Abstract


Echocardiography: The key to diagnosis
Echocardiography is the most important tool for diagnosing myocardial disease in cats, and has become widely available in veterinary practice to provide a non-invasive assessment of cardiac anatomy and function. The process of performing a basic echocardiographic examination is very intuitive, and examiners can quickly learn to recognise basic patterns of myocardial disease (Table 1). However, clinicians should be aware that echocardiographic results may show intra- and inter-operator variabihty, which is mainly dependent on operator skill and experience.1,2
General guidelines for echocardiographic classification of feline CM

NB. A definitive diagnosis of CM should
HCM = hypertrophic cardiomyopathy, HOCM = hypertrophic obstructive cardiomyopathy, RCM = restrictive cardiomyopathy, DCM = dilated cardiomyopathy, ARVC = arrhythmogenic right ventricular cardiomyopathy, UCM = unclassified cardiomyopathy. For explanations of IVRT, DT, E:A ratio and AR, see box on page 186
Most cats tolerate a full echocardiographic study without sedation. However, sedation may become necessary when the examination is made difficult by a restless patient or a stress-induced tachycardia. Although sedation may affect some echocardiographic measurements,3,4 the magnitude of observed changes in sedated cats is usually insufficient to influence the diagnosis. Furthermore, a study has recently shown that sedation with acepromazine and hydromorphone does not affect two-dimensional (2D) and M-mode echocardiographic measurements. 5 Propofol appears to induce a mild reduction in myocardial systolic velocity but does not alter diastolic function on tissue Doppler studies. 6
M-mode echocardiography
M-mode echocardiography allows accurate measurement of myocardial thickness and ventricular chamber diameter during the different phases of the cardiac cycle. Systolic anterior motion (SAM) is also documented on M-mode interrogation of the mitral valve as abnormal movement of the septal leaflet towards the interventricular septum (IVS) in early to mid-systole. Superimposition of colour Doppler on the M-mode interrogation of the mitral valve can document the blood flow turbulence during SAM. Left atrial (LA) size can also be measured on M-mode, although it is difficult to align the cursor through the body of the atrial chamber. 7
Hypertrophy of the myocardium is defined as diastolic wall thickness ≥ 6 mm, 8 and is often accompanied by increased left ventricular (LV) fractional shortening (FS). Left ventricular diameter ≥ 14 mm in end-systole is consistent with dilation and, in cases of systolic dysfunction, is also accompanied by reduced FS (≤ 28%). 9 However, a diagnosis of CM should never be based solely on these findings, since increased LV myocardial thickness can be seen in cats that are dehydrated and/or tachycardic (so-called pseudohypertrophy) 5 and FS can vary significantly in association with other conditions such as mitral valve insufficiency, hyperthyroidism, sympathetic stimulation and over-hydration.5,10–12 Furthermore, significant overestimation of myocardial thickness can result from erroneous positioning of the cursor across the papillary muscles (which can be very difficult to avoid when papillary muscles are prominent). Conversely, M-mode studies can fail to demonstrate regional hypertrophy, 11 and do not provide any information on abnormal blood flow, patchy fibrotic lesions or valvular insufficiency. Finally, assessment of right ventricular (RV) morphology and function is almost impossible on M-mode studies.
PART 1
In spite of these severe limitations, there is still a widespread tendency to concentrate on M-mode measurements and fail to notice other cardiac abnormalities that could be revealed by a more careful 2D examination, such as right-sided changes, valvular and pericardial abnormalities, or regional reduced hypokinesis.
