Abstract
Introduction
Pain in cats has many negative effects, both physiological and emotional. 1,2 It is now accepted that there is no such thing as ‘good pain’ following surgery and during treatment for trauma or disease — eg, pain that inhibits potentially deleterious movement after surgery. Pain delays recovery, impacts negatively on a patient's wellbeing, and disturbs the bond with its owner and also the veterinary team. 1,3
Studies have looked at the use of non-steroidal anti-inflammatory drugs (NSAIDs) for acute, especially perioperative, pain in cats. 4–7 Surveys have shown clinicians were more likely to treat pain in dogs than cats, 8,9 as a result of difficulties in recognising pain, lack of knowledge concerning the use of analgesics, and fear of drug side effects in cats. Less has been published on the management of chronic pain in cats, but it is recognised that signs may be subtle and include withdrawing from attention, decreased mobility, reduced interactions with humans and other animals, poor appetite and aggression. 10–12
Chronic pain can be regarded as pain that has persisted for more than 2–3 weeks, often persists months or years, and may continue beyond the anticipated healing time. Importantly, chronic pain can become dissociated from the inciting cause and be maladaptive, such that the degree of pain does not necessarily correlate with the pathology observed or perceived by the individual, and is not associated with healing. 12 Multimodal analgesia is commonly advocated, but it is becoming evident that NSAIDs will play a key role in managing chronic feline pain, especially musculoskeletal pain, just as they do in humans and dogs. 10,13–16 Until quite recently, while many NSAIDs have been available to treat dogs with degenerative joint disease (DJD), 17 only a restricted range has been licensed for short-term (up to a few days) use in cats. At the time of writing, at least one NSAID — meloxicam — has been licensed for long-term use in cats in many regions of the world, transforming our ability to manage pain in this species, and a second — robenacoxib — has been licensed for up to 6 days of therapy in cats (see Table 1, page 529). There is little doubt that others will become licensed for long-term use in the future, due to the recognition of the need and value for such NSAID therapy in this species. 10,11,13–15,18
NSAIDs licensed for systemic use in cats (NB not all drugs are licensed in all regions and veterinarians should refer to local information and regulations)

Clinicians are aware of their duty to promote animal welfare and relieve suffering, but are also often reminded of Hippocrates' advice to ‘first do no harm’. This is often rightly used to question whether an intervention will actually do more harm than good, and to withhold that intervention when doubts exist. However, we need also to recognise that withholding treatments such as analgesics can sometimes cause the greater harm, because we are no longer addressing the pain and suffering the animal is enduring. In drawing up these guidelines, the international panel of experts' purpose has been to review the current literature on long-term NSAID use in cats, and to provide practical guidance on their use. The overarching aim is to encourage more widespread and appropriate NSAID therapy, when cats will benefit from the use of these drugs. However, most of our knowledge of therapeutic mechanisms or adverse drug reactions is extrapolated from work in other species, as there is a paucity of published data relating to cats.
Common causes of chronic pain and inflammation in cats
One of the difficulties in managing pain in cats is its initial recognition. It is important, therefore, to be aware of common causes of pain and to have a high index of suspicion for signs and behaviours potentially related to pain. If something is painful to us, it is likely to be painful to a cat.
Degenerative joint disease
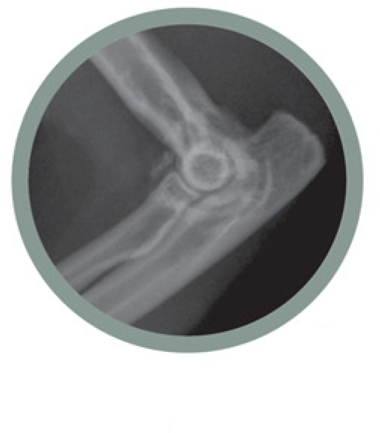
The most common cause of chronic feline pain is thought to be DJD, and this has been the subject of a number of important studies in the past 10 years. 11,15,16,18–24 From these studies, it is clear that DJD is very common, with radio graphic changes affecting up to 60–90% of cats (Figs 1 and 2), 18,24 that it affects both the spine and the appendicular joints, and that it occurs especially commonly in older patients. 18,24 The hips, stifle, shoulder, elbow, tarsus and spine are the most common sites affected, although other joints can also be involved. Studies based on radiographic findings have limitations, though, as the changes observed do not necessarily correspond to clinical disease, or the severity of clinical disease and pain. Nevertheless, where clinical disease is present many owners may simply assume a cat is ‘getting old’, and even educated and attentive owners may not necessarily appreciate suffering associated with DJD without veterinary observation and insight.

