Abstract

This month's JFMS has an interesting case report from a team of Brazilian vets struggling to cure a refractory case of feline sporotrichosis, a disease seen in epidemic proportions in that country. 1 Having failed to resolve the problem with a long course of oral itraconazole therapy, they resorted to administering a concentrated solution of amphotericin B intra-lesionally (0.8–1.0 ml of a 5 mg/ml solution) using a very fine needle. The treatment proved remarkably effective in this instance, with the patient cured after three injections given at 1-week intervals. The authors briefly reviewed the literature concerning the use of intra-lesional amphotericin B in feline, equine and human patients with localised fungal infections.
Infections of the skin and subcutis with saprophytic fungi occur sporadically in cats. These infections can be challenging to treat. Generally, they are more common in warmer climates, but even in cold climates (like the UK), certain fungal species seem to be problematic, eg, Alternaria species. 2 Generally, these are localised infections of immune competent cats. Pathogenesis usually involves penetrating injuries which introduce fungal elements (hyphae or spores) into the tissues. Cat scratch wounds are a common cause of this type of infection, and the naso-ocular region is often involved. 3 Puncture wounds of the distal limbs also account for some cases ( Fig 1 ).

Infection of the distal limb of a cat with Wangiela dermatiditis, a pigmented fungal organism that can grow in mammalian tissues as a yeast or hyphae. In this instance, surgery (A) was used to debulk the fungus impregnated tissue (B; close-up photo on a gauze swab) between the phalanges. Systemic antifungal therapy was used subsequently to eliminate residual fungal elements from the tissues. Intra-lesional amphotericin B may prove useful in cases such as this, either instead of or subsequent to surgery.
These cases are readily diagnosed using cytology, histology and mycologic culture (ideally at, or in collaboration with, a human reference laboratory as this allows definitive species identification by morphology and/or polymerase chain reaction methods and accurate susceptibility testing). Treatment generally involves surgical removal (or at least ‘debulking’) of the lesion, followed by a long course of systemic antifungal therapy. Typically a drug with a wide spectrum of activity like itraconazole, or better still, posaconazole 4 is selected for empiric therapy while awaiting results from the reference laboratory. The subsequent choice of agent(s) is based on in vitro susceptibility testing and information from the literature.
There can be problems, however: (i) some fungal isolates show resistance to many antifungal agents either in vitro or in vivo, (ii) the more modern drugs (posaconazole, voriconazole, caspofungin, liposomal amphotericin B) can be prohibitively expensive, (iii) there are sometimes toxicity considerations and (iv) therapy must be continued for many months, usually 2–3 months past complete resolution of the lesions. Finally, although complete surgical excision is ideal, infections tend to occur on the cat's face or distal limbs, areas where even marginal resection is difficult. Thus, debulking may leave quite a large amount of residually infected tissue ( Fig 1 ).
The notion that intra-lesional amphotericin B can be safely used as sole or adjunctive therapy adds a very worthwhile therapeutic weapon in the management of these cases. This is especially the case in veterinary medicine where the cost of medication or complex surgery can be a critical consideration.
But, we need to know a bit more before we can create firm guidelines.
What is the ideal concentration of amphotericin to use intra-lesionally? Is there any benefit in mixing amphotericin B with procaine?5,6
Must it be given in concert with systemic azole therapy?
What is the ideal volume to inject per lesion?
How frequently should doses be administered?
Can treatments be given under heavy sedation, or are they so painful that full general anaesthesia is required?
Does intra-lesional injection ever cause sloughing of the affected tissues?
Does it lead to scarring or permanent hair loss at the sites of injection?
Do cats become systemically unwell as a result of this therapy?
Do they require opiate pain relief after the procedure?
Can other water-soluble antifungal agents be given in the same way?
The only way to get these answers is by trying this approach in a carefully selected series of cases, and reporting the clinical outcomes in a meaningful number of consecutive patients. There is a reasonable amount of information concerning intra-lesional amphotericin B in human patients. For example, a recent case of alternariosis in a renal transplant patient was treated using stock amphotericin solution diluted with four volumes of 0.5% procaine hydrochloride to give a final amphotericin B concentration of 1 mg/ml; 1 ml was injected directly into the lesions twice a week for 5 weeks. The injections were well tolerated, in spite of moderate pain in the location of the injection. 6 Human experience, plus the current paper in JFMS, should provide us with a place to start.
This strategy may also be helpful in the management of pseudomycetomas in Persian cats caused by Microsporum canis, 7 combined with surgery (for the largest and/or most accessible lesions) and long-term azole therapy. This clinical situation is conceptually different, in that it appears to develop as a result of an inability to effectively mount an adequate immune response against a host adapted dermatophyte in particular breed(s) of cat. Intra-lesional amphotericin B may also have a place in the management of cutaneous infections cause by Leishmania species, Pythium species and Prototheca species.
Intra-lesional amphotericin B has proven to be useful in the hands of equine veterinarians treating nasopharyngeal fungal infections. 8 Perhaps it would prove useful also for those very frustrating infections of the sinonasal cavity with Neosartorya/Aspergillus species that penetrate the nasal bones to involve the nasal bridge, orbital ( Fig 2 A and C) and pharyngeal tissues.4,9 Computed tomography (CT)-guided infiltration of infected tissues with amphotericin B, caspofungin or voriconazole maybe a useful adjunct to therapy. This might be utilised instead of, or as well as, surgical cytoreduction ( Fig 2 B), and would permit sequential treatments along the lines described by Gremião and colleagues. 1 More information is required to determine whether this technique can be used safely when the optic nerve is so close.

