Abstract
Absorbed thyroid dose and effective half-life were determined in 46 hyperthyroid cats after treatment with a low dose (mean 111 MBq) of radioiodine intravenously. Thirteen of these cats had received iohexol for glomerular filtration rate (GFR) measurement within 24 h before treatment with radioiodine in view of another ongoing study at our institution. Pre-therapy values were obtained for total thyroxine (TT4) and for the thyroid to salivary gland ratio with sodium pertechnetate γ-camera imaging. All cats underwent post-therapy scans at 24, 48 and 120 h for evaluation of radioactive iodine uptake (RAIU) and the effective half-life of radioiodine. The absorbed dose was calculated from the cumulative activity with Olinda software. Both groups were comparable in age, TT4 and the ratio of thyroid activity to salivary gland activity. Statistical analysis revealed a significant decreased absorbed dose in the thyroid in the iohexol group. This decreased uptake was not accompanied by an decreased effective half-life of the radioiodine. The variation of inter-individual RAIU decreased in this group and more homogenous absorbed doses were obtained. No significant difference in outcome could be demonstrated. However, a tendency towards a higher number of residual hyperthyroidism in the iohexol group was noted (15 versus 6% in control group). This study demonstrates that iohexol interferes with the uptake of radioiodine in the hyperthyroid cat but does not provoke increased turnover. In this study, albeit including a small number of cats, outcome did not seem to be significantly affected.
The outcome of radioiodine treatment depends mainly on the absorbed energy dose in the diseased thyroid tissue. The resulting target dose depends on the amount of radioiodine trapped (radioactive iodine uptake or RAIU) and on its effective half-life in the thyroid.
Various agents have been reported to interfere with thyroid uptake of radioiodine. The most important agents are antithyroid medication and external iodide administration (Childs et al 1950, Goldsmith et al 1958, Moka et al 2002).
Among other possible drug compounds, iodide contrast agents may interfere with RAIU (Nygaard et al 1998, Yu and Shaw 2003). In an ongoing research study at our institution, several glomerular filtration rate (GFR) tracers were compared in a group of hyperthyroid cats and for practical reasons, immediately prior to treatment with radioiodine-131 (van Hoek et al 2007). Among other markers, iohexol, an iodide-containing agent, was used in this protocol.
The aim of this retrospective study was, therefore, to investigate whether single iohexol administration for GFR measurements interfered with radiodine therapy in hyperthyroid cats at treatment dosages currently used at our institution. To reach this goal the absorbed dose and effective half-life of radioiodine were calculated and the final outcome was determined.
Material and methods
In the period 2005–2006, 74 privately owned cats were presented and treated for hyperthyroidism at the Division of Veterinary Nuclear Medicine, Medical Imaging Department, Faculty of Veterinary Medicine, Belgium. Twenty-eight cases were excluded because incomplete information was available. Forty-six cats with raised TT4 and clinical signs of hyperthyroidism were included in this study. All cats underwent a pre-therapy planar static scan with sodium pertechnetate at our hospital. Regions of interest were drawn over the affected thyroid lobes and the salivary glands to measure radioactivity in these regions. The thyroid to salivary gland ratio was calculated and, based on previous studies, a ratio of more than one was considered pathological (Daniel et al 2002, Broome 2006) (Fig 1). Antithyroid medication was discontinued at least 1 week before radioiodine treatment.

Static pertechnetate images are acquired with the cat in ventral recumbency, neck against the camera. Regions of interests are drawn over the affected thyroid lobe (large arrow) and the salivary gland (small arrow) and a ratio is calculated from the activity measured. Note the lack of visualisation of the contralateral thyroid lobe due to suppression.
Thirteen hyperthyroid cats underwent a GFR study with iohexol prior to treatment with radioiodine (iohexol group). The study design was not randomised because not all owners opted for inclusion of their pet in the iohexol protocol as this protocol required multiple blood samples. Owners declined out of sentimental or practical reasons (handling difficulties of cats). All owners of the cats included in the GFR study signed an informed consent. All owners in both groups received information both orally as well as in the form of an informative brochure, regarding the diagnostic and therapeutic protocols followed. They were informed about the post-therapy scans as these are part of the routine procedure. The owners also received information on how to handle cats and litter when returning home.
