Abstract
Clinical Reports
Case 1
A 10-year-old female spayed Persian cat was presented at the Queen Mother Hospital for Animals (QMHA) at the Royal Veterinary College with a 7-day history of malaise and inappétence. She had been treated empirically, a week previously, for suspected cystitis with a single injection of meloxicam (0.3 mg/kg SC) and oral amoxicillin clavulanate (15 mg/kg bid PO) and N-acetyl D glucosamine for 6 days (125 mg bid PO). During this period inappetence worsened and lethargy and occasional vomiting were noted. On the sixth day of treatment, clinical examination revealed abdominal dilation and discomfort and radiographs demonstrated pneumoperitoneum. This finding prompted referral to the QMHA.
On presentation, the cat was quiet but responsive. She was tachycardie (220 beats per minute) with weak peripheral pulses, and was moderately dehydrated (7–8%). Abdominal palpation revealed generalised abdominal distension and tympany. Haematological evaluation demonstrated a normal neutrophil count but a moderate left shift and toxic changes, and lymphopenia (0.22 × 109/l; reference range [RR] 1.5–7 × 109/l). Serum biochemical analysis showed hypoalbuminaemia (23.4 g/l; RR 28–42 g/l), hypoproteinaemia (48.1 g/l; RR 61–80 g/l), elevated creatine kinase activity (5876 U/l; RR 52–506 U/l) and hyponatraemia (150 mmol/l; RR 153–162 mmol/l). Orthogonal abdominal radiographs showed a marked pneumoperitoneum and mild gaseous distension throughout the gastrointestinal tract (Fig 1). In the absence of a compatible history or external signs of trauma, spontaneous pneumoperitoneum was thought most likely to be associated with a gastrointestinal tract rupture.
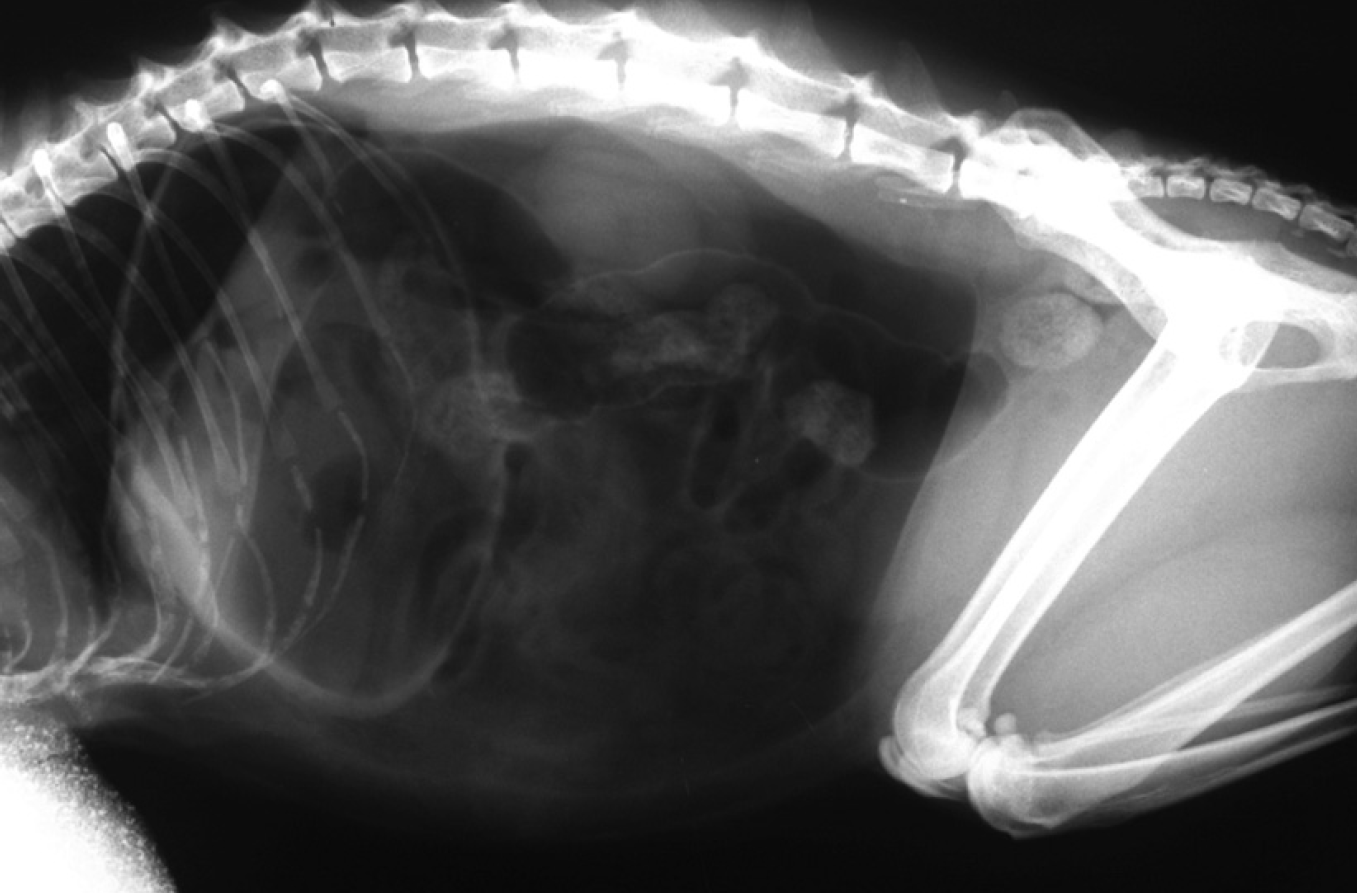
Left lateral radiographic projection of case 1. Marked pneumoperitoneum and mild gaseous dilation of the gastrointestinal tract are visible
After an initial period of stabilisation with fluid resuscitation, the cat underwent an exploratory coeliotomy. A large volume of free peritoneal gas was evident upon incision through the linea alba. Thorough inspection of the gastrointestinal tract revealed a small gastric perforation associated with the lesser curvature of the stomach, in the area of the pyloric antrum (Fig 2). The lesser omentum and the left middle liver lobe were adhered to the stomach at this site. A gastrotomy was performed to allow evaluation of the lesion.

Appearance of the ulcer in case 1, as viewed from the visceral gastric layer before incising through the gastric wall