Two-dimensional echocardiography
Two-dimensional echocardiography allows overall assessment of myocardial function and identification of various phenotypical expressions of myocardial disease (Fig 1). Diastolic measurements of the LV myocardium should be taken in four different wall segments in order to identify regional hyper-trophy. 11 Diagnosis of LV hypertrophy can be made when the hypertrophic segment (≥ 6 mm) occupies more than 50% of the LV myocardial area. 8

Phenotypical expressions of myocardial disease on 2D echocardiography. RA = right atrium, RV = right ventricle, LA = left atrium, LV = left ventricle, Ao = aorta, IVS = interventricular septum, FW = free wall
Papillary muscles can be measured on 2D echocardiography; their size is generally greater in cats with concentric myocardial hypertrophy. 13
Global biventricular function should always be assessed in 2D in order to identify regional dyssynchrony, hypokinesis or dyskinesis that could be related to a myocardial insult (ie, ischaemia and reparative fibrous infiltration). Paradoxical motion of the septum would suggest the presence of RV volume overload and/or pressure overload, 14 such as severe tricuspid insufficiency, pulmonary hypertension, atrial septal defect or arrhythmogenic right ventricular cardiomyopathy (ARVC).
Given the limitations of M-mode studies outlined above, LA size should be measured using 2D echocardiography. The LA diameter in normal cats is approximately 10 mm, both in short and long axis view, and, when indexed to the aortic diameter, should have a ratio < 1.5.5,7 However, volume depletion and overzealous fluid administration can lead to significant variations in LA size and hence potentially to misinterpretation of echocardiographic findings. 5 Increased LA size accompanied by an abnormal arching of the atrial septum towards the right atrium is suggestive of increased atrial pressure, which can lead to pulmonary venous congestion, and pulmonary oedema and/or pleural effusion. The presence of an echodense structure in the LA lumen or auricle can indicate a thrombus, which could predispose to aortic thromboembolism (ATE). Similarly, spontaneous echo contrast (‘smoke’) observed in the cardiac chambers indicates red blood cell aggregation and altered blood flow, 15 and may be considered a potential marker of LA thrombus or previous thromboembolism. 16
Two-dimensional echocardiography also allows the identification of RV enlargement, pericardial effusion and myocardial fibrotic lesions that can accompany different forms of CM.
Colour Doppler echocardiography
Colour Doppler allows the identification of flow turbulence and facilitates spectral interrogation. The presence of simultaneous turbulence in the left ventricular outflow tract (LVOT) and left atrium during early to mid-systole suggests dynamic LVOT obstruction and mitral regurgitation, which is consistent with SAM. Accurate colour Doppler settings (ie, pulse repetition frequency, gain, filters) are mandatory to avoid misleading artefacts.
Spectral Doppler echocardiography
Spectral Doppler interrogation of all four cardiac valves should always be performed to identify abnormalities that could mimic myocardial disease, such as mitral or tricuspid regurgitation, and aortic or pulmonic stenosis. Left dynamic outflow obstruction caused by SAM should be suspected whenever the aortic peak velocity exceeds the normal reference range, and the shape of the Doppler recording presents an abrupt acceleration in mid-systole, producing a concave, asymmetrically shaped waveform (see Part 1, Fig 3d). 17 Sometimes, SAM occurs only during stress or excitement and a simple method to detect the abnormality is to excite the cat by increasing the speaker volume during Doppler interrogation of the LVOT. A significant change in shape and peak velocity of the aortic wave would be consistent with dynamic LVOT obstruction.

Dorsoventral thoracic radiographs from a cat with myocardial disease. (a) Cardiomegaly and bi-atriaI enlargement (valentine-shaped heart) and patchy alveolar opacity (arrows) consistent with focal pulmonary oedema; (b) resolution of pulmonary oedema after intravenous administration of furosemide

A variety of histopathological lesions that can be observed in feline myocardial disease.
