Abstract
Polycystic kidney disease (PKD) is one of the most common genetic diseases in cats. It has been widely described in Persians and Persian-related cats and sporadically in other breeds. The purpose of the present paper is to describe the first reported case of PKD in a 12-year-old female Chartreux cat. The cat was referred with polyuria and polydipsia and enlarged and irregular kidneys at palpation. Multiple renal cysts and a single liver cyst were identified by ultrasound and the inherited pattern was confirmed by genetic test (polymerase chain reaction/restriction fragment length polymorphism (PCR/RFLP) assay). Chartreux cats should be included in the screening programme of PKD, and PKD should be always considered as a possible cause of chronic renal failure in this breed.
Autosomal dominant polycystic kidney disease (AD-PKD) is the one of the most prevalent inherited genetic disease in cats affecting 36–49.5% of Persians and Persians related cats such as Exotic Shorthairs and mix-breeds. 1–6 The disease is characterised by renal and less frequently hepatic and rarely pancreatic cysts, 7 that can be easily detected by ultrasound screening. A genetic mutation was identified in the PKD1 gene which was linked to AD-PKD. Two types of genetic tests are currently available (RFLP and real-time PCR). 8,9 The prevalence of the disease in other breeds is less known. It has been reported sporadically in Burmillas, domestic shorthairs, Himalayans, American Shorthairs, Scottish Folds, British Shorthairs and Ragdolls. 1,10,11
The purpose of this paper is to describe the first reported case (to the author's knowledge) of PKD in an adult Chartreux cat.
A 12-year-old spayed female Chartreux cat was presented to the referring veterinarian with a history of 1-month duration of polyuria and polydipsia and 3-day duration of vomiting.