The cats in the iohexol group received a dose of 65 mgI/kg IV (Omnipaque 300, 300 mgI/ml, Nycomed Imaging AS, Oslo, Norway) 24 h prior to treatment with 131I. All animals received radioiodine intravenously depending on TT4 and ratio thyroid/salivary gland. RAIU was measured post-therapy at 24, 48 and 120 h according to the standard operating protocol used at our hospital, with γ-camera imaging. Acquisition time was set at 5 s making sedation or anaesthesia unnecessary. A syringe with a known reference activity was measured on the γ-camera at the same occasion, using the same acquisition parameters. Regions of interest were drawn over the thyroid region, background and the reference activity for all time points. Thyroid volume was estimated on the pre-therapy pertechnetate images with dedicated software from Nuclear Diagnostics (Sweden). The effective half-life was determined from the single exponential fit through the thyroid activity points. Taking thyroid volume into account and using the Olinda software, a factor was calculated converting the cumulative activity to absorbed dose (Stabin et al 2005).
The outcome of the treatment was documented and based on TT4 results within the reference range at 6 months. General condition (weight, hair coat, clinical signs related to other non-thyroid disease and whether treatment was instituted for hypothyroidism) and attitude of the cats were also registered.
Statistics
Post-hoc group comparison was used to investigate whether the two groups were different on several important items which have been reported in literature to influence radioiodine uptake (Turrel et al 1984, Peterson and Becker 1995, Berg et al 1996, Nieckarz and Daniel 2001, Moka et al 2002). A Mann Whitney test was used to compare age, weight, pre-therapy TT4 level, time interval of discontinuation of antithyroid medication, the ratio of thyroid to salivary gland on the pre-therapy pertechnetate scans, mean injected activity of 131-iodine, injected 131I activity/bodyweight, the effective half-life of radioiodine and the absorbed dose between both groups. A χ2 test was used to determine laterality (unilateral versus bilateral involvement), pre-therapy with antithyroid drugs and outcome in both groups.
All analyses were undertaken using SPSS software (release 12). A level of α=0.05 was adopted to indicate statistical significance.
Results