(a,b) Degenerative joint disease of the elbow in a feline patient
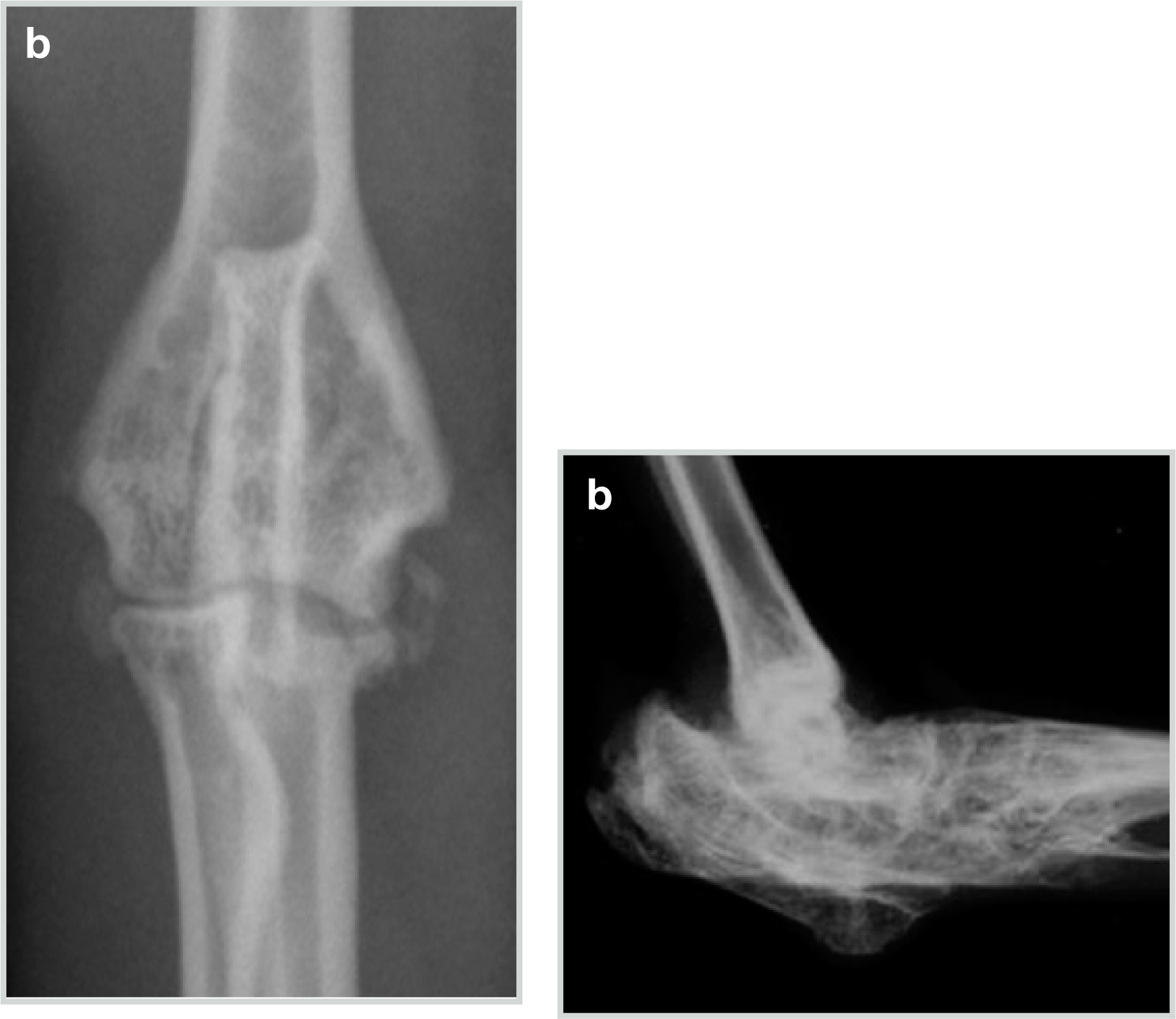
Anteroposterior (a) and lateral (b) radiographs of the hock of a Scottish fold cat with severe osteochondrodysplasia, showing destruction of joint spaces and extensive plantar exostosis. Courtesy of Kim Kendall
In the absence of medical intervention, many cats with DJD suffer pain and discomfort for years, greatly affecting their quality of life and the human/feline bond. It is vital that examinations of the older feline patient should specifically address whether DJD is present, through history and physical examination and, where necessary, radiology and therapeutic trials. Control of bodyweight, exercise and environmental modifications may help cats with DJD, as may other medical therapies. However, the dramatic responses reported to NSAIDs 13,21,23 indicate that there is a huge scope for safe, effective long-term NSAID therapy in the large cohort of aged cats with DJD (Fig 3).

Watson, a DJD sufferer, enjoying the benefits of daily NSAID treatment
Other diseases
There are many other feline diseases where control of protracted inflammation and pain is important. These include various cancers (Fig 4a), particularly where definitive treatment is not possible, or in some cases for the anti-neoplastic effect NSAIDs may offer. 25–28 Other common conditions associated with chronic pain where NSAIDs may form part of therapy include trauma, lymphoplasmacytic gingivostomatitis (Fig 5), 29 idiopathic cystitis, 30,31 skin disease and uveitis (Fig 6). In the last, both topical and/or systemic NSAID therapy may be valuable. 32 Through their antipyretic effect, control of fever with NSAIDs may also be valuable in some situations. A short therapeutic trial of an NSAID without a definitive diagnosis may sometimes be appropriate, using the response to treatment as a guide to diagnosis and further therapy. Informed client consent and close monitoring of the patient is mandatory, especially in such cases.