Russian Blue cat with bilateral orbital infection with a Neosartorya species immediately before (A) and during (B) surgical cytoreduction. In (A), one can appreciate how the globes have been pushed forward by infection within the orbit, while removal of fungus impregnated tissue (arrow) is shown in (B). There maybe a place for intra-lesional amphotericin B, voriconazole or caspofungin as an adjunct in cases such as this. A CT scan from a similarly affected cat is shown in (C); the arrow highlights tissue infiltrated with fungal elements that could be the target for intra-lesional amphotericin therapy. Note the erosions in the bone separating the orbit from the nasal cavity.
The concept of intra-lesional therapy might also be usefully extended to the treatment of focal bacterial infections caused by organisms with a propensity to be drug-resistant – such as mycobacteria that cause feline leprosy10,11 and related diseases and canine leproid granuloma syndrome12,13 – substituting amphotericin B with a water-soluble anti-bacterial agent, such as amikacin or clofazimine ( Fig 3 ).
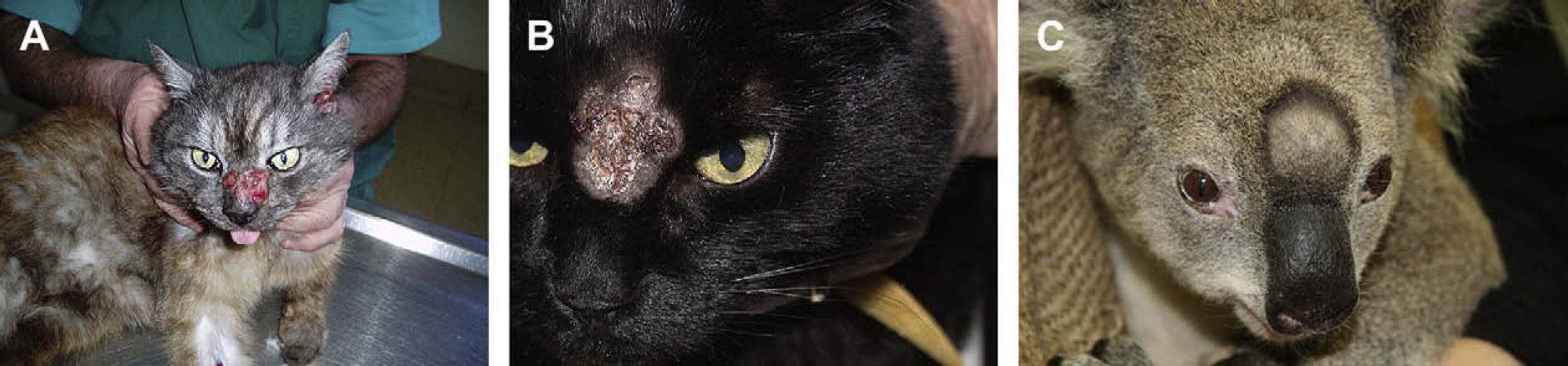
Cryptococcosis with involvement of the skin and/or subcutis in two cats (A, B) and a koala (C). Intra-lesional amphotericin B may have a place in the management of cases such as this, in concert with systemic therapy, although it was not used in these particular patients.
Finally, while discussing unconventional techniques for managing focal infections in a cost effective manner, there is some information that cryotherapy can be useful for certain superficial fungal diseases of the integument. 14