Thirteen hyperthyroid cats underwent a GFR study with iohexol (iohexol group). Thirty-three animals were grouped as control animals. All animals received minimal 81 MBq (2.2 mCi) and maximal 148 MBq (4 mCi) radioiodine intravenously. The cats from the iohexol group received 1.65–4.5 ml iohexol, based on weight (range 2.8–6.6 kg). Further details on pre-therapy and therapeutic data of both groups are presented in Table 1. In the iohexol group, the mean estimated absorbed thyroid dose was 53±10 Gy compared to the 131I alone group with an absorbed dose of 89±42 Gy. This difference was statistically significant (P<0.01). Variability of thyroid uptake was higher in the 131I alone group compared to the iohexol pre-treated group (Fig 2).
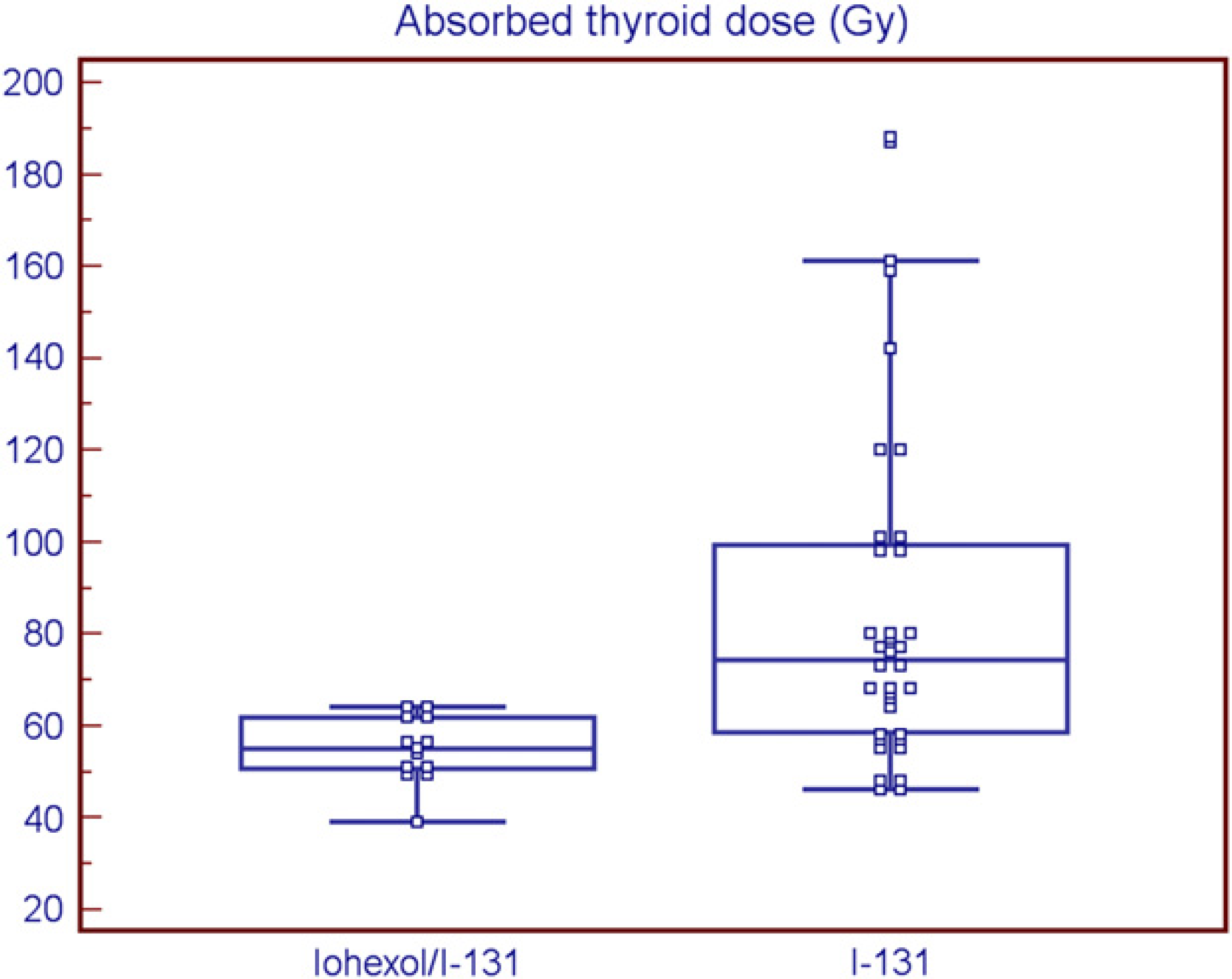
Absorbed thyroid dose in the iohexol/131I and the 131I groups calculated with Olinda software. Note the decreased variability in the iohexol/131I group.
Pre-therapy information and therapeutic data from both groups

The effective half-life of 131I was comparable between both groups (iohexol group: 2.2 d (1.6 d); non-iohexol group: 2.3 d (2 d) (P=0.89)).
Figure 3 demonstrates the outcome of treatment in both groups based on the TT4 values. A higher number of cats remained hyperthyroid in the iohexol group but significance was not reached (χ2: 0.221; P=0.638). We could a priori not randomise the cats in the two treatment arms of the study and, therefore, selection bias could not be ruled out. However, post-hoc analysis of potential factors that could influence the treatment outcome showed no significant difference between the two groups.
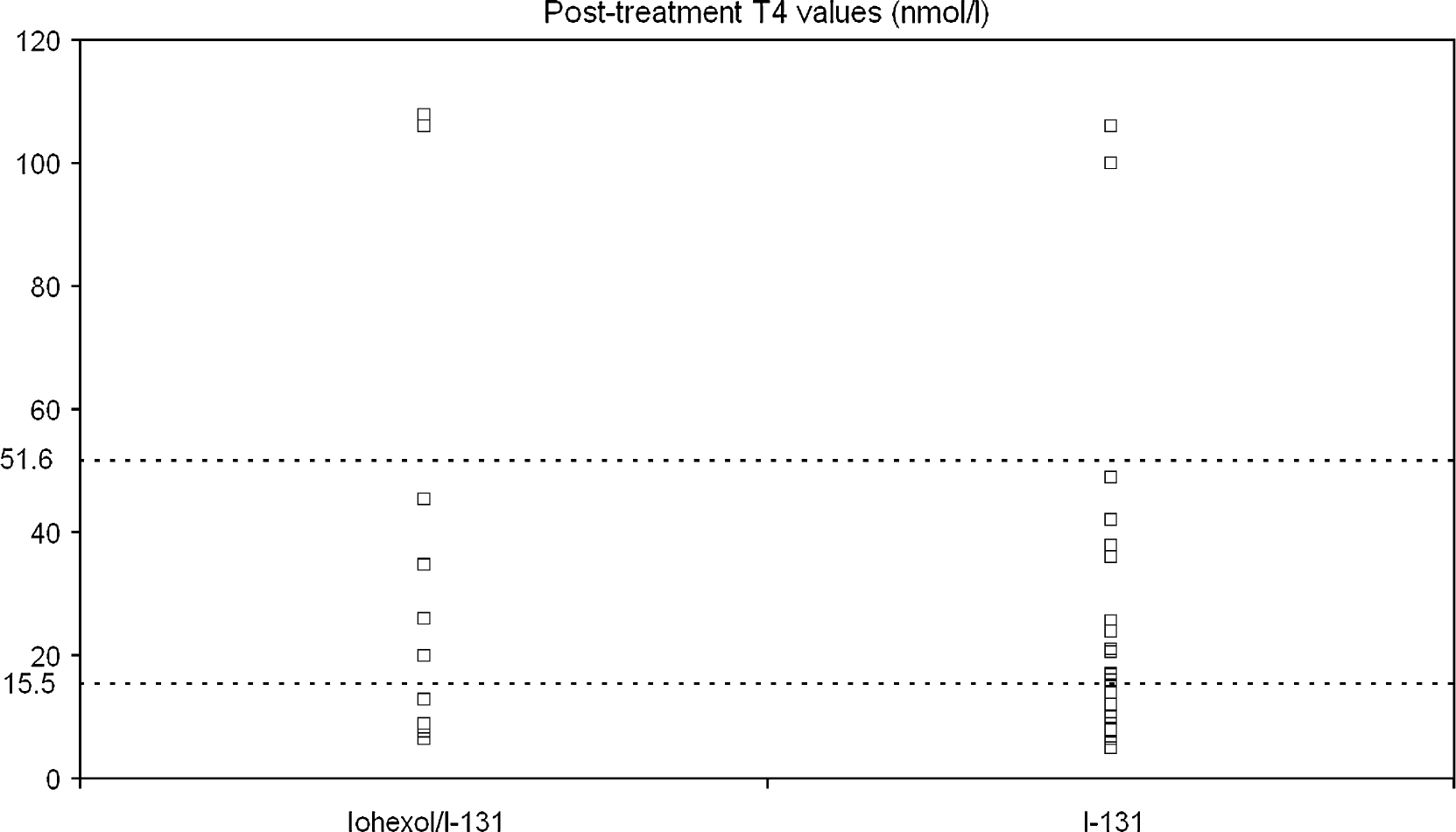
Therapy outcome based on TT4 values obtained in both groups 6 months after radioiodine treatment. The normal range of TT4 expressed in nmol/l is 15.5–51.6. Below normal limits were found in 15% of the iohexol group and in 6% the radioiodine treatment only group. Euthyroidism was seen in, respectively, 31% (iohexol group) and 39% and higher than normal TT4 values were found in 54% (iohexol group) and 55% in the control group.
Twenty-one cats had received antithyroid medication, six in the iohexol group and 15 in the control group. The difference was not significant. The withdrawal period did not differ significantly between both groups (range 14–35 d). In seven cats of the iohexol group, disease was limited to one thyroid lobe versus 19 in the control group. The difference in occurrence of bilateral versus unilateral disease was not significant.
Discussion
In this study we compared the absorbed therapeutic doses in hyperthyroid cats with and without prior use of the iodine-containing contrast agent, iohexol, and we evaluated therapy outcome in both groups based on TT4 levels. As the therapeutic effect of radioiodine largely depends on the absorbed dose, variations can be expected when uptake of radioiodine is hampered or when its turnover is increased (decreased effective