Transitional carcinoma of the bladder (a) and multifocal osteomyelitic bone lesions (b) in two feline patients. The tumour in the first cat was debulked surgically and the cat then received piroxicam; the second cat was given meloxicam in addition to antibiotics. Courtesy of Randolph Baral (a) and Emma Hughes (b)

(a-c) Severe and painful ulcerative and proliferative gingivostomatitis in three cats. (c) Courtesy of Alberto Barneto

Uveitis in a cat with toxoplasmosis. Courtesy of Carolyn O'Brien
NSAIDs and cyclo-oxygenase/lipoxygenase inhibition
The therapeutic benefits of NSAIDs include their antipyretic, analgesic and anti-inflammatory actions. They exert these effects mostly through inhibiting the production of prostaglandins (PGs) and leukotrienes (LTs) by the cyclo-oxygenase (COX) and 5-lipoxygenase (5-LOX) enzymes, respectively. 33–35 Most NSAIDs primarily inhibit the activity of COX enzymes. Although some also inhibit LOX enzymes, for currently licensed feline drugs this is generally short-lived in comparison with COX inhibition, and evidence of additional clinical efficacy from this is lacking. More effective dual COX/LOX inhibitors may become available in the future. 36–38
Two distinct COX isoforms (COX-1 and COX-2) have been identified as being responsible for the production of prostaglandins (Fig 7). 35 A third isoform has also been identified, initially known as COX-3, now described as a splice-variant of COX-1, which seems to have a role in the central control of pain. 38 Phospholipase A2 is the rate-limiting enzyme that initiates the COX pathway by liberating arachidonic acid (AA) from membrane-bound phospholipids. Both COX isoforms are then responsible for converting AA to PGG2 and PGH2 via identical enzymatic reactions. Following these initial steps, PGH2 functions as an intermediate substrate for the biosynthesis, by specific synthases and isomerases, of prostaglandins, prostacyclin and throm-boxanes. COX-1 converts AA to a range of molecules, including thromboxanes (TX), such as thromboxane A2 (TXA2), and prostaglandins, such as PGD2, PGE2 and PGF2, and prostacyclin (PGI2). COX-2 activity produces a narrower spectrum of prostaglandins, specifically PGE2, and prostacyclin.

Overview of the role of COX and LOX in prostanoid production
The prostaglandins play a major role in many aspects of normal physiology, including vascular homeostasis, gastroprotection, renal development and blood flow, blood clotting, reproduction, bone metabolism, wound healing, nerve development and growth, and immune responses. They are also involved in pathophysiological processes, including pain and inflammation, and cancer progression. However, much of our knowledge is extrapolated from other species, as there is a paucity of feline-specific data.
Expression of COX enzymes
Both COX-1 and COX-2 are enzymes that are constitutively expressed (normally present in tissues and at fairly constant concentrations), as well as induced (appear and/or increase in concentration in response to an inciting factor, often associated with inflammation). COX-1 is considered as predominantly constitutive, being expressed in almost all tissues, and involved in the production of prostaglandins responsible for ‘house-keeping’ functions, such as the cytoprotective effects in the gastric mucosa, normal platelet function and maintenance of renal perfusion. 39 Constitutive expression of COX-2 appears to be more restricted, 39,40 although it is present, along with COX-1, in the central nervous system, kidney, vascular endothelium, reproductive tract and gastrointestinal (GI) tract — sites where COX-2 activity contributes to homeostatic functions. 35,41 It appears that COX-2 has an important role in healing damaged mucosa in the GI tract, and although COX-2 has been shown to be constitutively expressed in the canine GI tract, 42,43 information on cats is lacking.
While COX-1 is the predominant constitutively produced enzyme, COX-2 is predominantly inducible and its production is dramatically upregulated during inflammation, in which it plays a central role. 44 The expression of COX-2 may also be upregulated in certain neoplasms, and in cats variable expression has been reported in transitional cell carcinomas, squamous cell carcinomas, mammary carcinomas and pancreatic carcinomas. 25–28,45–47 However, just as COX-2 has some constitutive expression, COX-1 expression also has a role to play in the inflammatory response. 39,40
COX and LOX selectivity, and NSAID adverse effects
Inhibition of COX-1, the enzyme predominantly associated with homeostatic functions, is reported to be the cause of most NSAID-induced side effects such as gastric ulcers and blood dyscrasias. In an attempt to avoid this, NSAIDs with a greater propensity to suppress COX-2 than COX-1, so-called ‘COX-2 preferential’ (or ‘COX-1 sparing’) NSAIDs, have been developed. Drugs that have negligible effect on COX-1 have been termed ‘COX-2 selective’ rather than ‘preferential’, although there is no recognised precise definition of these terms. 48
However, it rapidly became evident from human studies that COX-2 preferential or selective NSAIDs, while reducing some of the side effects classically associated with COX-1 inhibition, still caused adverse events such as acute renal failure, thromboembolic disease and gastric ulceration, 49,50 consistent with a physiological role for COX-2 in a number of tissues. For example, both COX-1 and COX-2 are expressed in mammalian kidneys. They are found within different cells of the kidney (macula densa, cortical ascending tubule, medullary interstitial cells), and play different roles, but both are important to preserve renal function during hypovolaemia. 50 Additionally, the inhibition of COX has been postulated to be associated with an increase in LOX activity, which can result in adverse effects on the GI mucosa. Furthermore, it has been suggested that dual inhibitors may be associated with fewer GI adverse effects than COX-1 or COX-2 inhibitors.
Although the COX/LOX selectivity of an NSAID may be important, this does not negate all potential side effects, and indeed evaluation of COX/LOX selectivity is not the only factor to consider when trying to predict the safety of an NSAID.