Pulsed-wave spectral Doppler echocardiography can be used to study mitral and pulmonary venous inflow patterns and isovolumetric relaxation time (IVRT), which provide a non-invasive assessment of impaired diastolic function in a variety of myocardial diseases (see box on page 186).18,19 An important limitation of studying mitral inflow is the fusion of the E and A waves at heart rates above 160 bpm, which makes it difficult to characterise the different inflow patterns. Recently, pulsed-wave Doppler has been used to measure LA appendix flow velocity, which appears to be decreased in cats with CM and can stratify patients at increased risk of spontaneous echo contrast and, possibly, thromboembolism. 20

Tissue Doppler imaging
Tissue Doppler imaging (TDI) is a technique that allows non-invasive quantification of regional myocardial function. 21 It provides a reliable evaluation of the systolic and diastolic myocardium and is independent of changes in preload in the diseased state. 22 Pulsed TDI shows lower early diastolic velocities, acceleration and deceleration in cats with HCM, especially along the longitudinal axis of the heart.22,23 Systolic impairment has also been documented by TDI in cats with HCM, irrespective of the presence of LVOT obstruction and congestive heart failure (CHF). 22 Colour TDI provides an estimation of the myocardial velocity gradient and mean myocardial velocity, and has been demonstrated to be a useful tool in the investigation of different myocardial diseases in humans. 24 Interestingly, diastolic impairment has been detected in Maine Coon cats before the occurrence of hypertrophy, both with pulsed TDI 25 and colour TDI, 26 although it is unclear whether it would be a sensitive enough screening test to detect genotypically affected cats with no hypertrophy.
Radial strain and strain rate
Radial strain and strain rate are novel techniques that provide non-invasive assessment of LV function and may have potential utility in the early detection of myocardial dysfunction in cats. 27
Further diagnostics
Thoracic radiography
Thoracic radiographs are invaluable for detecting cardiomegaly, pulmonary vein congestion and an interstitial/alveolar pattern consistent with CHR The distribution of alveolar infiltrate can be diffuse or patchy, in contrast to the more consistent dorsoventral location of cardiogenic pulmonary oedema observed in dogs (Fig 2). 28 Thoracic radiographs can also allow pleural effusion and ascites to be identified.
Cardiomegaly is not always present in myocardial diseases, especially when neither significant LV or LA enlargement nor pericardial effusion are present. The classic ‘valentine-shaped’ heart on dorsoventral (or ventrodorsal) views is caused by severe bi-atrial enlargement, which would be expected in the advanced stages of any feline myocardial disease, 29 and not only HCM as has been reported in the past.29,30
Cardiac magnetic resonance imaging
Cardiac magnetic resonance imaging (cMRI) is a relatively new diagnostic test that enables quantification of ventricular mass and ventricular function. In human medicine, cMRI is becoming extensively used in patients with specific myocardial disorders, such as ARVC, endocardial fibroelastosis and myocarditis. Cardiac MRI is also an important tool in the quantification of LV mass in patients with HCM, 31 and in the differentiation of infiltrative and inflammatory CM. 32 Recent studies have demonstrated that cMRI is an accurate method for identifying changes in LV mass in feline HCM but is not useful for identifying diastolic dysfunction in these patients. 6
The requirement for general anaesthesia, the long duration of scanning (approximately 1 h), ECG cardiac gating and high costs are important limitations hindering the widespread use of cMRI in feline cardiology. However, it is possible that new technologies will overcome these problems in the future.
Radionuclide imaging
In people, radionuclide ventriculography can be useful to identify ischaemic myocardial injury and can elucidate the nature of regional wall motion abnormalities. 32 Positron emission tomography (PET) and single photon emission computed tomography (SPECT) can provide combined molecular, functional and morphological image data of the cardiovascular system. 33 In veterinary cardiology, PET has been used to measure coronary flow reserve in healthy cats 34 and cats affected by HCM, 35 and revealed a lower flow in the latter group.
High costs and limited availability of this technology currently prohibit the routine use of PET scanning, although it may become more accessible and affordable in the future.
