Abstract
An 11-year-old neutered male domestic shorthair cat was presented with an epulis. A hemispherical mass, 8 mm in maximum diameter, without a peduncle and bright reddish in colour, was observed on the gingiva of the left mandible. Radiography failed to show any infiltrating osteolysis. The epulis was surgically removed via gingival incision around the margin to the depth of connective tissue layer. Histopathological examination indicated that the epulis contained a large number of multinucleated giant cells (MGCs) intermixed with mononuclear mesenchymal cells in a loose fibrovascular stroma. Mitotic cells were found, mainly in the centre of the mass. MGCs were stained positive by the tartrase resistant acid phosphatase (TRAP) staining, indicating osteoclasts activity. Immunohistochemical staining for proliferating-cell nuclear antigen (PCNA) was observed within the majority of mononucleated cells, whereas multinucleated cells did not stain. An osteoclast-like giant cell tumour was concluded in this case. The origin of epulis is likely from the periosteal tissue. The cat recovered uneventfully and no recurrence has been noted for 3 years thereafter.
Epulis is a non-specific clinical designation for a localised and exophytic growth of mass on the gingiva (Gardner 1996). In the veterinary literature, giant cell epulis (GCE), also called peripheral giant cell granuloma, has been mostly reported in the dog (Blood and Studdert 1999). This unspecific lesion is most frequently associated with local periosteal inflammation or trauma in humans (Bhaskar et al 1971). Epulis is rarely observed in the cat, with only two GCE cases reported in the literature (Schneck 1975, Rothwell et al 1988). The present report describes the clinical presentation of a new feline GCE and the histological and immunohistochemical characterisations of this lesion.
An 11-year-old neutered male domestic shorthair cat (weighed 3.8 kg) was referred to the Veterinary Medical Teaching Hospital with a firm, hemispherical, bright reddish epulis protruding beyond the gingiva of the fourth premolar of left lower jaw (Fig 1). According to the owner, the epulis had significantly enlarged in the 2 weeks since it had first been observed. At the time of referral the epulis measured approximately 8 mm in diameter and was occasionally surrounded by blood-tinged saliva. The cat showed normal appetite and stamina, no other clinical or clinicopathological abnormalities were observed. Blood figures (blood cell counts, haemoglobin and haematocrit) and serum chemistry (liver function, renal function, etc) were normal. The cat had no history of overt trauma in the gingival area before the appearance of the epulis. Radiography of the left mandible revealed no abnormalities of the mandible and there were no erosive or reactive changes associated with the lesion. The epulis was removed via gingival incision around the margin to the depth of connective tissue layer and the surgical wound was not sutured. Representative specimens were fixed in 10% neutral buffered formalin and submitted for pathological and immunohistochemical examinations.