There are several other issues to consider. Firstly, the risks of adverse events can be affected by tissue concentrations of the drug — where the extracellular fluid is of a lower pH than the intracellular fluid, ‘ion trapping’ of weakly acidic drugs, such as most NSAIDs, can occur with accumulation of the drug within cells (eg, the gastric mucosa). 51 The extent to which this occurs will vary between drugs but this local accumulation can affect the prevalence of side effects.
Secondly, differences are recognised between species in both the expression and distribution of the COX enzymes. 52–55 Very little feline-specific data are available, but there could be differences in susceptibility to adverse events as a result of such differences in cats.
Thirdly, there are substantial variations in the reported COX selectivity of an NSAID based on the type of in vitro assay used to measure COX-1 and COX-2 activity. These results vary depending on the species used to source the material for the assay; and, even when the assay is performed in tissue from the target species, different assays yield different results. 36,38,56 Additionally, differences in metabolism of drugs between species can result in differing selectivity. In the dog, tepoxalin is a dual inhibitor for a short period of time only; but, in the cat, tepoxalin pharmacokinetics indicate it is potentially a balanced COX and LOX inhibitor throughout its kinetic profile. 38
Other factors also affect the risk of adverse events — for example, age. Older humans are recognised to be at greatest risk of GI ulceration; and in human medicine pre-existing renal insufficiency, cardiovascular disease and hepatic disease are all relative contraindications for use of NSAIDs. However, management of pain in the geriatric patient becomes critical to quality of life. Therefore, careful selection of NSAIDs and their dose, and the use of adjunctive therapies (such as proton pump inhibitors to assist gastroprotection, other analgesics to modulate other parts of the pain pathway and reduce the required NSAID dose, and fluid therapy to minimise effects of hypovolaemia), must be considered rather than simply avoiding addressing pain in both humans 57–60 and veterinary species. 61 Patient selection, dose titration and ongoing monitoring for the early signs of toxicity are essential. 62,63
Panel recommendations
COX-2 selectivity
What does this mean for cats?
Because of species differences in expression of COX enzymes and in the in vitro COX selectivity assays, it is imprudent to generalise results from any single study. 64 With all these variables, it is not surprising that there is no simple answer to the question of whether a COX selective or a dual COX/LOX inhibitor is better, and indeed what the ‘ideal’ COX/LOX selectivity and profile of an NSAID is in the cat. It may indeed depend on the disease process and the individual being treated. Despite these caveats, and given the paucity of feline-specific data at present, we can only cautiously extrapolate knowledge based on data from other species.
Practical NSAID therapy in cats
Beyond the question of COX selectivity, many other factors are also important in choosing and using NSAIDs for long-term therapy in cats.
Compliance
Administering medication to cats can be challenging for owners, yet adequate therapy relies on good owner compliance. Along with NSAIDs, many cats will be receiving other medications and the ‘administration burden’ may be daunting for owners, leading to inconsistent dosing. To help long-term use, a drug should ideally be highly palatable and taken voluntarily by the cat — for example, in food or as a treat — and veterinary pharmaceutical companies undertake much research into this. 65 Published studies suggest meloxicam liquid is highly palatable in cats, 13,16 with one study suggesting it was significantly more palatable than ketoprofen tablets. 16 Other drugs may be compounded in specific flavours that are appealing to individual cats. However, it is important to follow all regulations and compliance policies for drug com-pounding, 66 which are different throughout the world, and to consider the potential effect of compounding on bioavailability and stability/shelf-life.
Additionally, owners must be consistent and remember to administer the drug. Based on the long duration of action of many NSAIDs in cats, this should be at a set time on treatment days. Creative reminder systems may help ensure cats receive medication on the correct day(s), at the correct time(s) and at the correct dose. Giving medication along with a daily food ration (which should also be done for safety) can provide a built-in reminder system for owners, and encourage owner involvement in the monitoring process.
Dosing — intervals, frequency, timing and the ‘lowest effective dose’
Short-term pharmacokinetic data are available for a number of NSAIDs in cats, which form a basis for dosing intervals. While many NSAIDs are metabolised via glucuronidation in the liver, and the relative deficiency of glucuronyl transferase enzymes in cats may lead to a prolonged half-life for some of these drugs, 37,56 others, such as piroxicam and meloxicam, 56,67 are metabolised by oxidation. Single doses of many approved/licensed NSAIDs for acute pain in cats seem to have a duration of action of around 18–20 h. 56 However, it is not known if such prolonged pharmacokinetics are necessary for appropriate efficacy. For example, meloxicam and robenacoxib have a serum half-life of approximately 24 and 2 h, respectively, 68,69 yet both have been shown to be effective for daily treatment of musculoskeletal pain in cats by virtue of their European licences.
For most of the NSAIDs used in cats, it is not known if repeated long-term dosing alters the pharmacokinetics or pharmacodynamics of the drug. In one study, the administration of flunixin daily for 7 days appeared to result in more rapid metabolism of the drug after 7 days and decreased pharmacodynamic effects, 70 although the same did not appear to be evident during daily administration of meloxicam for 7 days. 68 Additionally, information on the apparent efficacy of daily versus every-other-day or less frequent dosing is anecdotal, with no controlled studies yet published. Daily dosing of meloxicam at less than the labelled dose for a mean of 5.8 months was considered to be clinically effective and associated with minimal adverse effects in one non-blinded study, 13 although efficacy was not measured objectively or with a validated assessment system. However, due to the inter-cat variability of pharmacokinetics with administration of a variety of NSAIDs, it is likely that daily dosing may be appropriate for some cats, while longer intervals might be appropriate for others.