Electrocardiography
Cats with myocardial disease may present with a variety of electrocardiographic abnormalities, including morphological changes consistent with chamber enlargement, arrhythmias and conduction abnormalities, the most common being left anterior fascicular block (LAFB), which is observed in approximately 20–30% of cardiomyopathic cats.36,37. Although electrocardiography (ECG) can be unremarkable at presentation in many cats with CM, in the author's experience ventricular arrhythmias, including paroxysmal ventricular tachycardia, can be documented on 24 h ECG (Holter) recording in almost all cats with myocardial disease. This suggests that standard ECG is a relatively insensitive diagnostic test in cats with myocardial disease.
Histopathology
Post-mortem histopathology can identify a variety of myocardial lesions in cardiomyopathic cats. These include myofibre hypertrophy and disarray, focal to multifocal or extensive myocyte necrosis and degeneration, interstitial fibrosis, and fibrous or fibrofatty myocyte replacement. Major or intramural coronary arteries can show medial and intimal thickening associated with increased connective tissue elements. In some cases occlusive fibrinous thrombosis can be observed. Endomyocardial infiltrates of mononuclear cells, macrophages and occasional neutrophils (Fig 3) are often associated with symptomatic endomyocarditis in cats.17,38–42
Although some histological changes can be indicative of a particular form of CM, they are unlikely to be pathognomonic. Fibrous or fibrofatty myocyte replacement, for example, is a common sequela of the myocardial ischaemia and tissue necrosis that is frequently observed in all forms of cardiomyopathy.
Ante-mortem identification of infiltrative and inflammatory changes can be obtained by transvenous endomyocardial biopsy. However, use of this procedure is highly controversial due to the associated risks of RV perforation and malignant ventricular arrhythmias. Furthermore, patchy myocardial lesions may be missed by the random biopsy sampling.
Genetic tests
Genetic mutations responsible for HCM have been identified both in Maine Coons 43 and Ragdoll cats, 44 although not all Maine Coons or Ragdolls with HCM show this particular mutation. Furthermore, the mutation in the two breeds is located in different regions of the same gene and other mutations are likely to be identified in the future. Studies are in progress to identify similar mutations in other predisposed breeds, such as the Norwegian Forest Cat and Sphinx.
Genetic tests can identify individuals predisposed to a particular mutation and may be useful for screening programmes. However, the disease can still originate from a different mutation that has yet to be identified.
Prognosis
The median survival time of cats with HCM that survived the first 24 h after initial examination has been reported as 596–732 days (Table 2). Patients affected by ATE and concurrent myocardial disease carry a poorer prognosis (184 days). 29 Other forms of CM carry a less favourable prognosis still, with a reported median survival time of 132 days for RCM and 11 days for DCM. 37 Identification of a primary cause is a critical element influencing survival time since some forms of CM, such as taurine deficiency, hyperthyroidism or sustained tachycardia, can potentially be corrected, with subsequent reversal of remodelling and resolution of myocardial dysfunction.
Median survival times of cats with CM surviving the first 24 h after initial examination

HCM = hypertrophic cardiomyopathy, RCM = restrictive cardiomyopathy, DCM = dilated cardiomyopathy, ARVC = arrhythmogenic right ventricular cardiomyopathy, UCM = unclassified cardiomyopathy
In an attempt to identify risk factors and establish an accurate prognosis in cats with myocardial disease, many clinical features have been suggested as being significant.
In one study of cats with HCM, those with heart rates ≥ 200 bpm appeared to have a reduced survival compared with those with lower heart rates. 52 However, a later study reported no statistical association between heart rate and survival time, and whether or not tachycardia represents a negative prognostic factor remains controversial. 53
Clinical signs associated with CHF were found to be a negative prognostic factor in cats with HCM in two retrospective studies.52,53 In one of these studies, LA size, age, subjective evidence of RV enlargement and the performing of thoracocentesis were negatively associated with survival time, but only LA size and age were significant predictors in multivariate analyses. 53 In the same study, LA and LV diameter were also associated with a higher incidence of ATE, in contrast to the findings of a subsequent retrospective study of 127 cases of ATE. 54 However, in none of the above studies did the authors specify the method used to measure LA dimensions. It is well documented that the M-mode method of measuring the left atrium has inherent limitations and tends to produce different values when compared with 2D evaluations.7,55 For this reason, the association between LA dimension and risk of ATE remains controversial.