Grossly the lesion appeared to be a pinpoint perforation at the centre of a thickened ulcerated area 1 cm in diameter. This area was resected by a full thickness incision with 1 cm margins (Fig 3) and submitted for histopathology. The gastric defect and gastrotomy were closed in two layers. There was a minimal volume of free peritoneal fluid. Intraoperative cytological assessment of the fluid demonstrated a mixed neutrophilic/pyogranulomatous inflammatory response; no bacterial agents were identified. The abdomen was lavaged thoroughly and closed routinely and an oesophagostomy tube was placed to facilitate feeding postoperatively.

Intraoperative views of the small perforation within the gastric ulcer of case 1. The lesion is being resected with 1 cm margins of normal-looking gastric wall. Stay sutures help with manipulation of the stomach and tissue to be resected
Microbial culture of the free peritoneal fluid did not yield any bacterial growth. Histological evaluation of the resected tissue showed areas of necrosis, acute inflammation and new and maturing granulation tissue extending through the full thickness of the stomach wall to an area of inflammatory/ healing reaction in the serosa and abdominal fat. This was consistent with a perforating gastric ulcer. There was no evidence of neoplasia in the evaluated sections.
The cat remained hospitalised for 6 days after surgery. She was treated with empirical antibiotics (cefuroxime 20 mg/kg tid IV and metronidazole 10 mg/kg tid IV), both started perioperatively, omeprazole (1 mg/kg sid PO) and sucralfate (250 mg bid PO). Oesophagostomy tube feeding was employed as she remained inappetent in the hospital. Once discharged, she started eating well and the feeding tube was removed at the time of skin staples removal, 12 days after surgery. She was reported to be clinically normal at that time.
Nineteen months post-surgery, there had been no further occurrence of digestive or urinary tract signs.
Case 2
A 3-year-old male neutered Burmese cat was presented to the QMHA for evaluation of abdominal distension and discomfort. The cat had a 1-week history of lethargy and inappetence. Potentiated amoxicillin and meloxicam had been prescribed. He had been treated 9 months previously for multiple pelvic fractures following a road traffic accident. A right iliac fracture had been managed surgically using a bone plate and screws, while left iliac, pubic and ischial fractures had been managed conservatively. The cat had reportedly slowly but steadily recovered. He had, however, continued to be mildly lame on his right pelvic limb and had intermittently been given meloxicam for dogs (one drop a day with food; ie, approximately 0.1 mg sid PO) until presentation.
