential being a pulmonary carcinoma.
Because of the poor prognosis the cat was euthanased. Post-mortem examination was permitted and the resultant histopathological diagnosis was pulmonary carcinoma.
Fluid analysis results



KEY POINTS
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
