Abstract
The objective of this paper was to evaluate the efficacy of a hypofractionated radiation protocol for feline facial squamous cell carcinoma (SCC). Twenty-five histologically confirmed SCCs in 15 cats were treated with four fractions of 7.6–10 Gy each, with 1 week intervals. The equipment used was a linear accelerator Clinac 2100 delivering electron beam of 4 or 6 MeV, and a bolus of 5 or 10 mm was used in all lesions. Of the lesions, 44% were staged as T4, 16% as T3, 8% as T2 and 32% as T1. Of the irradiated lesions, 40% had complete response, 12% had partial response and 48% had no response (NR) to the treatment. For T1 tumors, 62.5% had complete remission. Mean overall survival time was 224 days. Owners requested euthanasia of cats having NR to the treatment. Mean disease free time was 271 days. Side effects observed were skin erythema, epilation, ulceration and conjunctivitis, which were graded according to Veterinary Radiation Therapy Oncology Group (VRTOG) toxicity criteria. Response rates found in this study (52%) were lower when compared to other protocols, probably due to technique differences, such as fractionation schedule, bolus thickness and energy penetration depth. However, the hypofractionated radiation protocol was considered safe for feline facial SCC. Modifications of this protocol are being planned with the objective of improving the cure rates in the future.
Radiation therapy is a cancer treatment modality. Its main goal is the death of neoplastic cells sparing the healthy adjacent tissue. 1 The squamous cell carcinoma (SCC) is the most common skin tumor in cats, and it is related to the ultraviolet light exposure. 2–4 The nose, eyelids and ears, especially of white cats, are more commonly affected. 2–4 Bowen's disease or multiple SCC in situ is a rare condition that affects pigmented and haired skin of cats, and it is not related to ultraviolet light exposure. 5 Several treatment modalities have been used in the treatment of SCC, which include surgery, 6–9 cryosurgery, 10,11 systemic 12,13 and intralesional chemotherapy, 14 hyperthermia, 15 photodynamic therapy, 16 external beam radiation therapy 17–20 and plesiotherapy, 21,22 with different results.
Radiation therapy is applied in multiple treatments, called fractions, which is important for a better tumor control and minimization of side effects. 2,4,17 Several radiation protocols have been tested for cutaneous SCC, including a monday–wednesday–friday schedule, 17,23 hypofractionated 18 and accelerated protocols. 19,20 A hypofractionated protocol has lower financial costs, greater convenience and fewer anesthesia procedures needed. The aim of this study was to determine the effectiveness and safety of a hypofractionated radiation protocol in cats with cutaneous SCC.
Materials and methods
The present study was approved by the local institutional Bioethics Committee. Fifteen cats with single or multiple SCCs were treated with a hypofractionated electron beam radiation protocol, in a total of 25 irradiated lesions. All tumors were located in the face of cats and were diagnosed as SCCs by histopathology. Bowen's disease was not diagnosed in any cat of this study.
Staging was undertaken according to the WHO classification for feline tumors of epidermal origin (Table 1). 24 Each cat was evaluated with hematological examination, blood urea nitrogen (BUN) and creatinine. Thoracic radiographs were made, including lateral and ventrodorsal views. No sedation was needed for this procedure. When regional metastasis was suspected, a fine needle aspiration of mandibular lymph nodes was made.
World Health Organization classification for feline tumors of epidermal origin, modification by Owen. 24

Treatments were undertaken on saturdays in the Clementino Fraga Filho University Hospital of the Universidade Federal do Rio de Janeiro (UFRJ), at the Radiation Therapy Unit. The equipment used was a linear accelerator Clinac 2100 delivering electron beam. The protocol used was four fractions of 7.6 or 10 Gy each, with 1 week intervals, in a total dose of 30.4 or 40 Gy. Tumor volume was considered the visible lesion and was calculated using a length×width×height model. The planned target volume was determined previously to the treatment with a felt pen, and a 1 cm margin was added to the tumor volume. Animal positioning depended on the facial location of the SCC. Cats with nasal tumors were positioned in sternal recumbency, and those with pinnae or temporal tumors were positioned in lateral recumbency. In many cases, folded towels were used as support for the head. In advanced nasal SCC with lip extension, a device for mouth opening was used. In all cats, a bolus was used on the tumor to allow radiation dose to be deposited superficially in the skin. Bolus thickness (5 or 10 mm) and energy (4 or 6 MeV) were applied according to tumor depth. The distance between focus and skin was 100 cm.