On presentation, the cat was quiet but responsive, with temperature, pulse and respiratory rates within normal limits. His abdomen was moderately distended and abdominal pain was elicited on palpation. Haematological analysis demonstrated moderate neutrophilia (25.7 × 109/l; RR 2.5–12.5 × 109/l) with 13% bands and toxic changes in the neutrophils. Serum biochemical analysis revealed hypoalbuminaemia (21 g/l; RR 28–12 g/l), hypoproteinaemia (55.9 g/l; RR 61–80 g/l), azotaemia (urea 16.3 mmol/l; RR 6.1–12 mmol/l), hyperbilirubinaemia (14.4 μmol/l; RR 0–3 μmol/l), hyponatraemia (142.9 mmol/l; RR 153–162 mmol/l), hypochloraemia (103.6 mmol/l; RR 110–121 mmol/l) and hyper-kalaemia (5.8 mmol/l; RR 3.8–5.3 mmol/l). Results of feline leukaemia virus (FeLV) and feline immunodeficiency virus (FIV) serological tests were negative. Orthogonal abdominal radiographs showed marked pneumoperitoneum in association with gaseous distension of the small intestinal tract. Ultrasound evaluation confirmed pneumoperitoneum and revealed free peritoneal fluid. Abdominocentesis yielded serosanguineous fluid, cytology of which was consistent with a septic neutrophilic inflammation with the presence of intracellular bacteria. However, microbial culture yielded no bacterial growth.
The presenting signs, physical findings and laboratory results for the three cats were essentially non-specific … There were no signs referable to gastric bleeding, decompensated shock or pain on abdominal palpation.
A diagnosis of pneumoperitoneum and generalised ileus secondary to a presumptive perforated viscus was made, and an emergency exploratory coeliotomy was performed. Adhesions were found between the parietal surface of the stomach and the head of the spleen, the left medial and quadrate lobes of the liver and the greater omentum. After careful division of these adhesions, two perforations were found on the parietal surface of the gastric body. These lesions were debrided and resected with 1 cm margins of stomach wall and the defect was closed in two layers using 2 metric polydioxanone (PDS II; Ethicon) in a simple continuous pattern. The gastric repair was further omentalised. The peritoneal cavity was lavaged copiously with warm sterile saline and the abdominal wall closed routinely.
The cat recovered well from the procedure and was bright and eating the day after surgery. He was initially kept on maintenance intravenous fluid therapy, cefuroxime (20 mg/kg tid IV), metronidazole (10 mg/kg bid IV) and enrofloxacin (5 mg/kg sid IV), which had been started perioperatively and empirically, sucralfate (250 mg tid PO) and ranitidine (2 mg/kg bid PO). However, 3 days after surgery, the cat had a seizure, which was treated successfully with a single bolus of diazepam. This seizure resulted in post-ictal blindness. Treatment with metronidazole and enrofloxacin was discontinued due to the possible neurological and ophthalmic side effects of these drugs. Histopathological examination of the two debrided lesions showed mixed inflammation, along with congestion, hyperaemia, haemorrhage and occasional areas of necrosis within the gastric wall. There was no evidence of neoplasia. These findings were consistent with perforated gastric ulcers.
Six days after surgery, the cat was discharged with a 5-day course of sucralfate (250 mg tid PO) and ranitidine (2 mg/kg bid PO). At the time of suture removal, 12 days after surgery, the owners reported that he was clinically normal and his sight was improving. Thirty-two months after surgery, the cat was clinically normal except for a persistent, but occasional, mild lameness in his right pelvic limb. Meloxicam administration had been discontinued and this did not appear to have negatively affected the cat's level of comfort and exercise.
Case 3
A 14-year-old male neutered domestic short-hair cat was presented to the QMHA with a 1-day history of acute inappetence, lethargy and vomiting. The cat had undergone routine dental work under general anaesthesia 7 days prior to presentation and had been on a 5-day course of clindamycin. He had also been administered prednisolone (5 mg sid PO) for 3 months to palliate discomfort related to bilateral coxofemoral osteoarthritis. Abdominal radiographs performed at the local veterinary practice to investigate abdominal distension revealed a marked pneumoperitoneum, prompting referral.
The cat presented depressed but responsive. His vital parameters were within normal limits, with the exception of an increased respiratory rate (60 bpm) and effort. Moderate dehydration (7%) was noted. Abdominal palpation confirmed marked distension but no overt discomfort could be elicited.
