Abstract
Osteoarthritis is the most common arthropathy of mammalian species including cats. Cartilage degradation is central to the disorder and here we present, for the first time, an in vitro model of feline cartilage degradation which will be useful for further studies in this target species. Feline articular cartilage explant cultures were maintained for 28 days and in the presence of oncostatin M with and without interleukin (IL)-17, tumour necrosis factor (TNF), IL-1α, or IL-1β. Media samples and digested cartilage explants were analysed for glycosaminoglycan (GAG) and collagen content. The combination of IL-1β and OSM, both at 20 ng/ml, was able to promote GAG release to the greatest extent at 14 days. At 28 days, all groups showed relatively high release of GAG. At 14 days, only IL-1β and OSM in combination were associated with a statistically significant increase in collagen release over and above control tissue. IL-1β dose-response studies showed that an IL-1β dose of 10 ng/ml promotes a statistically significant increase in GAG breakdown when used with OSM, and higher doses of IL-1β did not result in significantly greater response. The model demonstrated both GAG and collagen degradation and will be of use for further understanding of feline cartilage metabolism and for screening of potential structure-modifying agents to be used in cats.
Osteoarthritis (OA) is the most common arthropathy to affect mammalian species. A key component of OA is the gradual loss of articular cartilage within the synovial joint. There have been multiple studies which have explored the pathology and biochemistry, as well as clinical and radiological features of canine OA. By comparison, as a species, the cat has received little attention in this area. An experimentally-induced model of OA in the feline stifle joint has been used, to a limited extent, to study the pathological processes of OA. 1–7 However, in recent years, there have been several clinical and radiological studies that have highlighted the prevalence of OA in the domestic feline population. 8–10 In addition, there is an increasing interest in providing therapies for cats with OA.11
A central tissue in OA is articular cartilage. Although studies of feline cartilage and chondrocytes are very limited, 12–14 these studies, and the many others in several other mammals, indicate that cartilage is a complex avascular tissue adapted both to allow the almost frictionless movement of joints and to dissipate load. 15 The single cell type of articular cartilage, the chondrocyte, is the architect of an organic extracellular matrix (ECM) composed mainly of type II collagen and glycosaminoglycans (GAGs), of which aggrecan is the major constituent. The triple helical type II collagen molecule confers tensile strength on the tissue and the GAGs imbibe water such that approximately 70% of the wet weight of cartilage is water. The GAGs thus provide a swelling pressure that resists compressive load. 16 In OA, degradation of the organic matrix is upregulated through the activity of two families of metalloproteinases, matrix metalloproteinases (MMPs) and A disintegrin and metalloproteinase with thrombospondin motifs (ADAMTSs).
In vitro models of cartilage degradation have been used to study the anabolic and catabolic processes occurring in articular cartilage, taken from a variety of species, for over 20 years. 17–22 Furthermore, such models are often used to screen candidate therapeutic agents, 23,24 or to test the effects on cartilage of agents used for long-term symptomatic relief of OA pain. 25,26 One common model system is to use explants of articular cartilage and initiate cartilage degradation with one or more catabolic cytokines. Commonly used catabolic cytokines include interleukin-1 (IL-1), 27 tumour necrosis factor (TNF), 28 oncostatin M (OSM), 29 and more recently, IL-17. 30–32 However, the literature 33,34 and our own experience indicate that there can be significant inter-species differences in the response to various recombinant cytokines, and it is therefore important to develop models for each target species.
To our knowledge, degradation models of feline cartilage have not been reported. We hypothesised that feline cartilage would respond to various candidate recombinant catabolic cytokines (OSM, TNF, IL-1α or IL-1β, IL-17) as measured by increased release of ECM components into the culture media. Further to this, we aimed to characterise that response and thus produce a reliable model of feline articular cartilage degradation for further studies of candidate therapeutic agents which may be relevant for feline OA.
