Abstract
A 6-year-old female neutered Burmese cat received a 10 times overdose (5 mg/m2) of vincristine, administered in error. Supportive therapy, including administration of calcium folinate, was instigated within 8 h. Despite treatment, the patient exhibited deterioration in renal and respiratory function and died 72 h after overdose. Necropsy was performed within 24 h of death. Gross examination revealed pulmonary oedema and a pale brown liver with a prominent lobular pattern. Histological examination revealed marked apoptosis and necrosis of the bone marrow myeloid series, and mild to moderate apoptosis and necrosis of the erythroid and megakaryocyte series. Multifocal necrosis of the renal tubules, hepatocytes, and small intestinal crypt epithelium was also observed. Use of calcium folinate as a rescue therapy following vincristine overdose in humans has been previously documented. If treatment is to be successful in cases of vincristine overdose in cats, then a more complete understanding of the pathogenesis of vincristine toxicity in companion animal species is required.
In the UK, vincristine is the second most widely used chemotherapeutic agent employed to treat dogs and cats with neoplasia. 1 A vinca alkaloid, it is considered to be cell-cycle specific. Vincristine inhibits tubulin polymerisation, preventing mitotic spindle formation and thus mitosis. 2
Vincristine overdose has been previously reported in humans, and may occur following errors in calculating and recording dosages. 3 Here we describe, for the first time to the authors' knowledge, the pathological lesions associated with accidental overdose of vincristine in a veterinary species.
A 6-year-old female neutered Burmese cat was being treated with the low dose COP chemotherapy protocol 4 following diagnosis of gastric lymphoma. Complete clinical remission was promptly attained. The patient was not shown to be neutropenic at any time and did not exhibit evidence of renal dysfunction. At the eighth weekly administration of intravenous vincristine an erroneous 10 times overdose (5 mg/m2) was administered. The patient was not scheduled to receive a concurrent dose of cyclophosphamide.
Within 8 h of overdose, intravenous crystalloid therapy was introduced (5 ml/kg/h) (Aquapharm 1; Animalcare) together with intravenous furosemide (2 mg/kg tid) (Dimazon; Intervet UK), calcium folinate (2 mg/kg q3 h for 12 h then q6 h for 48 h) (Lederfolin; Wyeth Pharmaceuticals), and subcutaneous metoclopramide (0.5 mg/kg tid) (Metoclopramide; Hameln Pharmaceuticals). Signs consistent with nausea were seen from 14 h after overdose; folinic acid injection was associated with ptyalism and sometimes induced unproductive vomiting. After 24 h the patient was subdued and hypothermic (36.8°C). Respiratory rate was increased and respiratory crackles were auscultable so the fluid administration rate was adjusted. Plasma potassium and urea concentrations were monitored (Table 1) and potassium supplementation (potassium chloride; Antigen Pharmaceuticals) was administered with the intravenous crystalloids. After 48 h the patient no longer appeared nauseous so metoclopramide therapy was withdrawn. Subcutaneous dexamethasone (0.1 mg/kg OD) (Dexadreson; Intervet UK) and intravenous ranitidine (2.5 mg/kg bid) (Zantac; Glaxosmithkline UK) were administered. Indices of renal function were seen to deteriorate (Table 1); increased intravenous fluid administration caused respiratory distress and was, therefore, limited. The patient was not pyrexic at any point. The patient died 72 h after overdose.
Results of serial blood evaluations made using a portable blood analyser (iSTAT portable clinical analyser, Heska Corporation) during the 72 h following recognition of vincristine overdose to a cat

NP=not performed.
At necropsy, the lungs were found be homogenous and aerated. The pleural surfaces glistened, and translucent fluid discharged from the cut pulmonary surfaces, indicating mild oedema. The liver was pale brown, with a prominent lobular pattern. All other organ systems were macroscopically unremarkable.
For histological investigation, tissue samples from all major organs were fixed in 10% formalin and submitted for routine paraffin embedding; 3 μm sections were mounted on glass slides and stained with haematoxylin and eosin.
Histological evaluation of the bone marrow revealed that approximately 95% of the bone marrow area comprised erythrocytes and mature adipocytes. Only small numbers of erythroid and megakaryocyte precursors, and occasional myeloid precursors, were present, along with moderate to large numbers of cells with pyknotic or karyorrhectic nuclei present diffusely (Fig 1).