The drugs used for anesthesia were acepromazine maleate (0.03 mg/kg IM), meperidine HCl (3 mg/kg IM), ketamine HCl (2–4 mg/kg IV) and diazepam (0.2 mg/kg IV). In only one cat (E) another protocol was used, due to ketamine resistance, that consisted of acepromazine maleate (0.03 mg/kg IM), meperidine HCl (3 mg/kg IM) and tiletamine/zolazepam (3 mg/kg IV).
The neoplastic lesions were observed and photographed weekly during the treatment, and 30 and 60 days after its end. After this period, follow-up information was gathered through examinations, telephone conversations and photographs sent by clients. Maximum follow-up time was 2 years. Response was graded as complete response (CR) when the tumor completely regressed, partial response (PR) when there was a 50% or more reduction in the tumor size, and no response (NR) when less than 50% reduction in tumor size or tumor progressed. Owners requested euthanasia of cats having NR to the treatment.
Disease free interval was calculated from the last radiation treatment to the first detection of tumor return; considered as a new lesion in the previous radiation field. It was calculated only in the lesions that achieved CR. Survival was calculated from the beginning of therapy to the day of death or euthanasia. The Kaplan–Meier survival plot was used for survival analysis, and Spearman's rank correlation was used to determine the relationship between variables. Spearman's coefficient (Rs) ranged from 0 (no correlation) to 1 (maximum correlation), and varied according to the number of variables used.
Reactions to radiation therapy were graded according to the toxicity criteria of the Veterinary Radiation Therapy Oncology Group (VRTOG) (Table 2). 25
Toxicity criteria of the VRTOG (Veterinary Radiation Therapy Oncology Group). 25

These criteria are adapted from the guidelines established by the VRTOG. Only skin, oral cavity, eye and ear are shown, because these were the organ systems irradiated in this study.
Results
All cats were totally (53.3%) or partially (46.7%) white, and had frequent sun exposure. There were 14 females and only one male, with mean age of 11 years (7–17 years). Only three cats were intact. Nine lesions (9/25) had been submitted to previous treatments, including surgery (5/9), fused silver nitrate stick (3/9), and cryosurgery (1/9) (Table 3).
Information about the 15 cats with 25 lesions of squamous cell carcinomas treated with radiation therapy.

N=none.
The most commonly affected site was the pinna (40%), followed by nasal planum (32%), eyelid (16%) and tempora (12%). According to WHO classification, 24 eight lesions were staged as T1, two were staged as T2, four staged as T3 and 11 staged as T4 (Table 3). None of the cats had evidence of regional or distant metastasis at initial presentation. One cat (D) was euthanased in the third radiation treatment due to pulmonary metastasis, diagnosed by radiography and histopathology, made at necropsy.
Concomitant diseases were present in four cats, and included chronic renal failure, hypertrophic cardiomyopathy, feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV). The cats with positive status for FIV and FeLV had no clinical or hematological evidence of disease, and were considered asymptomatic.
None of the cats had anesthetic complications during radiation treatments. Energy of 4 MeV and 6 MeV were used in 17 and eight lesions, while bolus thickness were 5 mm and 10 mm in 14 and 11 lesions, respectively. In the total of 25 lesions irradiated, 10 (40%) were considered as CR, three (12%) as PR and 12 (48%) as NR (Table 4 and Fig. 1). T1 and T2 lesions had 55.5% of complete remission, while T3 and T4 had 31.25%. If only T1 lesions were considered, complete remission would be of 62.5%. Owners requested euthanasia of cats having NR to the treatment. Three SCC lesions are still in complete remission and seven lesions recurred. Maximum follow-up time was 2 years, with median of 5 months, and no cat was lost to follow-up.
Radiation techniques, survival, disease free interval, follow-up and acute reactions information of the cats with squamous cell carcinoma treated with radiation therapy.

Cat still alive.