Unfortunately there is no practical way to determine which cats might be ‘fast’ metabolisers and which slower. Additionally, probably as a result of their high protein-binding, which may enable NSAIDs to persist in inflamed tissue sites for longer than in plasma, the anti-inflammatory and analgesic activity of these drugs often persists longer than would be predicted from their serum half-life. This may enable daily dosing even for drugs with a relatively short half-life. 38,69 Indeed persistence at the site of inflammation has been demonstrated in an experimental study of robenacoxib in cats. 67 It is, therefore, unlikely that a set mg/kg dose and dosing schedule will work equally well for all cats; furthermore, variations in the level of pain may alter the cat's needs over time.
Very little attention has been given to the best time of day to administer NSAIDs to cats to achieve the most beneficial effect, a concept termed chronotherapy. 71 Theoretically, long-term dosing may result in a pharmacokinetic and pharmacodynamic steady-state. However, ‘peaks and troughs’ may still occur. If the peak beneficial effect on lameness occurs at say 5 h after dosing, treatment may be tailored to achieve maximum clinical effect when the cat is most active. The timing, therefore, may depend on a cat's lifestyle. Alternatively, an owner may find that at ‘peak effect’ the cat is more comfortable, it rests for longer and may choose to administer the drug to promote resting and sleeping at the most suitable time for the household.
Panel recommendations
Dosing frequency
Dosing — accuracy
Dosing accuracy will depend on the formulation of the drug. Liquids are more easily measured, and can be delivered in small volumes. Thus incremental increases or decreases in dose are potentially more readily achieved. However, differing dispensing methods can potentially result in wide variations in doses. Tablets or caplets are not always easy to divide and therefore delivering a small dose may be problematic and inaccurate. Intact tablets will provide a different dose to cats of different weights, which may not be a problem when the drug is licensed for a dose range, as for example robenacoxib, but may be a problem if a very precise target dose is required. Repeat subcutaneous injections may be another option in some cats and with some owners, although no NSAIDs are currently licensed for long-term use by this route.
Panel recommendations
Dosing accuracy X

Use of a dedicated dosing syringe is advisable
Dosing — switching drugs
There is little objective data available on the best way to transition therapy from one NSAID to another, and feline-specific information is lacking. There is concern about changing from aspirin to another NSAID in other species due to COX-2 dependent adaptive mechanisms that may occur during therapy. 38,61 However, there is uncertainty about the need for or timing of any ‘washout’ period with other NSAIDs. 38,61
Panel recommendations
Switching between NSAIDs
Monitoring efficacy
There is no validated assessment tool for acute or chronic feline pain, although studies are ongoing. 72 In studies evaluating the efficacy of NSAIDs in cats with musculoskeletal pain, improved mobility, and in particular the willingness to jump and the height of the jump, have been the most obvious signs of improvement, 13,21 and another study found increases in mobility with administration of an NSAID. 15 One key feature of chronic pain assessment is owner involvement and observation, especially as pain may manifest in different ways in individual cats. 56,73 It has been postulated that four behavioural domains — mobility, activity, grooming and temperament — are particularly useful to both clinicians and owners in assessing chronic musculoskeletal pain and monitoring the response to therapy. 23
When treating animals with long-term diseases, an overall assessment of ‘quality of life’ may be beneficial; this includes, but is not limited to, pain. An assessment tool may need to be individually designed since what is important to each patient will be different: can the cat climb trees, hunt, play with other pets in the household, and so on? 15 This was the thinking behind the use of client-specific outcome measures in a recent study. 15 Owners should keep a regular journal or diary of the cat's activities, as changes in mobility and behaviour may be subtle and occur slowly. The owner is the best person to judge and track the cat's behaviour and demeanour. It may only be obvious from consulting the ‘diary’ that a change in treatment is needed.