Materials and Methods
Feline cartilage explants culture system
Feline articular cartilage was obtained within 24 h of death from the tibial condyles of cats euthanatized at the owner's request for reasons unrelated to joint disease and with the owner's consent for tissue collection. Joints were assessed for gross pathology and only grossly healthy joints were used. Cartilage slices were washed three times in Hanks balanced salt solution (HBSS) (Invitrogen, Paisley, UK) containing 300 U/ml penicillin, 300 μg/ml streptomycin and 7.5 μg/ml amphotericin B. Cartilage slices were then sectioned into explants of approximately 2 mm2, mixed and allowed to stabilise for 24 h at 37°C in a Petri dish containing Dulbecco's modified Eagle medium:nutrient mix F-12 (DMEM/F-12) plus 100 U/ml penicillin, 100 μg/ml streptomycin, 2.5 μg/ml amphotericin B, 2 mM glutamax and 46 μg/ml l-proline. Explants were then transferred to individual wells of 24-well tissue culture plates (in quadruplicate). Each well contained 500 μl DMEM with the supplements previously described, with the addition of 50 μg/ml ascorbic acid (Sigma–Aldrich, Dorset, UK).
Cultures were maintained for 28 days, with media collected and replaced every 7 days. After 28 days, cartilage explants were digested with 1 mg/ml papain in 0.1 M sodium phosphate buffer, pH 6.5, containing 5 mM N-acetyl cysteine and 5 mM EDTA for 16 h at 65°C.
All cytokines were recombinant human cytokines obtained from PeproTech EC, London, UK. Explants were maintained with 20 ng/ml OSM with or without another cytokine (50 ng/ml IL-17, TNF or IL-1α or 20 ng/ml IL-1β). Cytokines were refreshed with every media change. Media samples from each collection point and digested cartilage explants were analysed for GAG content using the dimethylmethylene blue (DMMB) assay, 35 and for collagen content using the microtitre plate hydroxyproline method.36
Evaluation of OSM in combination with IL-1α, TNFα and different concentrations of IL-1β
A dose–response experiment using feline articular cartilage was established using the method described above, with 20 ng/ml OSM with or without 50 ng/ml TNF or IL-1α or 50, 20 or 10 ng/ml IL-1β. Media and cartilage were analysed for GAG and collagen as described previously, as well as determining MMP-13 activity using a quenched fluorescent peptide substrate.37
MMP-13 fluorogenic substrate assay
MMP-13 enzymatic activity was assessed in feline cartilage (n=2) explant culture medium using a fluorogenic substrate (MCA-Pro-Cha-Gly-Nva-His-Ala-Dpa-NH2) (10 μM; Calbiochem), which is preferentially cleaved by MMP-13. 37 The assays were performed at 37°C in black 96-well plates with assay buffer (50 mM HEPES, 200 mM NaCl, 1 mM CaCl2, 0.01% (v/v) Brij-35, pH 7.3). Serial dilutions of active MMP-13 containing media from a lipopolysaccharide (LPS)-stimulated canine monocyte cell line (DH82) were included on each plate to ensure a linear relationship between MMP-13 activity and substrate cleavage over the required range. DMEM was included in order to determine the assay background.
In order to obtain values for total (latent and active) MMP-13 levels in the explant medium, activation of the latent enzyme was performed by incubation with 0.5 mM p-aminophenylmercuric acetate (APMA) at 37°C for 1 h prior to addition of the substrate.
Total MMP-13 generated fluorescence were measured (FL ×800 microplate reader; Bio-Tek Instruments), adjusting the excitation wavelength to 325 nm and emission to 393 nm after incubation of samples with substrate for 3 h at 37°C. The data were analysed by KC junior software (Bio-Tek).
Statistical analysis
Loss of GAG and collagen was expressed as percentage of totals at 7 or 14 and 28 days. Kruskal–Wallis tests followed by Dunn's post-tests were used to investigate differences between cytokine-stimulated explants and control tissue.