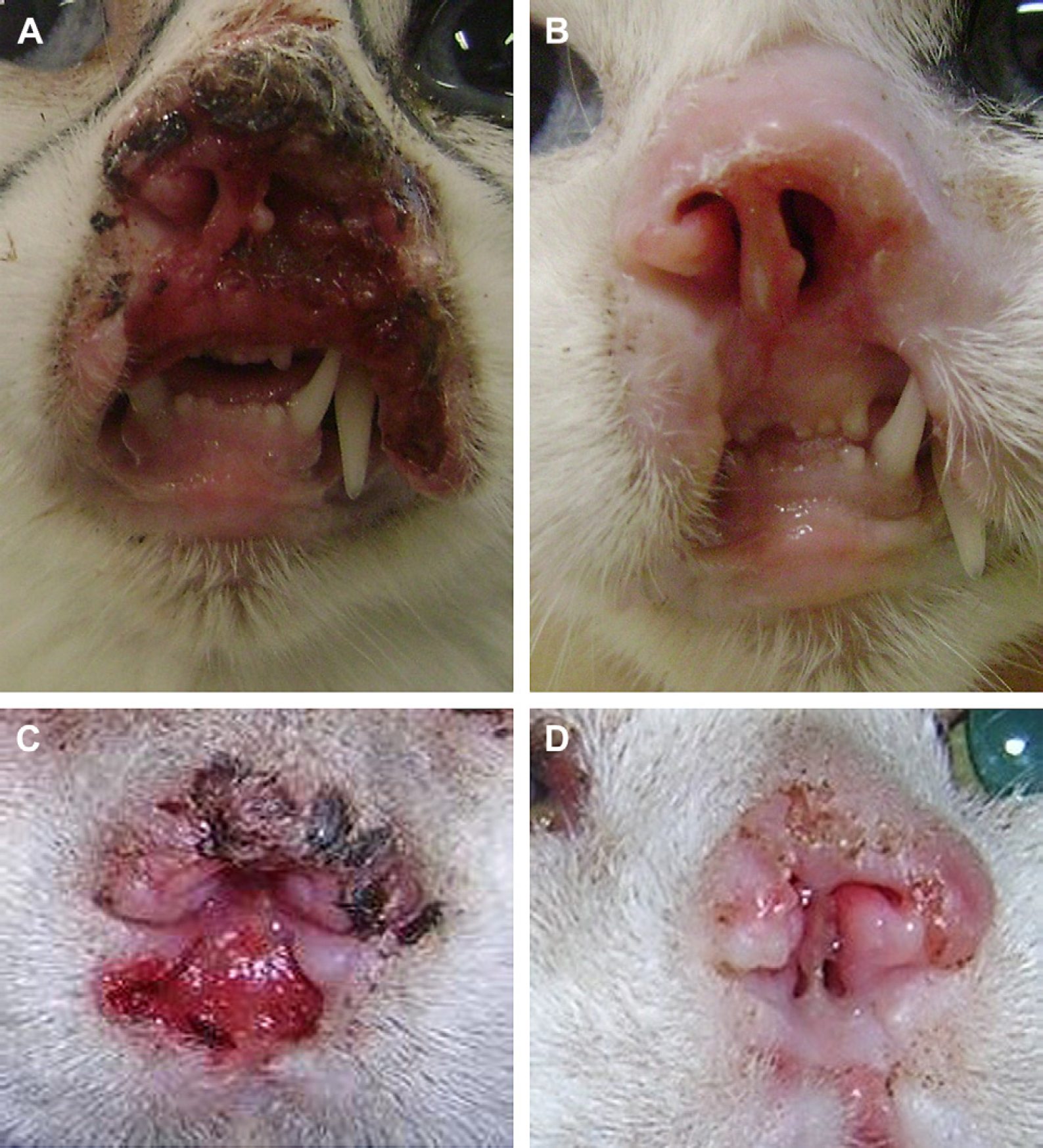
Hypofractionated radiation therapy for feline squamous cell carcinoma. Photograph of lesion 9 before (A) and after (B) radiation treatment, showing CR. Photograph of lesion 10 before (C) and after (D) radiation treatment, showing CR.
Acute reactions occurred in 12 (48%) lesions and were considered mild/moderate, according to VRTOG. 25 Reactions included alopecia, conjunctivitis, skin erythema and ulceration (Table 4 and Fig. 2). Only one acute reaction of conjunctivitis required treatment with topical non-steroidal anti-inflammatory medication and Elizabethan collar. Late reactions were not yet observed in the surviving cats.

Acute reactions to hypofractionated radiation therapy observed in cats with squamous cell carcinoma. (A) Conjunctivitis in cat H. (B) Erythema in cat C. (C) Epilation in cat J. (D) Nasal ulceration in cat E.
Statistical analysis
Mean survival time of the cats treated with hypofractionated radiation therapy was 224 days (16–750). Mean disease free interval of the lesions that achieved CR was 271 days (64–720). Kaplan–Meier survival plot was used for survival analysis only (Fig. 3).

Kaplan–Meier survival plot of 15 cats with squamous cell carcinoma treated with hypofractionated radiation therapy.
Spearman's rank correlation showed lack of correlation between tumor staging and tumor response to radiation treatment (Rs=−0,341; P=0.096). Thus, staging the SCC lesions as T1, T2, T3 or T4 did not affect its response to the hypofractionated radiation therapy.