A study of coagulation markers (thrombin-antithrombin complex, D-dimers and fibrin degradation products) has shown that 45% of cats with HCM are in a hypercoagulable state. However, coagulation results are not correlated with LA size and an association between hypercoagulability and the risk of thrombosis has yet to be documented in cats with HCM. 56
A major limitation of retrospective studies to establish prognostic factors is the inherent variability of such studies in terms of, for example, the management of patients following diagnosis (different clinicians may choose different drugs and doses), and the severity of the cardiac disease at presentation and at euthanasia. Additionally, criteria for classification of myocardial disease change over time and are influenced by several subjective evaluations.
Biomarkers
Cardiac troponin-I (cTn-I) is a sensitive and specific marker of cardiac myocyte injury and its plasma concentration is increased in a variety of cardiac diseases, including HCM in cats.45,46 An elevated cTn-I level is not pathognomonic for HCM, but simply indicates ongoing myocardial damage, which may be present in any myocardial disease. However, this assay may serve as a useful adjunct to help determine diagnosis and/or prognosis. 32 The natriuretic peptides ANP (atrial natriuretic peptide) and BNP (brain natriuretic peptide) are found in higher concentrations in the heart of cats with HCM. 47 Similarly, their level is increased in the circulation of cats with cardiac disease.48–51 Natriuretic peptide measurement can be clinically useful as an initial screening test for cats with suspected cardiac disease. The N-terminal fragments of ANP and BNP, namely NT-proANP and NT-proBNP, are more stable, have a longer halflife and are therefore easier to analyse. In particular, NT-proBNP concentrations are positively correlated with LA size and pressure, 51 representing a useful tool for assessing cardiac disease severity and, potentially, prognosis.
The asymptomatic patient: A treatment dilemma
Whether or not an asymptomatic cat with CM should be treated is controversial. Anecdotal reports claim improvement in physical activity levels in asymptomatic cats with HCM treated with diltiazem or β-blockers. 28 In addition, a recent pilot study has demonstrated a possible reduction in myocardial damage in cats with compensated HCM following daily administration of atenolol, as suggested by a significant reduction in circulating cTn-I. 57 However, randomised placebo-controlled studies are still lacking and clinical benefits of diltiazem or β-blockers in asymptomatic cats have yet to be proven.
ACE inhibitors have also been advocated in cases of HCM, both in asymptomatic and symptomatic cats. 58 In that particular retrospective study, significant changes in cardiac dimensions were identified by echocardiography, together with an improvement in life expectancy, after administration of enalapril. Unfortunately, recent prospective, controlled studies have failed to demonstrate significant effects of ACE inhibitors (benazepril and ramipril) in cats with subclinical HCM.59,60 The potassium-sparing agent spironolactone has shown anti-remodelling properties in human patients with asymptomatic cardiac disease, as well as in patients with mild CHF (NYHA class I and II). 61 It is not unreasonable to hypothesise that spironolactone may produce a similar effect in cats with asymptomatic CM, although controlled clinical studies would be necessary to confirm this. Regrettably, a recent study failed to demonstrate improvement of diastolic function and LV mass in Maine Coon cats with HCM treated with spironolactone for 4 months. Furthermore, in that study, one-third of the treated cats developed severe ulcerative facial dermatitis. 61 However, to the best of the author's knowledge, this side effect has never been reported in Europe.
Cats with asymptomatic forms of CM but with echocardiographic evidence of intracavitary thrombi, spontaneous echo contrast or severe LA dilation may benefit from antithrombotic prophylaxis to reduce the risk of ATE. This could theoretically be achieved by administering low-dose aspirin, 54 clopidogrel 62 or a combination of the two drugs, although such combination has not shown clinical benefit in humans. 63 However, results of controlled prospective studies are necessary to prove the prophylactic efficacy of these treatments.