The cat was slightly depressed, clearly underweight, and moderately dehydrated (6%). The kidneys were enlarged and dented at palpation. Systolic blood pressure was high (220 mmHg). Serum biochemical analysis revealed moderate uraemia (24.59 mmol/l; normal range 3.57–11.78 mmol/l) and severe creatinaemia (397.8 μmol/l, normal range <176.8 μmol/l), mild hyperglicaemia (8.08 mmol/l, normal range 2.97–5.50 mmol/l) and hypercholesterolaemia (6.52 mmol/l, normal range 1.82–3.9 mmol/l). The cat was negative for feline immunodeficiency virus, feline leukaemia virus and feline coronavirus. The value of T4 was normal. Urinalysis was unremarkable (specific gravity: 1030, normal range 1015–1045; pH: 5.80, normal range 5.70–7.20; absence of glucose, bilirubin, blood, leukocytes and ketonic acid, urinary protein/creatinine ratio: 0.43, normal range <0.60). The result of urine culture was negative.
The cat underwent ultrasonographic abdominal examination at the Veterinary Teaching Hospital of the University of Parma. A 7.5–10 MHz linear transducer (Megas GPX, Esaote, Genova, Italy) was used, set at 10 MHz. Both kidneys presented several round anechoic or hypoechoic cavities, with a smooth, sharply demarcated thin walls and a distinct far-wall border. The greatest number of cysts was located in the cortex. The cysts were very large, and completely altered renal contour and parenchyma (Fig 1). The volume of both kidneys was increased, and the distinction cortex–medulla was deformed. The left kidney showed a mild pelvic dilation. The liver showed a single cyst (2×1 cm) with thin septa located in the right median lobe (Fig 2). The reminder of the abdominal organs was normal.
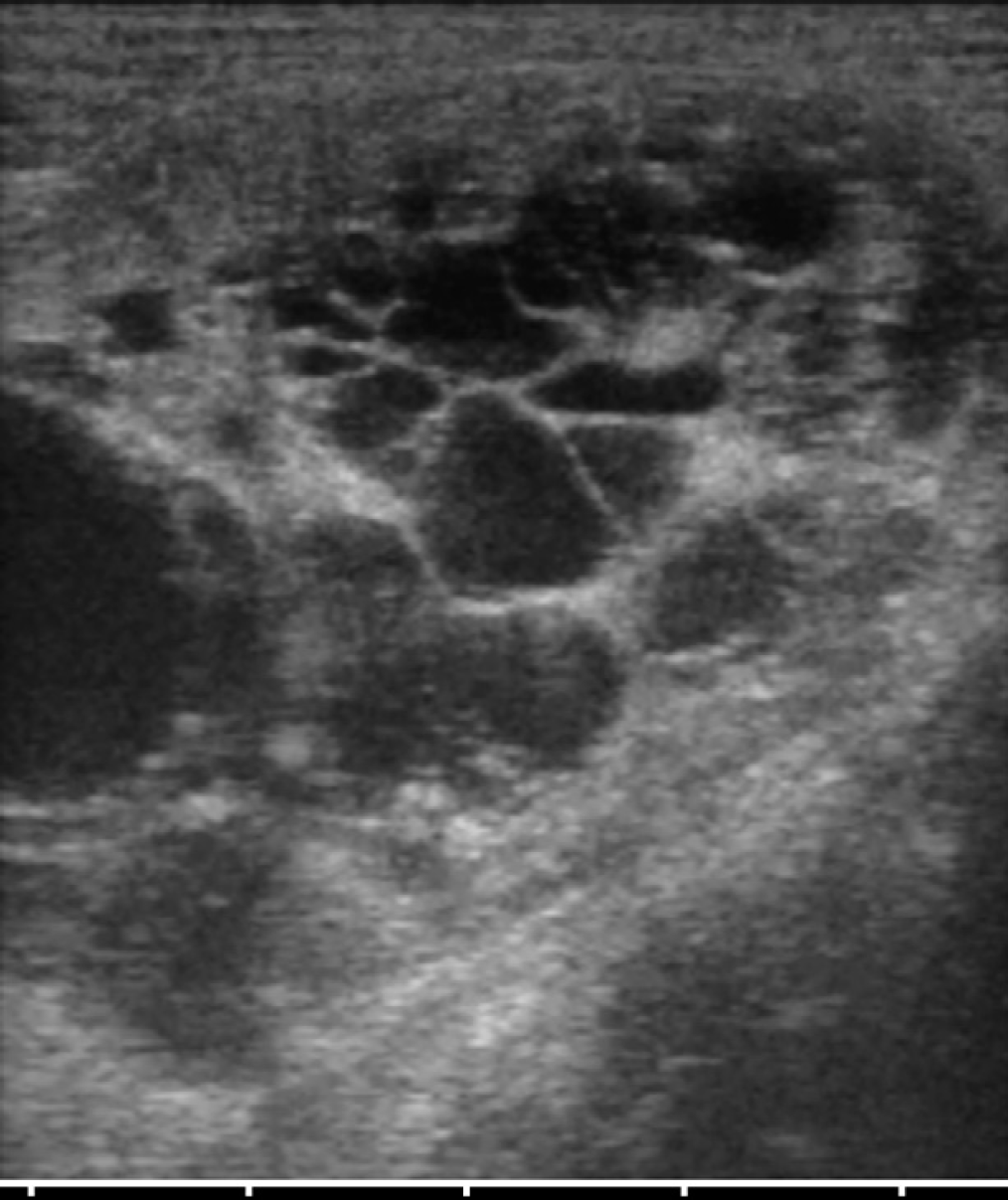
Several cystic structures in the renal parenchyma that completely altered normal architecture.
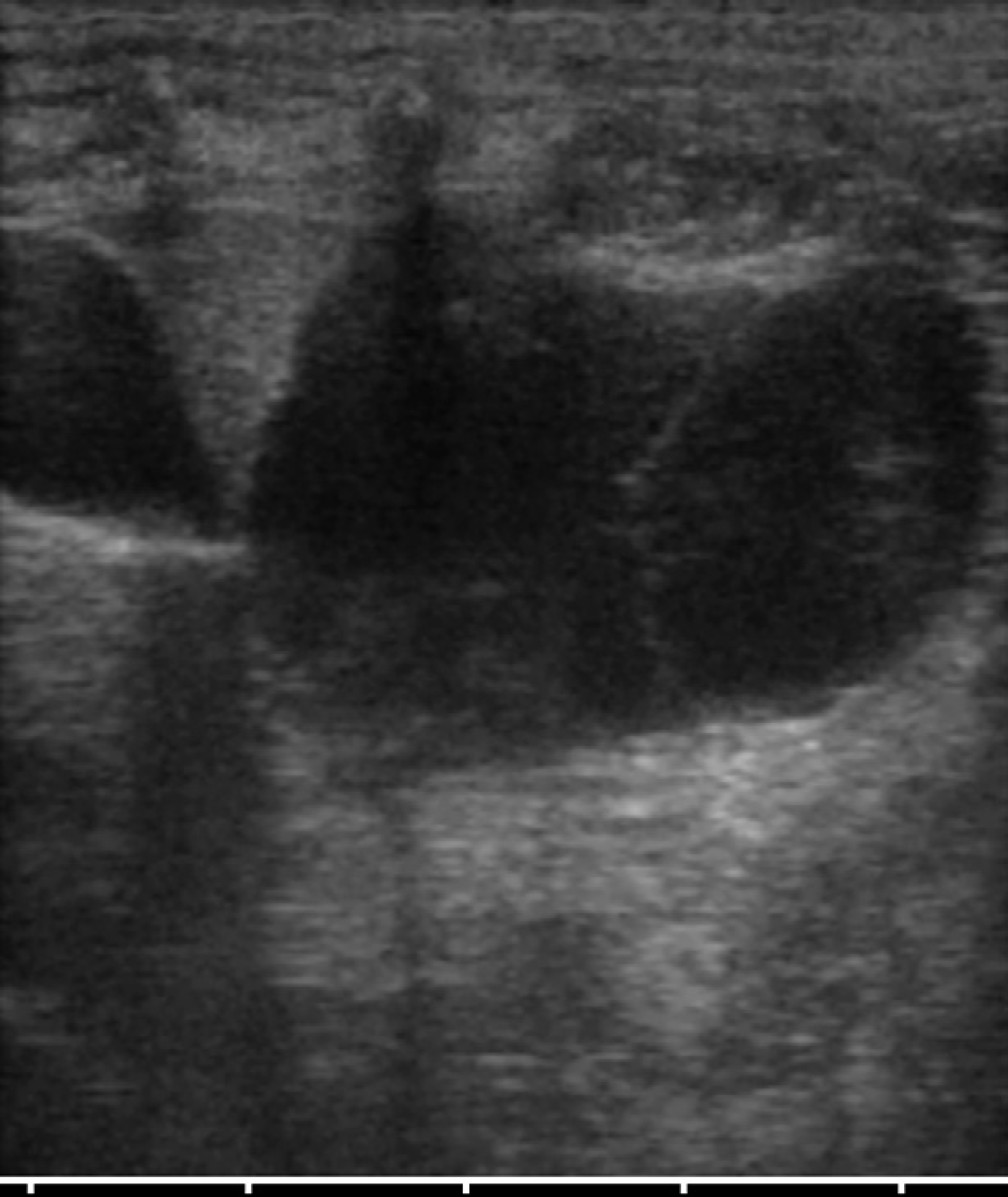
A single cyst with thin septa in the left medial hepatic lobe.