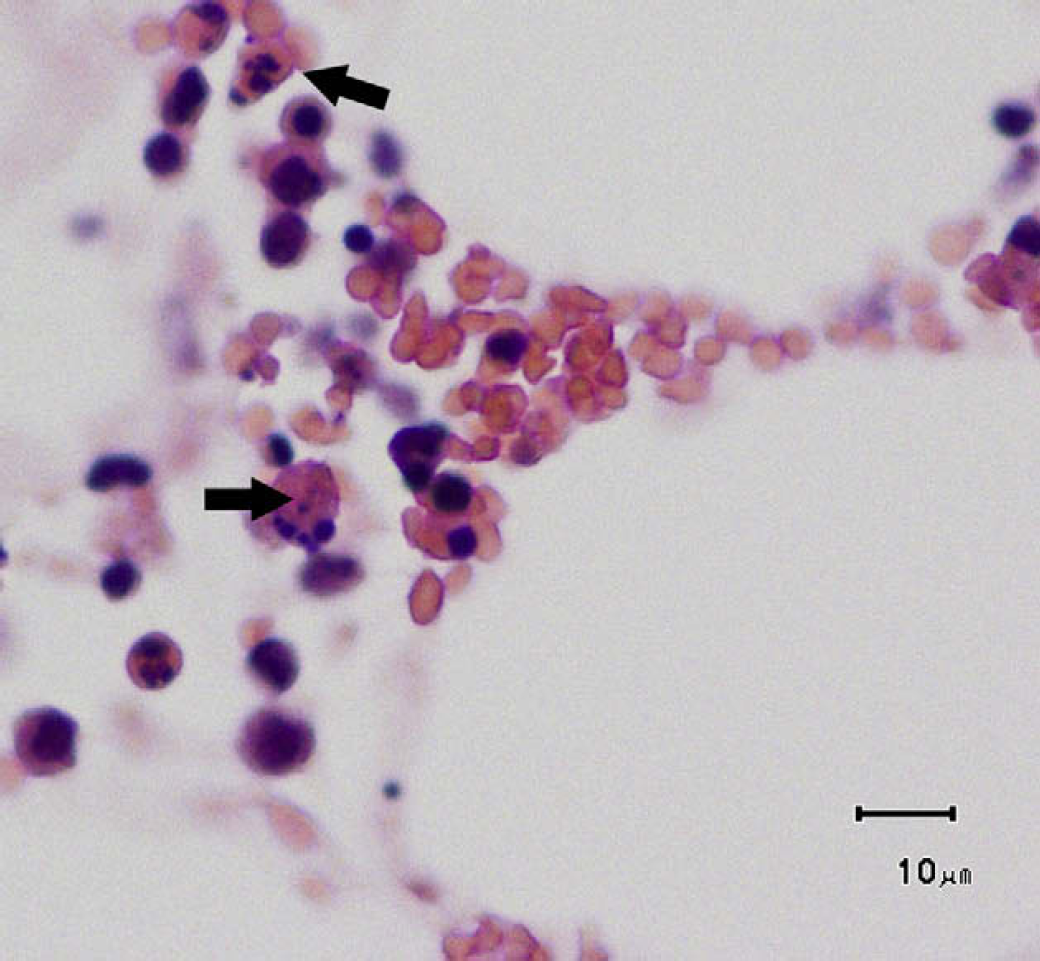
Photomicrograph of bone marrow. Pyknotic and karyorrhectic cellular debris (arrows) are present throughout. Haematoxylin and eosin stain, bar=10 μm.
Within the kidney, necrotic tubular epithelial cells were present (Fig 2), and multifocal cortical tubular epithelial cells exhibited mitoses, including atypical examples.

Photomicrograph of renal cortex. Necrotic tubular epithelial cells (arrowheads) are present within the renal tubules. Haematoxylin and eosin stain, bar=10 μm.
Randomly scattered throughout the liver were moderate numbers of necrotic hepatocytes (Fig 3). Mild hepatocyte anisocytosis and occasional binucleate hepatocytes were observed. Hepatocytes exhibited mitoses at a rate of one per one to two high power fields. There was diffuse Ito cell hyperplasia and hypertrophy. Intracytoplasmic accumulation of fine, golden granular pigment was present multifocally in centrilobular (zone 3) hepatocytes. The pigment stained positively with periodic acid-Schiff stain. Examinations using Perl's stain, for iron containing pigment, and Fouchet's stain, for bile pigment, proved negative. The histological appearance and staining properties of the pigment were consistent with lipofuscin.

Photomicrograph of hepatic parenchyma. Necrotic hepatocytes (arrows) are present multifocally. Haematoxylin and eosin stain; bar=10 μm.