Haematological analysis demonstrated a marked anaemia (PCV 16%) with signs of regeneration, as well as a moderate neutrophilia (16.3 × 109/l; RR 2.5–12.5 × 109/l) with a left shift and a lymphopenia (0.18 × 109/l; RR 1.5–7 × 109/l). Biochemical analysis showed hypoalbuminaemia (24.2 g/l; RR 28–42 g/l) and hypoproteinaemia (52.8 g/l; RR 60–80 g/l), increased creatine kinase activity (2200 U/l; RR 52–506 U/l), hyponatraemia (150 mmol/l; RR 153–162 mmol/l) and hypokalaemia (3.5 mmol/l; RR 3.8–5.3 mmol/l). Results of FeLV and FIV serological tests were negative.
After a short period of fluid resuscitation, the cat underwent exploratory coeliotomy. On incision of the linea alba, a small amount of serosanguineous peritoneal fluid was noted. A full-thickness perforation of the gastric fundus was identified adjacent to the greater curvature of the stomach. The lesion was resected with 1 cm margins and the defect closed using a 50 mm GIA stapler. The gastric site was further oversewn with 1.5 metric polydioxanone in a simple continuous pattern. The peritoneal cavity was lavaged copiously with warm sterile saline and the abdominal wall was closed routinely after placement of a Jackson-Pratt drain connected to an active suction system. The procedure was completed with the placement of an oesophageal feeding tube.
Microbial culture of the peritoneal fluid yielded a scanty growth of Pasteurella species, sensitive to most antibiotics tested, including cephalexin. Histopathological examination of the resected tissue was consistent with a gastric perforated ulcer and associated subacute gastritis and local peritonitis. There was no evidence of a neoplastic basis for the ulcer. The cat received a whole blood transfusion on the day following surgery. From then, he recovered slowly but steadily until he was discharged 8 days after initial presentation. While hospitalised, he had been administered cefalexin (20 mg/kg bid PO), omeprazole (1 mg/kg sid IV), sucralfate (250 mg tid PO) and prednisolone (1 mg sid PO) to avoid iatrogenic hypoadrenocorticism. The cat resumed spontaneous oral feeding once back home and his oesophagostomy tube was removed along with the skin staples 12 days after surgery. He was reportedly normal at that time.
Four months post-surgery, the cat was reported to have had no further occurrence of digestive signs and the signs related to his coxarthrosis seemed to be well controlled by the judicious administration of meloxicam (0.05 mg/kg sid PO).
Discussion
Gastric or gastroduodenal perforations are defined as being spontaneous when they arise in the absence of foreign body ingestion, gastric dilatation and volvulus, external trauma, leakage at previous gastrotomy or anastomotic sites, or iatrogenic trauma. 1 Typically, spontaneous perforations are associated with a gastric ulcer or neoplastic process.
Gastroduodenal ulceration, and potentially subsequent perforation, has been uncommonly reported in cats. The identified, or presumed, causes of gastric and duodenal ulcers in cats include neoplasia (lymphosarcoma and adenocarcinoma),2,3 inflammatory bowel disease, 4 stress related to surgery, 5 gastric hyperacidity due to systemic mastocytosis3,6,7 or gastrinoma,8,9 carprofen administration, 10 hypereosinophilic syndrome, 11 bacterial granuloma, 12 and toxicity following ingestion of Dieffenbachia leaves. 13 However, a cause for ulceration/perforation was not found in a significant proportion of reported cases.3,14–17
The three cases presented here represent the only cats with spontaneous gastric perforation seen at our institution over the past 3 years. The presenting signs, physical findings and laboratory results of these cats were essentially non-specific. This is in agreement with previous reports.1,3,18 In particular, there were no signs referable to gastric bleeding (haematemesis, melaena), decompensated shock and pain on abdominal palpation. Only one of the cats presented with anaemia. Conversely, anaemia was detected in all cats with gastric ulcers in the largest series of feline patients. 3
The most informative diagnostic test performed in our series was abdominal radiography. By demonstrating pneumoperitoneum, in the absence of recent abdominal surgery or abdominocentesis, an indication to explore the abdomen was presented. The cytological results of the later performed abdominocentesis in case 2 provided further support for surgical exploration. Indeed, spontaneous pneumoperitoneum may be associated with rupture of a gastrointestinal or urogenital viscus,16,19 transmural migration of gas across bowel with vascular compromise, 19 or infection with gas-producing bacteria within the abdominal cavity, 20 all of which should be managed surgically. Furthermore, surgery allowed a definitive diagnosis to be made through identification of the lesions and harvesting of specimens for histopathology.