Clinical management of the symptomatic patient
Ideally, treatment of feline CM should be aimed at resolving all the underlying pathogenetic mechanisms of the disease, such as diastolic and systolic dysfunction, dynamic outflow obstruction, ischaemia, arrhythmias, neurohormonal activation and hypercoagulability status. In reality, with the exception of taurine supplementation in cats with taurine-deficient DCM, such ideal treatment is not available and no drug at present has convincingly demonstrated its ability to improve survival and/or quality of life in cats with myocardial disease.
Diuretics and other treatments for CHF
In practice, the patient presenting with clinical or radiographic signs of CHF should be treated accordingly. In cases of acute respiratory distress, stress should be minimised and cage rest and oxygen supplementation should be instituted promptly. Pulmonary oedema is generally controlled by intravenous administration of furosemide q 4–6 h until a normal respiratory rate is achieved. Dyspnoea secondary to pleural effusion can be successfully managed by thoracocentesis. Sedative medication with acepromazine and butorphanol may help to alleviate the respiratory distress. In extreme cases, where the patient does not respond to acute diuresis, airway suctioning and mechanical ventilation can be considered. 28 Once pulmonary oedema is sufficiently controlled, furosemide can be given orally at the lowest effective dose.
A similar approach should be taken in chronic and non-acute cases in order to reduce the negative side effects of diuresis, including reduced ventricular preload, hypotension, prerenal azotaemia and hypokalaemia, which can predispose to anorexia and severe ventricular arrhythmias. The risk of hypokalaemia could be reduced by concomitant administration of a potassium-sparing agent, such as spironolactone, although sufficient data of clinical efficacy in cats are not available.
Calcium channel blockers