The spleen exhibited multifocal, mild lymphocytic depletion. The white pulp appeared relatively inconspicuous and the collagenous trabeculae appeared more prominent than expected.
Within the small intestinal crypts there were multifocal cells with pyknotic and karyorrhectic nuclei, indicating epithelial cell necrosis. Moderate numbers of extravasated erythrocytes were present within the intestinal lumen. There was no evidence of gastrointestinal lymphoma.
The cause of death of the patient was considered to be a combination of acute renal failure and pulmonary oedema.
Enquiries of the UK Veterinary Medicine Directorate's Suspected Adverse Reaction Surveillance Scheme did not reveal any previous reports of vincristine overdose in cats (D. Bird, personal communication, 2008). As far as can be ascertained, this is the first report documenting the use of calcium folinate as a therapeutic agent following vincristine overdose in a cat, and the first report to describe the pathological lesions seen following vincristine overdose in a companion animal.
Calcium folinate has been previously administered to human patients following vincristine overdose. 5,6 Folinic acid rescue is used to reduce methotrexate toxicity as folinic acid partially overcomes the methotrexate-induced inhibition of dihydrofolate reductase and thymidylate synthetase in DNA synthesis. 6 However, the rationale behind the use of folinic acid in cases of vincristine toxicity is less clear, and indeed the value of this agent as a rescue therapy has been disputed in vincristine toxicity trials in mice. 7
The efficacy of folinic acid treatment in cases of vincristine overdose in humans is difficult to elucidate from individual case reports and small case series where confounding variables may be present. 5,6
It is not possible to ascertain whether the administration of calcium folinate ameliorated the clinical signs in this patient. Indeed, the possibility of direct adverse effects of folinic acid or interaction between calcium folinate and vincristine cannot be completely discounted, especially in view of the perceived clinical association between folinic acid administration and ptyalism and unproductive vomiting. Human reports have not documented adverse interactions between calcium folinate and vincristine. 5,6 The present report highlights the necessity for further evaluation of the use of calcium folinate as a therapeutic agent following vincristine overdose in cats.
The present case illustrates the challenges associated with managing a patient which has received an overdose of vincristine. As 10–21% of administered vinca alkaloid doses have been shown to undergo renal elimination, 8 intravenous crystalloid therapy was administered to enhance renal excretion of the vincristine. Development of pulmonary oedema either as an adverse drug reaction 9 or a cytokine mediated inflammatory response 10 was also a concern, hence the administration of furosemide and dexamethasone.
A potential limitation of this report is that no haematological data was obtained following the overdose. Haematological parameters were normal on the morning prior to the overdose, and it was felt that neutropenia would not have had time to develop in the subsequent 72 h treatment period. Indeed in human reports, leukopenia manifests most usually from the fifth day following overdose, although it can be seen as early as the third day 6 with a nadir recorded at 10 days. 5 Histologically, there was no evidence of septicaemia in this patient, consistent with the view that the patient was unlikely to have been neutropenic at the time of death.
In this case, there was histological evidence of diffuse, marked apoptosis and necrosis of the bone marrow myeloid series; diffuse, mild to moderate apoptosis and necrosis of the erythroid and megakaryocyte series; multifocal acute renal tubular necrosis; multifocal hepatic necrosis; and diffuse, moderate necrosis of the small intestinal crypt epithelium. The perturbation in bone marrow cellular composition and the gastrointestinal signs are well-documented effects of vincristine overdose in humans. 11,12 The lesions within populations of dividing cells are consistent with the mode of action of vincristine. 13
It has been suggested that non-cardiogenic pulmonary oedema, sometimes seen following administration of anti-cancer agents in humans, may be due to direct damage to alveolar type 1 pneumocytes or pulmonary vascular endothelial cells, eliciting a cytokine mediated inflammatory response. 10 However, in the present case there was no histological evidence of damage to either type 1 pneumocytes, or pulmonary vascular endothelial cells. It is most likely that the pulmonary oedema noted in this case reflects severe renal compromise and fluid administration. This is in contrast to the reported manifestation of pulmonary oedema in a cat as a suspected adverse reaction to vincristine. 9 In that case, severe pulmonary oedema developed within hours of administration of standard vincristine doses.
This case report documents pathological lesions following vincristine toxicity in a cat. With the now widespread usage of chemotherapeutic agents in veterinary practice, further cases of accidental cytotoxic drug overdose are likely. If effective treatment of such cases is to be attempted, further characterisation of the pathogenesis of vincristine overdose in cats is required. Documentation of cases of accidental overdose of chemotherapeutic agents is critical to facilitate an evidence-based approach to treatment of these patients.