Results
Feline cartilage explants culture system
Total GAG and collagen release (at 14 and 28 days) in response to culture with individual cytokines are illustrated in Fig 1. Several cytokines were able to increase release of GAG at 14 and 28 days (Fig 1a and c). The combination of IL-1β and OSM, both at 20 ng/ml, was able to promote GAG release to the greatest extent at 14 days. At 28 days, all groups showed relatively high release of GAG.
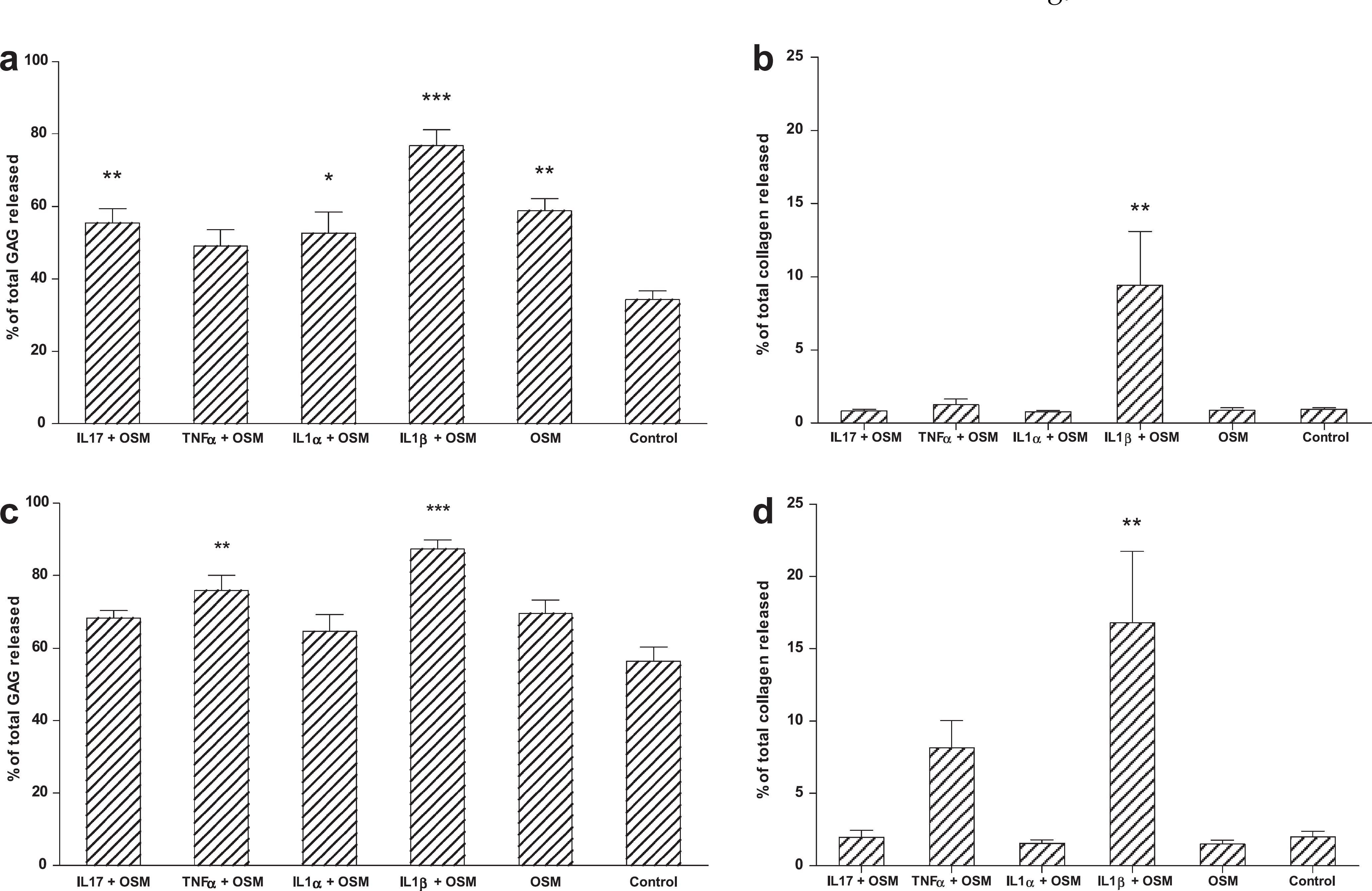
Degradation and release of proteoglycan and collagen from feline cartilage explants treated with cytokines. Explants were maintained with 20 ng/ml OSM with or without another cytokine (50 ng/ml IL-17, TNF or IL-1α or 20 ng/ml IL-1β). Accumulative levels of proteoglycan released into the medium up to days 14 and 28 of the culture period are shown in (a) and (c), respectively. (b) and (d) show the accumulative release of collagen degradation products up to 14 and 28 days, respectively. Mean±SEM. *P≤0.05, **P<0.01, ***P<0.001.