NSAIDs and concomitant disease
Renal disease
Prostaglandins play an important role in mammalian renal physiology, helping to autoregulate vascular tone, glomerular filtration rate (GFR), renin production and sodium/water balance. When renal haemodynamics are normal, prostaglandins appear to have a minimal role. In keeping with this, a recent study evaluating the effect of 5-day therapy with meloxicam in healthy adult cats showed no alteration in GFR based on iohexol clearance studies, 74 and similarly in healthy cats undergoing anaesthesia there is evidence of its safety when standard care is taken to avoid hypovolaemia and hypotension. 5 Under conditions of low effective renal blood flow, however, prostaglandins become crucial in maintaining renal function and GFR. Prostaglandin inhibition by NSAIDs may reduce renal blood flow and GFR and can result in the potential complication of acute kidney failure (AKF) in humans. 75
Both COX-1 and COX-2 enzymes appear to be important in maintaining renal function, but their relative importance and physiological role may differ between species; 56,74 for example, a recent immunohistochemistry study demonstrated greater COX-2 expression in the kidneys of dogs with chronic renal disease than in cats. 55 These observations suggest that the propensity for NSAIDs to cause AKF may vary between species. In humans, the risk of AKF is regarded as low, and can occur with both non-selective and COX-2 selective NSAIDs, although the risk may vary between individual agents. 50,75–79 In general, the risks for NSAID-induced AKF in humans are higher with conditions causing renal hypoperfusion (eg, dehydration, hypovolaemia, congestive heart failure), with old age (occult renal disease) and pre-existing renal disease, with concomitant drug therapy (eg, diuretics, angiotensin converting enzyme inhibitors [ACEIs]) and with higher doses of NSAIDs. The resultant AKF is usually reversible, provided it is detected in time. 50,63,76,77,79–81 The use of NSAIDs also carries a small risk of inducing hyperkalaemia in human patients, which is higher in those with existing renal disease and those on potassium supplements. 50,57,75
In human medicine, the role of NSAIDs in chronic kidney disease (CKD) is much less clear. While some studies have suggested that NSAIDs may be a risk factor for developing CKD (so-called ‘analgesic nephropathy’), 82–84 or in the progression of existing CKD, 85 others have found no evidence of a causal association, 86,87 and the difficulties in interpreting trial data have been highlighted. 88 Where studies have suggested a link between CKD and NSAID use, the risk appears to be low, and may be exacerbated by heavy use of one or more NSAIDs. 85,88,89
Two retrospective studies evaluated the safety of NSAIDs in a total of 76 older cats, including some cats with stable CKD. In both studies, cats received oral meloxicam (approximately 0.02 mg/kg/day) on a long-term basis for osteoarthritis. One study included three cats with International Renal Interest Society (IRIS) stage 3 CKD 90 and an additional 10 cats without CKD that had serum creatinine concentrations monitored, 13 and the other included 22 cats with IRIS stage 1–3 CKD. 14 Neither study showed any significant difference in the development or progression of renal dysfunction in treated cats, compared with age- and disease-matched controls, over an average period of 6 months 13 or more than 1 year. 14 Another study evaluated 73 cats that received oral piroxicam at an average dose of 0.2–0.3 mg/kg/day for between 1 and 38 months. In that study, no significant changes were seen in serum renal or hepatic parameters within the first month of therapy in 43 cats that had follow-up samples collected. 91 During prolonged therapy, five cases of renal insufficiency were detected in 58 cats receiving piroxicam, but as other therapies were being received, as the cats had underlying neoplasia, and as they were an older population without any controls, it was impossible to know if any of these were related directly to piroxicam therapy. 91 It has been demonstrated that cats with CKD have higher circulating levels of gastrin 92 and, as such, these cats may be at increased risk of GI adverse effects when NSAIDs are used.
Panel recommendations
Renal disease
Gastrointestinal disease
Because of the physiological role of COX in maintaining the normal gastric mucosal barrier, upper GI bleeding has been the most common serious complication associated with NSAID use in humans. Indeed, the GI tract has been considered the major site for NSAID toxicity in both humans and animals, including cats. 13,16,21,48,57,93 In one study of the long-term use of piroxicam in 73 cats with neoplasia, 91 vomiting was the most commonly reported adverse effect (occurring in 16% in the first month), although there was evidence that other therapies (eg, chemotherapeutic agents) contributed to the reported prevalence. During long-term use of oral meloxicam at a dose of 0.1 mg/cat, vomiting was reported in 2/46 cats (4%). 14 Direct topical injury to the GI mucosa may also occur and contribute to adverse GI effects. 38,59 Although studies in cats are lacking, in humans and other species, COX-1 has a major role in maintaining the mucosal integrity. However, COX-2 expression is also thought to be important, especially for repair of injured mucosa. 48,49,94,95
Factors that have been recognised as increasing the risks of GI adverse events in humans include higher doses of NSAIDs, the specific NSAID used, increased age, previous NSAID-associated GI disease, liver disease, pre-existing GI ulcers, and concurrent anti coagulant or glucocorticoid use. 57,59,63,77 Some of these risk factors have also been noted in dogs. 38 In humans, the two main strategies to prevent GI adverse events with NSAIDs are to use COX-1 sparing drugs, and/or a combination of an NSAID and a mucosal protectant such as a prostaglandin analogue (eg, misoprostol) or a proton-pump inhibitor (eg, omeprazole). 49,57,59,62,63,77 In cats, NSAID-associated gastric and intestinal ulceration and perforation is recognised and, in the current absence of species-specific studies, data from humans are considered relevant. 56
Panel recommendations.
Panel recommendations
Cardiovascular disease
Inhibition of COX activity by NSAIDs can have a number of potential adverse cardiovascular effects in humans. These are uncommon or rare, but include occasional exacerbation of congestive heart failure (CHF) and/or hypertension due to water and salt retention mediated by COX-1 and COX-2 suppression in the kidneys; reduced platelet aggregation and bleeding as a result of inhibition of COX-1 mediated platelet thromboxane production; and thromboembolic disease due to inhibition of COX-2 mediated endothelial prostacyclin production. 77,96–98 While ex vivo inhibition of platelet thromboxane has been demonstrated for a number of NSAIDs in cats, studies have not been able to demonstrate a clinically beneficial effect in preventing thromboembolic disease, or in promoting unwanted bleeding. 56,99 Currently, there are no data on the potential effects of NSAID therapy on blood pressure or CHF in cats, or on whether COX-2 selective agents may have a prothrombotic effect in certain individuals, such as those with a propensity to develop thromboembolism.