The final diagnosis was PKD and liver disease causing chronic renal failure that was classified at stage III in IRIS classification, non-proteinuric and hypertensive. To confirm the inherited pattern a genetic test was performed (PCR/RFLP) on blood sample, that resulted positive for PKD1 gene mutation.
The cat was treated with an intravenous infusion of Ringer's lactate (2.5 ml/kg/h IV) and ranitidine (2 mg/kg bid IV) for 2 days by the referring veterinarian. The cat was then discharged on a diet with a low content of phosphate, proteins and sodium. Ranitidine was continued for another 5 days. Enalapril (0.25 mg/kg sid PO) was prescribed to manage hypertension, with re-check of the blood pressure and urea and creatinine the following week and then, every 2 weeks. At follow-up 2-months later, the cat was doing well and no complications were reported by the owner, except for the persistence of high systolic blood pressure (200 mmHg). Enalapril was then replaced by amlodipine (0.625 mg sid PO). At the re-check 2 and 4 weeks later the systolic blood pressure was decreased (160 mmHg). At follow-up 4 months later the blood pressure was stable under 165 mmHg. However, the disease is irreversible and progressive, and the prognosis is guarded.
AD-PKD is an inherited disease identified in Persian cats, Exotic Shorthairs, mixed breeds and sporadically in other breeds. 1,10,11 The most important clinical manifestation is chronic renal failure. Cysts form from renal tubular cells and eventually separate from the nephron as they develop. As cysts enlarge, they compress surrounding normal renal parenchyma and lesions of chronic interstitial nephritis occur. As the disease advances, cysts increase in size, and renal volume increases. For these reasons, PKD is considered a progressive disease, and the prognosis is guarded. 1,12,13
The cat of the present report had clinical evidence of renal disease such as polyuria and polydipsia, with moderate azotaemia at the age of 12, managed successfully with conventional medical approach. Enalapril was inadequate to control high systemic blood pressure, which was subsequently stabilised with amlodipine. Amlodipine is actually the treatment of choice for feline hypertension. 14
Ultrasound was of value in the diagnosis of PKD, in evaluating the size and the number of the cysts and in demonstrating a cyst in the liver. High frequency transducers should be used to have a good resolution (>7 MHz). The genetic test was essential to confirm the inherited pattern.
According to the literature, ultrasonography has sensitivity of 91% at 9 months of age, increasing with the age of the cat, while specificity is said to be 100%. 1–3,5,12 In a recent study, however, ultrasound resulted to have a sensitivity of 96.2% at the age of 3 months. 15
The prevalence of PKD in Italy is about 41% in Persians and Exotic Shorthairs. 4 The prevalence of PKD in the Chartreux cats in Italy is unknown. The Chartreux cat Italian Club currently does not follow a screening programme for PKD, because there are no reports of the disease in this breed.
Once the disease is reported in a specific breed, it becomes essential to establish a prevalence and to start a screening programme in order to document the eradication process. Eradication of the disease should be achieved by screening all cats used for breeding and retaining only unaffected cats for future breeding programmes. 2 Unfortunately, the relatives and antecedents of the cat in this report could not be found and tested.
The progressive nature of PKD that leads to irreversible renal failure, should raise the interest of veterinarians and breeders of cats that have current or past links with Persians. Actually, Chartreux cats have been crossed with British Blues, Russian Blues and Persian Blues since the end of World War II to 1977. From 1970 to 1977 the registries of British Blues and Chartreux cats were amalgamated, being again separated after 1977. 16 This could be the potential route by which the AD-PKD gene was introduced to the breed.
In conclusion, this report points out that Chartreux cats should be included in the screening programme of PKD, and PKD should be considered as a possible cause of chronic renal failure in this breed.
Footnotes
Acknowledgements
The authors thank Mr Giuseppe Bertaccini for technical assistance.