The collagen degradation data obtained over the same time points (Fig 1b and d) show that at 14 days, only IL-1β and OSM in combination were associated with a statistically significant increase in collagen release over and above control tissue. After 28 days, further collagen loss had occurred in response to stimulation with IL-1β and OSM. However, at this time point, TNF-treated cultures also showed loss of collagen, but this degradation was not significantly different from control.
In order to determine whether the IL-1β/OSM-stimulated loss showed a dose dependency, a range of IL-1β concentrations were tested in combinations with OSM, alongside combinations of TNF and IL-1α with OSM, OSM alone, and control untreated cultures (Fig 2) over 14 (Fig 2a and b) and 28 days (Fig 2c and d). As expected, there was evidence of GAG degradation within 14 days (Fig 2a). Again, only IL-1β and OSM in combination were associated with a statistically significant increase in collagen release over and above control tissue at 14 days (Fig 2b). This was seen at an increased level after 28 days of treatment with all IL-1β concentrations, but no other treatments (Fig 2d). The data obtained show that at both time points, an IL-1β dose of 10 ng/ml promotes a statistically significant increase in GAG breakdown when used with OSM, and higher doses of IL-1β did not result in significantly greater response. In terms of collagen loss, neither TNF nor IL-1α showed any synergy with OSM in driving this degradation.

Degradation and release of proteoglycan and collagen from feline cartilage explants treated with cytokines, including a range of IL-1β concentrations (10 ng/ml, 20 ng/ml or 50 ng/ml). Medium was collected up to 14 days (a and b) and up to 28 days (c and d) in order to measure accumulative loss of proteoglycan (a and c) and collagen (b and d). Mean±SEM. *P≤0.05, **P<0.01, ***P<0.001.
Total MMP-13 activity data obtained using a fluorogenic substrate assay (Fig 3) supported this finding with IL-1β concentrations of 10, 20 and 50 ng/ml all synergising with OSM to upregulate levels of activity relative to the untreated controls.

Stimulation of MMP-13 activity released from cartilage explants treated with OSM and a range of concentrations of IL-1β. Medium collected from cytokine-stimulated feline explant cultures was analysed using the fluorogenic substrate assay. Levels of MMP-13 measured in medium collected at 7, 14, 21 and 28 days are shown in (a) for OSM in combination with 10 ng/ml IL-1β, 20 ng/ml IL-1β and 50 ng/ml IL-1β. Control unstimulated cultures are shown alongside. Average MMP-13 activity produced over the 28-day culture period is shown in (b). Mean±SEM. *P≤0.05, **P<0.01, ***P<0.001, with comparisons made to the control for each time point in (a).
At 14 days, all concentrations of IL-1β upregulated levels of MMP-13 to equivalent levels when used in combination with OSM (Fig 3a). However, after 14 days MMP-13 levels released from explants treated with 10 ng/ml IL-1β showed a sustained response, whereas higher concentrations of IL-1β resulted in diminished production of the enzyme. Overall, this resulted in higher levels of MMP-13 being produced in response to the lowest concentration of IL-1β used in combination with OSM (Fig 3b).