Calcium channel blockers have been advocated as an effective treatment for symptomatic feline HCM for many years. 64 Diltiazem has a lesser effect on the systemic vasculature and inotropic state than verapamil, and it is generally the calcium channel blocker of choice for the treatment of feline HCM. The claimed beneficial therapeutic effects of diltiazem are due to its positive lusitropic and coronary vasodilating properties and include increased LV filling, reduced heart rate, increased venous oxygen tension, improved echocardiographic parameters and resolved radiographic abnormalities. 64 However, due to its pharmacokinetics, diltiazem needs to be administered every 8 h, and many clients struggle to comply with this dosing regimen, administering the drug only once or twice daily. Unfortunately, the administration of the extended release formulation is accompanied by significant side effects. 65
Drugs commonly used in the clinical management of feline myocardial disease
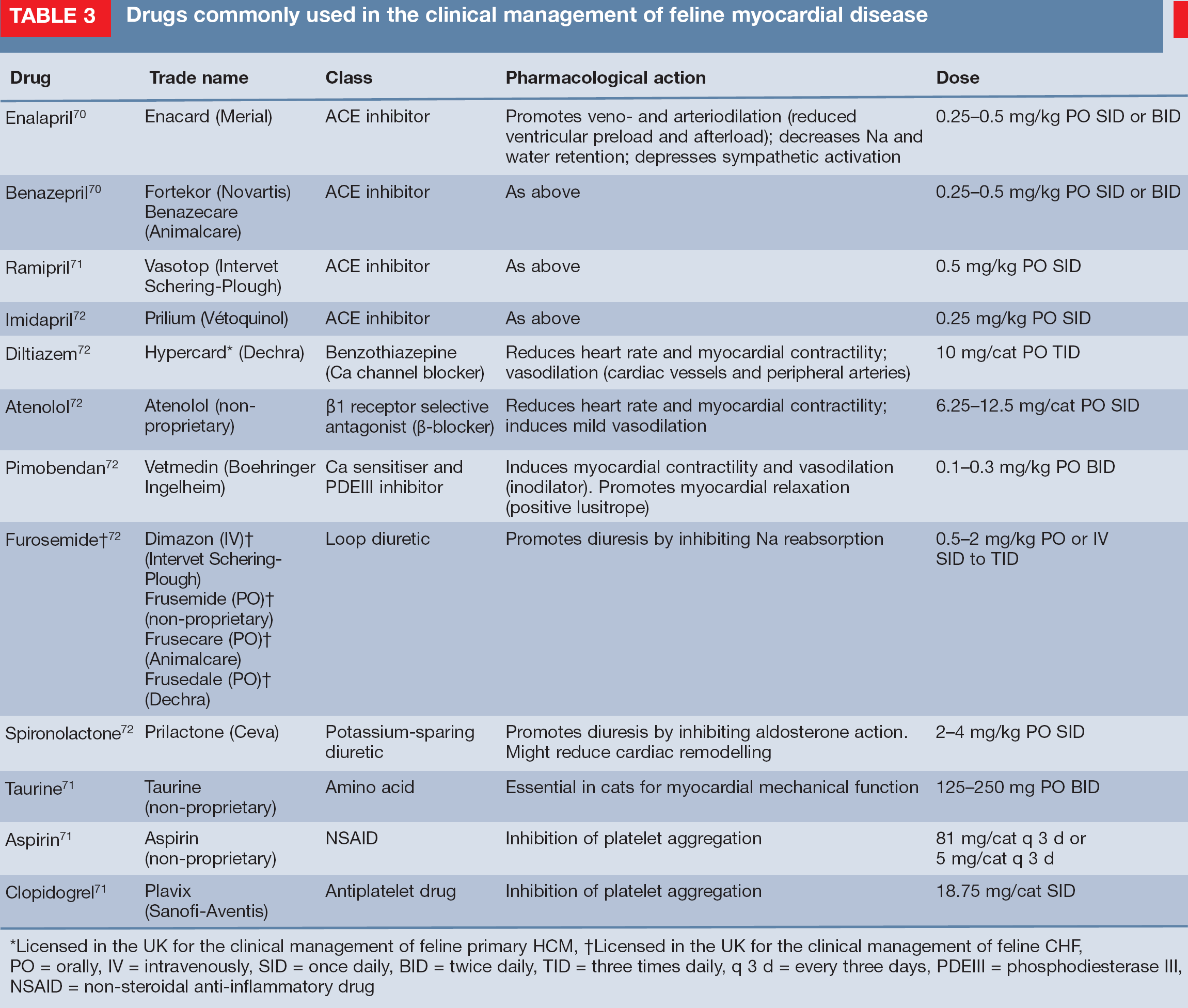
Licensed in the UK for the clinical management of feline primary HCM
Licensed in the UK for the clinical management of feline CHF, PO = orally, IV = intravenously, SID = once daily, BID = twice daily, TID = three times daily, q 3 d = every three days, PDEIII = phosphodiesterase III, NSAID = non-steroidal anti-inflammatory drug
Beta-blockers
Beta-blockers have also been suggested as a useful treatment for feline CM since they can provide heart rate and arrhythmia control, relieve LVOT obstruction and lessen myocardial oxygen demand. 28 Atenolol, a selective β-1 agonist, is generally preferred over other β-blockers (ie, propranolol) because it reduces the risk of bronchospasm. Also, it can be administered only once or twice daily, versus the recommended three times daily administrations of propranolol.
Interim results of a prospective, doubleblind, multicentre, controlled study comparing the clinical efficacy of atenolol, diltiazem and enalapril in cats with diastolic heart failure revealed that cats receiving atenolol and furosemide survived for a significantly shorter time than cats treated with furosemide alone. 66 Similarly, survival rates for patients receiving diltiazem were poorer than for cats on furosemide alone, while cats in the enalapril group did as well or better than the placebo group. However, these differences were not found to be statistically significant. 66 Therefore, a prudent approach should be taken when recommending any of these drugs, in combination with furosemide, for long-term treatment of feline CM.