Liver disease
In humans, NSAID-induced hepatotoxicity is an uncommon or rare event. It is regarded as an idiosyncratic reaction mediated by hyper-sensitivity or a metabolic aberration, possibly as a result of genetic polymorphism, although with salicylates it has a predictable dose-dependent occurrence. 77,100,101 The risk of this may be higher in patients receiving other potentially hepatotoxic drugs and varies between different NSAIDs, with toxicity usually developing within the first 6–12 weeks of therapy. 100–102 Idiosyncratic hepatotoxicity has also been reported in dogs receiving NSAIDs. 103
Severe hepatotoxicity following clinical use of NSAIDs has not been reported in cats, although this may simply reflect the lower prevalence of NSAID prescribing in this species. 56 Although NSAIDs are metabolised by the liver, pre-existing liver disease does not appear to predispose to NSAID-induced hepatotoxicity. 38 As drug metabolising pathways are often well preserved in liver disease, withholding NSAID therapy in such patients may not necessarily be required without evidence of significant liver dysfunction, 38 although reducing doses in severe/advanced liver disease is recommended in humans. 104 In humans, pre-existing advanced liver disease may be a risk factor for NSAID-associated renal and GI adverse events. 63,81
NSAIDs and concomitant drug therapy
Glucocorticoids
Concomitant use of glucocorticoids and NSAIDs carries a well-characterised increased risk for adverse GI events in humans and dogs, 38,56 with an estimated 2- to 15-fold greater risk for peptic ulcer disease in humans. 59,63
Panel recommendations
Concurrent drug therapy
Panel recommendations
Liver disease
Angiotensin converting enzyme inhibitors and diuretics
The use of ACEIs (and/or angiotensin receptor antagonists) and/or diuretics along with an NSAID carries a well-recognised risk for development of acute NSAID-associated renal adverse events in humans, 63,75,81,105 and there is evidence of a higher risk when all three drugs are used together. 105 Both ACEIs and NSAIDs may individually result in altered renal haemodynamics and reduced GFR, thus together the risk may be compounded, and the use of diuretics may lead to volume depletion and a greater renal dependence on prostaglandins to maintain GFR. 105
Anticoagulants
Although COX-1 inhibiting NSAIDs may suppress platelet thromboxane production and reduce platelet aggregation, clinically significant bleeding as a result of this is rare in humans and, to date, has not been reported in cats. 56,61,63 However, NSAIDs may appreciably potentiate the effect of warfarin, and other highly protein-bound drugs, through competitive protein binding 63 and the use of these drugs together should be avoided.
Monitoring cats receiving long-term NSAID therapy
Adverse drug events (ADEs) related to NSAID use most commonly affect the GI system, liver, kidneys and platelet function, but lessons learned from the long-term use of these agents in dogs suggest that this class of drug is often used inappropriately and without screening and monitoring. 106
While the need for, and benefit of, NSAID therapy in many situations is clear, screening and monitoring is important for the clinician, owner and patient, to help minimise the likelihood of ADEs occurring. Until further data become available, especially from pharmacovigilance studies, suggested protocols for screening and monitoring cats on long-term NSAID therapy have to be based on a knowledge of the use of these drugs in both animals and humans, and it is important that protocols are adapted to the individual needs of the patient.
Testing and screening before treatment
Thorough patient evaluation before commencing therapy is crucial, with a view to identifying concurrent conditions or therapies that may impact on NSAID administration, and allowing informed client consent to be obtained.
Panel recommendations
Screening before therapy

Blood pressure measurement should ideally be performed as a screening measure before NSAID therapy in cats
Screening during treatment
In dogs, most NSAID-related ADEs occur between 14 and 30 days (range 3–90) after the start of treatment. 107 However, it is recognised that the time for an ADE to develop is extremely variable, probably being dependent on the individual drug, the dose and the individual patient. In humans, hepatotoxicity is usually reported within the first 6 months of therapy, with more than 60% of cases reported in the first 3 months, 102 whereas acute renal failure is usually reported early, often within the first few days or weeks of commencing drug administration. 78
Based on appropriate use of NSAIDs in other species, the prevalence of ADEs is low in healthy patients. However, the frequency of certain ADEs increases in some patient groups, and these can therefore be classified as having a ‘higher’ or ‘lower’ risk. This approach enables treatment and monitoring plans to be adjusted according to perceived risks. 57,59,60,62,63,81 Critically, ADEs are typically reversible with prompt recognition and intervention. Categorising patients as having a higher or lower risk of ADEs has clear benefits and should be equally applicable to cats, although at present this has to be based largely on knowledge of ADEs in other species, due to the lack of feline-specific data.