Discussion
The development of an in vitro model of feline cartilage degeneration is advantageous for the study of potential structure-modifying agents in feline OA. Samples in this study were from a small number of cats without evidence of OA, and we have not been able to address background variables such as gender and age and how they might affect the results in the model. In addition, we have only studied healthy cartilage but this is likely to result in a more consistent model than using osteoarthritic cartilage.
We have identified that OSM and IL-1β in combination produce predictable release of GAG and collagen. It is of interest that neither canine nor equine cartilage undergo collagen degradation in response to this cytokine combination (A Vaughan-Thomas and J Innes, unpublished observations); however, previous work has shown that it is effective in degrading cartilage of other species including bovine and human, 38 and in our study it has been shown that feline cartilage is also susceptible. This may indicate that species vary in their response to recombinant human cytokines due to slight differences in the primary structure of the chondrocyte cell-surface receptors. Previous studies in other species have noted the synergy between IL-1 and OSM in inducing collagen and GAG degradation in cartilage.39
The GAG release observed in our model occurred in a time frame which is similar to models in other species. We observed very significant release of GAG at 7 and 14 days but by 28 days the continued low level turnover in the control explants resulted in reduced differences between groups as the cumulative release of GAG reached almost 80% and was able to catch up somewhat with the cytokine-stimulated groups. Thus, as in other species, the window for studying GAG release in this model is 0–14 days. The GAG released is likely to be primarily associated with degradation of aggrecan by ADAMTS-4 or ADAMTS-5 although studies to confirm this in the cat are lacking at present. Certainly, these enzymes are known to be key activities in the cytokine-stimulated release of aggrecan in models in other species 17,40,41 and ‘aggrecanase’ activity is known to be upregulated in clinical cases of canine OA. 42 Further studies will be required to investigate the activity of ‘aggrecanases’ in cats.
Collagen release is typically a slower event and typically occurs subsequent to GAG release in cartilage explant models such as this in other species. Surprisingly, in our experiments, the release of collagen from feline cartilage was measurable within the first week of exposing the tissue to the cytokine combination. This is a rapid response, relative to other species, 33,38,43 and may indicate that feline cartilage may be more ‘primed’ to elicit a degradative response on the collagen network. Degradation of the collagen network appears to be an irreversible step in the progression of OA and any agent that is able to reverse, stop or retard this process is likely to be therapeutically beneficial. However, this rapid loss of collagen also makes the feline system a useful model in shortening the timescale of the collagen degradation process which increases the efficiency of the model and reduces research costs. Thus, the model may be useful for broader discovery programmes aimed at blocking collagen degradation.
In this model, we have demonstrated increased MMP-13 activity in response to catabolic cytokines. MMP-13 is regarded as having a key enzymatic activity in the cleavage of triple helical type II collagen in cartilage in other species. 44–46 Our results suggest that MMP-13 may also have a key activity in that regard in feline cartilage. Work in human cartilage has indicated that a combination of IL-1 and OSM is a potent inducer of MMP-13 activity, perhaps through pro-MMP-13 activation by MMP-14.39
In summary, we have described an in vitro model of feline cartilage degradation that could prove a useful tool in drug discovery and development. With the expanding feline populations in the developed world, the longer life expectancy of cats, and the increased awareness of feline OA, such a model is likely to be critical to develop improved therapeutic options in this species.
Footnotes
Acknowledgements
This manuscript is dedicated to the memory of Dr Anne Vaughan-Thomas, a dear colleague and friend. The work, including Natalie Gabriel's salary, was supported by Hill's Pet Nutrition, Topeka, Kansas, USA. The authors are grateful to Dr Bill Schoenherr, Hill's Pet Nutrition, for advice and support throughout these studies.