Discussion
In this study, 10/25 SCC irradiated lesions had a CR and three had a PR to hypofractionated radiation therapy, giving a response rate of 52% – lower than previous papers. Several issues may have lead to these results, including the high number of advanced lesions (T3 and T4) included, and technique differences, such as fractionation schedule, bolus thickness, kind of energy and penetration depth.
Cox 23 used orthovoltage and Cobalt 60 megavoltage in 11 cats with nasal SCC, and the protocol used was 6–10 fractions of 3.75–4.4 Gy, three times a week. The paper does not mention the general response rate, but 61.5% of the cats survived 1 year, 23% survived 2 years and 15% survived 3 years. Theon et al 17 treated 90 cats with SCC with orthovoltage, using 10 fractions of 4 Gy, three times a week. T1 tumors had 56% of CR, but general response rate was not mentioned. Of the cats, 74.3% survived 1 year. Fidel et al 19 used an accelerated protocol of protons with energy of 72 MeV and total dose of 40.4–44.8 CGE (60Co Gy equivalent). The energy (protons) was chosen based on its superior depth-dose distributions, and the accelerated fractionation protocol was used with the objective of overcoming tumor repopulation capabilities. Of the cats, 60% had CR and 33% had PR. Vos et al 26 used orthovoltage in six cats with advanced nasal SCC, with a protocol of 12 fractions of 4 Gy, three times a week, associated with intralesional chemotherapy. Carboplatin was injected inside the tumor 30 min before radiation fractions, acting as a radiopotentiating agent. With this protocol, 100% of complete remission was achieved. Melzer et al 20 used an accelerated electrons beam protocol (that consisted of 10 fractions of 4.8 Gy during four consecutive days and minimum intervals of 6 h) in 17 cats with SCC. The energy of 5–16 MeV was delivered by a linear accelerator and bolus thickness was 2–4 mm. With this protocol, 94% of the SCC lesions had complete remission.
It is notable that the type of energy (photons, protons or electrons), equipment (orthovoltage or megavoltage), energy depth and fractionation schedules were extremely varied in previous literature. Only one paper 20 used the same energy (electrons) of the current study, and therefore results could be comparable. However, the fractionation schedules were quite different.
The fractionation of the radiation treatment is important for several reasons. The presence of cells in various stages of the cell cycle (M, S, G1, G2, and G0) is a major factor in the response to radiation, as cells in S phase are more resistant, and those in phases G1 and G2 are more sensitive to the effects of radiation. 27 During the interval between fractions, cells in S phase enter the cell cycle and progress to the responsive stages, in a process known as redistribution. 4,27 It is feasible to think that many cells that were not in the sensitive phases when irradiated should be re-irradiated as they progress in the cell cycle. 1,27 With 1 week intervals between fractions, tumor repopulation occurs. Although hypofractionation has advantages, including greater convenience and lower cost, it allows tumor repopulation.
Other important difference between Melzer's paper and the current study is the energy penetration depth. The energy intensity and bolus thickness determine radiation depth in the neoplastic tissue. Melzer et al 20 used 5–16 MeV, with bolus of 2–4 mm, while we used 4–6 MeV with bolus of 5 or 10 mm. It is possible that our energy depth was not adequate for advanced SCC lesions.
Advanced lesions, including T3 and T4, were the majority of the irradiated lesions (60%), and may have interfered in response rates, even if unproved by our statistical analysis. Probably, the small number of treated lesions of this study might be considered low for statistical conclusions. It is known from previous literature that advanced SCC tumors respond poorly to radiation therapy when compared to superficial lesions. 17,19 In addition, they interfered in survival times, because radiation therapy was believed to be the last treatment option for those cats with advanced lesions and if treatment was unsuccessful, euthanasia was undertaken.
Although dose per fraction was extremely high when compared to other protocols, which in theory could lead to severe acute reactions, these were considered mild/moderate. According to the literature, the acute reactions in the skin are erythema, dry or moist desquamation and epilation. 2,4,28 The late reactions can occur months or years after radiation therapy, and include depigmentation, irreversible alopecia, fibrosis, necrosis, fistulas formation, chronic conjunctivitis, keratoconjunctivitis sicca and cataracts. 2,4,28 In this study, the side effects observed were considered mild/moderate according to VRTOG and included erythema, epilation, ulceration and conjunctivitis, which were easily resolved with supportive treatment. There were no late reactions to treatment until this date, but it is still possible that they will occur in the next months or years.
The hypofractionated radiation protocol presented in this study was well tolerated by the cats, and lead to a complete remission of 40% and partial remission of 12%, with an overall response rate of 52%. However, further modifications of this protocol are being planned, including energy depth and bolus thickness, with the objective of improving the cure rates in the future.
Footnotes
Acknowledgements
The funding for this research was provided by CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico).