None of the three cases in this report required decompression of the pneumoperitoneum prior to anaesthesia. Tension pneumoperitoneum — defined as a life-threatening, massive accumulation of gas within the peritoneal cavity, necessitating immediate needle decompression to improve cardiorespiratory embarrassment — has been reported in one cat. 17
It is possible, although speculative, that meloxicam and prednisolone administration contributed significantly to the aggravation of pre-existing gastric lesions to the point that perforation developed, requiring surgical attention.
In human medicine, the introduction of COX-2 preferential and selective NSAIDs has increased NSAID use, which has coincided with an increased incidence of non-fatal digestive perforations and haemorrhages in the presence of NSAID administration. This observation may also be relevant to the feline species.
Surgery has been documented to significantly increase the survival rate of cats with a pneumoperitoneum or with gastric ulcers.3,19 It resulted in an excellent outcome in the three cats of our report. A thorough exploration of the abdominal cavity was performed with a view to locating the lesions, detecting imperforated ulcers via the adhesions or mass effect they are usually associated with, 3 and identifying extraintestinal neoplasms that may have caused gastric ulceration/perforation. The compromised ulcerated areas were debrided with 1 cm of healthy-looking gastric wall, as is the current recommendation for surgical management of gastric ulcers, 21 and the site of partial gastrectomy was closed primarily. In all cases, careful inspection of the gastric mucosa was performed through the partial gastrectomy window or through the original gastrotomy to ensure that no other ulcerative lesion was present. More extensive surgery, such as a Billroth I, has been performed successfully in a cat with a spontaneous duodenal perforation. 10
The location of the perforating gastric ulcers in our cases was consistent with previous reports, which described them as being almost exclusively pyloroantral or fundic in cats with non-neoplastic gastric lesions. 3 All cats not only survived long term but also rapidly regained a level of comfort and activity equivalent to that prior to surgery, similar to a previous study. 3 These reports compare favourably with the 17% survival rate reported in the only study focusing on the outcome of cats presented with spontaneous gastroduodenal perforations. 1
Two of our cats with spontaneous gastric perforation had been given potentiated amoxicillin and meloxicam before the onset of their presenting signs. Potentiated amoxicillin has not been described as having ulcerogenic effects. By contrast, meloxicam has previously been implicated as a causal factor of gastroduodenal perforation in dogs. 22 Generally speaking, NSAIDs, including the most recently developed COX-2 preferential NSAIDs, have been demonstrated to be potentially ulcerogenic in dogs.1,22–24 Despite experimental evidence of the ulcerogenic effects of NSAlDs in cats, 15 NSAlD-induced gastroduodenal ulceration has only been reported in one clinical case. 10 The administration of injectable carprofen, which at the time was a non-licensed form of NSAID in cats, was incriminated. So, to the authors' knowledge, this study represents the first report of likely meloxicam-associated gastric ulceration/ perforation in cats. To our knowledge, this is also the first report implicating chronic administration of prednisolone in spontaneous gastric perforation.
It should be emphasised, however, that anti-inflammatory drugs are typically administered when another disease process is present. In our cases, these disease processes were post-traumatic degenerative osteoarthritis and urinary bladder inflammation. In this respect, it is impossible to ascertain that the primary disease process and the likely stress related to it, in terms of discomfort, reduced appetite and visits to the vet, did not play a part in the aetiology of the gastric ulceration/perforation. In the third reported case, it is also likely that the stress and reduced gastric perfusion related to the anaesthesia and dental procedure played a part in the occurrence of gastric ulceration. 25
Although similar studies do not exist in cats, studies on the more recently developed COX-2 preferential NSAIDs, particularly meloxicam and carprofen, have demonstrated an excellent safety profile with regards to adverse effects on the gastrointestinal tract, both experimentally and clinically.26–30 It is likely that the aetiology of gastric ulceration and perforation was multifactorial in the reported cases. It is possible, although speculative, that meloxicam administration (either as a single subcutaneous injection or chronic oral administration, complying with recommended doses and licensed use) and prednisolone administration contributed significantly to the aggravation of pre-existing gastric lesions to the point that perforation developed, requiring surgical attention.
Several authors in the veterinary literature have recommended that practitioners should be aware of the potential development of gastroduodenal ulcerations and perforations, particularly with the increasing use of NSAIDs.24,30 In human medicine, the introduction of COX-2 preferential and selective NSAIDs has increased NSAID use, which has coincided with an increased incidence of non-fatal digestive perforations and haemorrhages in the presence of NSAID administration. 31 This observation may also be relevant to the feline species. The challenging diagnosis of gastric ulceration/perforation in the cat should highlight the importance of thorough patient evaluation in the presence of nonspecific clinical signs and a history of antiinflammatory drug administration.