half-life). In this study we found a significant decreased absorbed dose combined with less inter-individual variation, after iohexol administration without apparent effect on the effective half-life of radioiodine in the thyroid. It is generally accepted that iodine-containing contrast agents hamper the uptake of tracer doses of radioactive iodine in the thyroid due to release of iodide from these compounds or the presence of free iodide contamination products (Grayson 1960, Laurie et al 1992). Due to presence of this free iodide, RAIU may be inhibited as a result of competition for the iodine symporter and/or may be associated with autoregulatory phenomena in association with acute increased iodide supplementation leading to rejection of the iodide from the thyroid with increased kidney excretion (Ingbar 1972). The extent and duration of interference of RAIU in humans depend on the amount of free iodide, on the contrast agent involved, on time interval after contrast administration and on thyroid function (Childs et al 1950, Sternthal et al 1980, Yu and Shaw 2003). A study in normal and hyperthyroid human patients demonstrated a temporary reduction of radioiodine uptake after iohexol with a range from 43.6 to 80% of the initial RAIU depending on thyroid function (Nygaard et al 1998).
A decreased variation in the absorbed dose in the iohexol group compared to the non-iohexol group of cats was also found. Bacher et al (2002) reported a similar decrease in variation in RAIU in human patients premedicated with cold iodide for thyroid blocking purposes during treatment with 131I-lipiodol (Bacher et al 2002). This decreased variation may be attributable to individual physiological fluctuations in the thyroidal iodide trap and, consequently, differences in the amount of iodide accumulated are less after external iodide supplementation. The outcome of radioiodine treatment is predominantly dependent on its uptake and energy absorbed in the diseased thyroid tissue. In human patients with hyperthyroidism, high variation in radioiodine uptake is present (<10 to >80%) resulting in a wide variety of absorbed energy doses when a fixed dose regime is used (Moka et al 2002). Similar findings in the cat were reported by Sjollema et al (1989) who found a low RAIU that was within narrow limits in euthyroid cats compared to the high variation (16.2–87.7%) in uptake in hyperthyroid cats (Sjollema et al 1989). Therefore, it can be hypothesised that the addition of free iodide may result in less individual variation in absorbed radioiodine and may allow better prediction of the individual absorbed dose when the fixed dose regime is applied.