Panel recommendations
Classification of patients
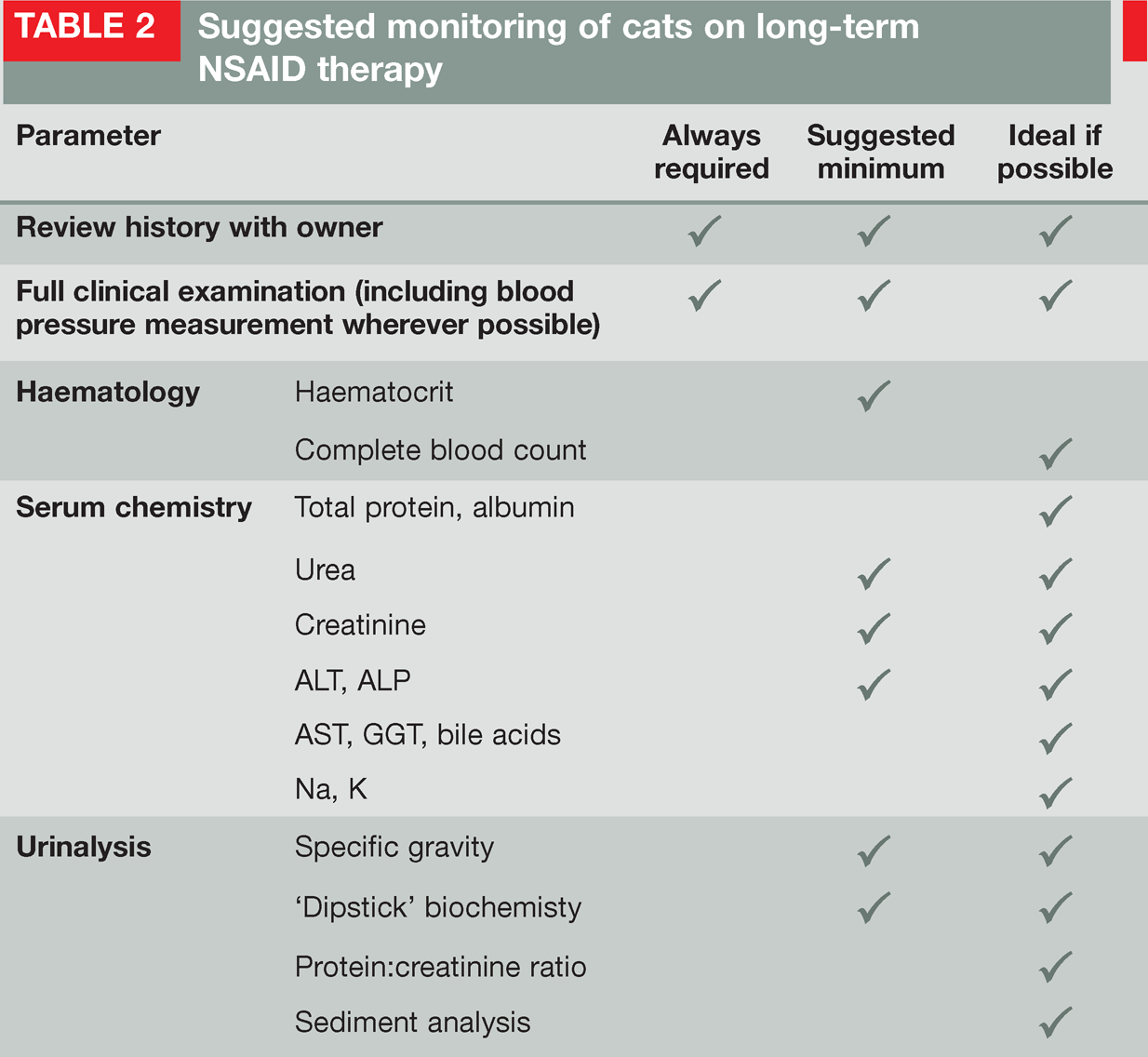
Suggested monitoring of cats on long-term NSAID therapy
Panel recommendations
Monitoring during therapy

Client leaflet advising on safe use of NSAIDs. The leaflet may be downloaded from www.isfm.net/toolbox and www.catvets.com/professionals/guidelines/publications
Adverse events and adverse event reporting (pharmacovigilance)
If ADEs are encountered or undesirable effects are seen, these should be managed as appropriate. If adverse GI events are observed, NSAID therapy should be withheld, and appropriate supportive therapy introduced, until any mucosal lesions have healed. If therapy is re-instituted, it should be done so at the lowest effective dose with consideration given to the concomitant use of omeprazole (0.7–1.0 mg/kg PO q24h) or misoprostol (5.0 μg/kg PO q8h), 99,108 and/or a different NSAID where licensing permits.
Hepatotoxicity or acute renal failure are usually reversible in other species on drug withdrawal and appropriate supportive therapy, providing they are detected early enough — this emphasises the importance of patient monitoring and of ensuring clients are involved in this process. In humans, it is recommended that a three-fold increase in ALT should lead to cessation of NSAID therapy. Milder increases may prompt more attentive monitoring, with further investigations being warranted if the ALT fails to return to baseline concentrations. 57 Re-institution of alternative NSAID therapy after hepatotoxicity or acute renal failure should be done very cautiously. Increases in blood pressure have been documented in other species with NSAID therapy, and this should be monitored in cats — antihypertensive therapy or more intense antihypertensive therapy should be used as appropriate.
All ADEs should be reported to the relevant pharmaceutical company and regulatory board to help the patient and enable accurate collation of information so that we can learn more about when and why they occur.
Acknowledgements
Boehringer Ingelheim Animal Health GmbH generously provided an educational grant to help facilitate the development of these guidelines.
SUMMARY POINTS
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