Taurine supplementation
Taurine-deficient DCM cases can be successfully treated with taurine supplementation, in addition to supportive therapy to control the clinical signs of CHF. Echocardiographic evidence of improved systolic function is generally seen within 6 weeks of supplementation. 67
Inotropic drugs
Cats with echocardiographic evidence of systolic dysfunction not related to taurine deficiency may benefit from positive inotropic drugs.
Dobutamine infusion can be considered in severe cases, especially those with accompanying severe hypotension. Less severe cases may benefit from an oral positive inotropic medication. Digoxin, for example, has been used for many years in cats, but careful dosing and periodic plasma measurements are necessary to reduce the risk of toxicity. Pimobendan can also be considered in all cases of feline CM that are accompanied by systolic dysfunction. Although this drug is not licensed for use in cats, its administration is well tolerated and leads to significant improvement in the demeanour and appetite of treated cats in association with resolution of clinical signs. 68 In addition to its positive inotropic properties, pimobendan has a positive lusitropic effect, which may also have benefits in other forms of feline myocardial dis-ease. 69 However, further studies are necessary to confirm this hypothesis.
Other therapies
Whenever a primary cause of myocardial disease is suspected or recognised, every attempt should be made to manage it directly. For example, sustained tachycardia can induce systolic dysfunction (tachycardiomyopathy) but the myocardial lesions can be reversible if the arrhythmia is successfully controlled. Similarly, corticosteroids may control the inflammatory changes in cases of myocarditis and, where the cause is bacterial (eg,
KEY POINTS
Case notes
History Tommy's previous history was unremarkable. He was an indoor/outdoor cat, routinely vaccinated, wormed, treated for fleas and fed a commercial feline maintenance diet.
General impression At presentation, Tommy was in good bodily condition (4.3 kg) and fully responsive. However, he appeared rather uncomfortable and had an anxious expression. He exhibited a rapid respiratory rate (66 breaths per minute), with shallow, open-mouth breathing, accompanied by severe abdominal effort and dilated nostrils (pictured right).

Physical examination and laboratory findings
Thoracic auscultation revealed a fast heart rate (> 200 bpm) with an audible irregular gallop rhythm. Crackles and wheezes could be heard throughout the lung fields. Systolic blood pressure measurement revealed a moderate hypotension (95 mmHg). Routine biochemistry and thyroid profile
Clinical management Sedatives (acepromazine and buprenorphine) and oxygen supplementation
Further diagnostic tests Once stabilised and more comfortable, imaging was undertaken. Echocardiography (see right) showed severe bi-atrial dilation with normal left ventricular dimensions; a fibrotic lesion was observed as a band of echogenic tissue bridging the ventricular lumen (arrow). Spectral Doppler interrogation of the mitral valve inflow showed increased E wave velocity, shortened DT and reduced A wave velocity. Thoracic radiography (above right) revealed cardiomegaly, and venous engorgement and an alveolar pattern indicative of pulmonary oedema.

WHAT IS YOUR ASSESSMENT?
The presence of bi-atrial enlargement is indicative of dilated cardiomyopathy (DGM).
Normal left ventricular (LV) dimensions rule out an underlying cardiomyopathy.
Normal LV dimensions accompanied by severe atrial dilation and fibrotic lesions are highly suggestive of a restrictive form of cardiomyopathy (RGM).
The spectral Doppler interrogation of the mitral valve inflow is indicative of hypertrophic cardiomyopathy (HCM).
Oral furosemide, ACE inhibitors and diltiazem.
Oral furosemide, ACE inhibitors and atenolol.
Oral furosemide, atenolol and diltiazem.
Oxygen therapy, intravenous furosemide and potassium supplementation.
Answers 1(c) 2(d)
Footnotes
Acknowledgements
I wish to thank Dr Anibal G. Armién, University of Minnesota, for sharing with me the beautiful pictures of his pathological specimens. Thank you also to Dr Kit Sturgess for his useful comments and criticisms, which improved the content of this article.