In this study no apparent effect was seen on the effective half-life (wash-out) of radioiodine. This last finding pleads against an iodide induced increased turnover of radiodiodine from the thyroid. Childs et al (1950) demonstrated in hyperthyroid human patients that in the higher dose ranges of administered inorganic iodide (1–10 mg) the competition for the iodide pump precluded further iodide trapping with a fast wash-out of radioiodine as a consequence. Low doses stable iodide (1–100 μg) did not hamper the trapping function and resulted in rapid organification and low wash-out of the radioiodine (Childs et al 1950). The iohexol dose (65 mg/kg) used in our study is low, compared to a study in normal rats (300–600 mg/kg) (Yu and Shaw 2003). The amount of free contaminant inorganic iodide in iohexol has been estimated to range from 1.48 to 20.8 μg/ml (Laurie et al 1992). In our study, based on the injected volumes of iohexol (range 1.65–4.5 ml, 300 mg/ml), the calculated amount of contaminant cold iodide would range between 34 and 94 μg when considering the maximal amount of 20.8 μg/ml in iohexol. These levels of iodide are of a similar order to these that did not provoke increased clearance from the thyroid in Childs' study (Childs et al 1950).
Although not ideal, in our situation we had to treat the animals with radioactive iodide 24 h after the iohexol administration from a practical and economical point of view.
Despite our expectation that interference of the contrast with uptake of radioiodine would be present and might influence the results of radiotherapy, the outcome did not differ significantly between both groups. The percentage of cats with normal TT4 was approximately the same in both groups. Many cats had TT4 below the lower limit 6 months after therapy even with a dose considered in the low range (mean 111 MBq (2.97 mCi)) (Peterson and Becker 1995, Slater et al 2007). No clinical signs of hypothyroidism were reported in either group and no cat had to receive medication for hypothyroidism. The reported incidence of low TT4 in literature varies considerable when dose ranges of 75–227 MBq (2.1–6.13 mCi) are used (Meric and Rubin 1990, Peterson and Becker 1995, Nykamp et al 2005). Incidence of low TT4 ranged from 8 to 56% at 1 month post-therapy (Meric and Rubin 1990, Nykamp et al 2005) and from 3 to 30% at 3 months and more (Meric and Rubin 1990, Nykamp et al 2005). Therefore, the higher incidence of below normal TT4 values in this series may be due to the low number of cats in face of the high variability in the incidence of hypothyroidism after radioiodine treatment. This may again be related to the previously discussed high individual variability in RAIU in hyperthyroid patients. The fixed dose regime may be insufficient for some and too high for other patients.
Another possibility is the presence of concurrent severe non-thyroid disease which has been reported to be associated with subnormal TT4 levels in 21–59% of cats (Peterson and Gamble 1990). All cats were clinically examined, albeit by different veterinarians at different practices, and no concurrent illness was reported at this 6 months check. However, neither a thyroid stimulating-hormone (TSH) test nor a diagnostic pertechnetate scan was systematically performed and, therefore, we cannot completely exclude the possibility of non-thyroid disease.
The percentage of cats with residual hyperthyroidism was slightly higher in the iohexol group (15 versus 6%) which was not significantly different from the control group. However, a limited number of cats were included in the iohexol group, and the investigation of a larger group is necessary to come to definite conclusions.
Besides the effect on absorbed dose of radioiodine for therapeutic purposes, it should also be considered that, when radiodiodine is used for diagnostic purposes, iohexol administration may affect the diagnostic performance of this test. Potential interference with uptake of pertechnetate may occur. However, no statistical significant difference was found in this study in the ratio of thyroid/salivary activity between both groups. Both salivary glands and thyroid cells have similar symporters at their disposal. If iohexol would interfere with both systems, it is possible that both effects cancel each other out, as a ratio is calculated for diagnostic purposes.
Conclusions
In this study decreased absorbed dose accompanied by less individual variation and without an alteration in radioiodine turnover was observed in a small group of hyperthyroid cats pre-treated with iohexol for GFR measurements. In this small group no significant impact was noted on therapy outcome.