A firm, hemispherical, bright reddish epulis protruded beyond the gingiva of the fourth premolar of the left lower jaw.
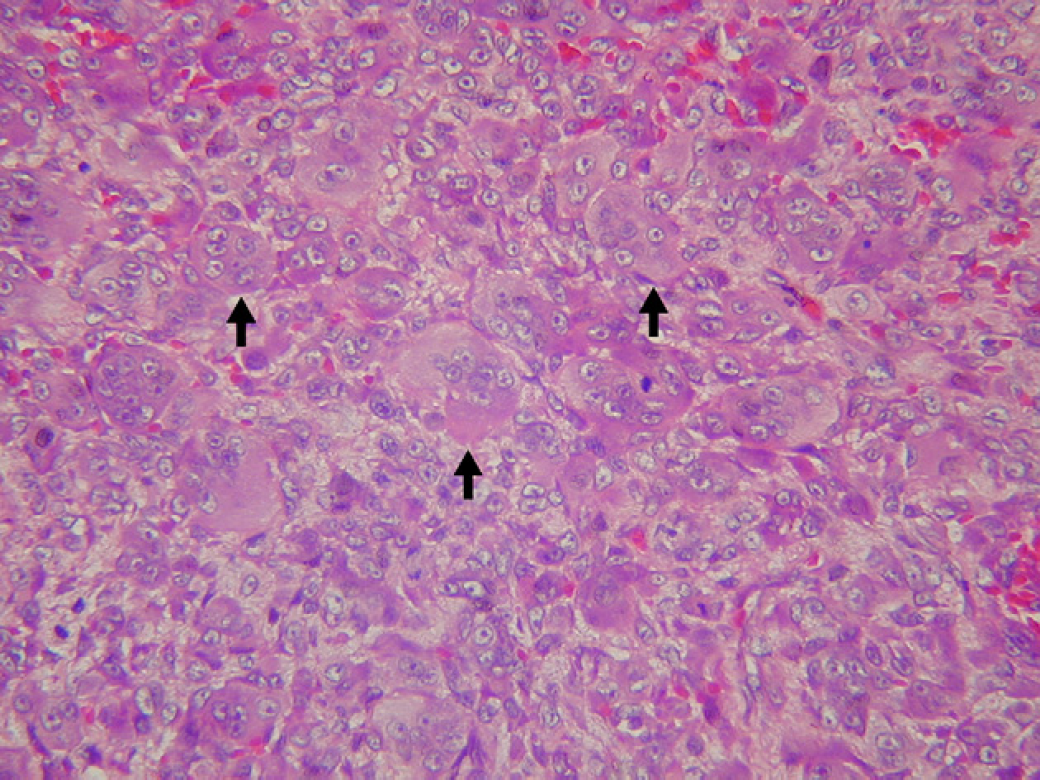
Histological examination revealed that the epulis was covered by a layer of stratified squamous epithelium, with superficial ulcerative and haemorrhagic areas. Neoangiogenesis was observed mainly in association with areas of ulceration. There was a clear demarcation between the mass and the normal tissue but no encapsulation. The mass was composed of a loose fibrovascular stroma and three distinct components: multinucleated giant cells (MGCs) containing three to more than 30 characteristic ovoid and spindly shaped nuclei, scattered throughout the mass (Fig 2); round-shaped mononuclear cells with round nuclei and a single round or ovoid nucleolus that were evenly distributed throughout the mass; and spindly shaped mononuclear cells arranged as cellular bundles mainly distributed in the central portion of the mass. Mononucleate cells and giant cells were not arranged in groups around a causal agent, as in some granulomas. Mitotic figures were detected in mononuclear cells localised mainly in the central portion of the lesion (Fig 3) and fewer in the periphery. Histological sections were stained by the Masson's trichrome method and counterstained by aniline blue and light green. Positively stained areas were seen only in the central area and constituted less than 5% of all tissues. Osteoclast-like MGCs were identified by tartrase resistant acid phosphatase (TRAP) staining (Tiffee and Aufdemorte 1997), in which all osteoclast-like MGCs scored positively (Fig 4). In addition, immunohistochemical staining using antibodies against the proliferating-cell nuclear antigen (PCNA) (PC10: sc-56, Santa Cruz Biotechnology, USA) (Mighell et al 1996) demonstrated that the majority of mononuclear and some stromal cells had PCNA-positive nuclei (Fig 5). Based on the histopathological and the immunohistochemical findings, a diagnosis of osteoclast-like giant cell tumour was made.
Haematoxylin and eosin, magnification 400×. MGCs (arrows) containing numerous round and ovoid nuclei are seen throughout the tumour mass.

Haematoxylin and eosin, magnification 400×. Multinucleated, mononucleated and fibroblast-like cells arranged compactly. Mitotic figures (arrows) are seen in the central area.

TRAP stain, magnification 400×. Strong positivity for MGCs (arrows) and some mononucleated cells (empty arrows) is observed.

PCNA stain, magnification 400×. Positive staining (arrows) is presented in the nuclei of mononucleated and stroma cells.
Histological findings in the cat reported here displayed many features reported in the literature (Schneck 1975, Rothwell et al 1988), which were characterised by large numbers of MGCs and round mononuclear mesenchymal cells in a loose fibrovascular stroma. Focal ulcers with adjacent haemorrhage and infiltration of inflammatory neutrophils are suggestive of infection/irritation secondary to local trauma resulting from the tumour formation. In this case no causal agent was found. The entire lesion was well circumscribed without invasion to the deeper tissue, indicating the focal nature of the lesion. This type of epulis has been called ‘peripheral giant cell granuloma’ (Schneck 1975, Rothwell et al 1988). Bonetti et al (1990) suggested that human giant cell granuloma exhibits a phenotype distinct from other giant cells found in chronic inflammation and that these cells might be true osteoclasts. In a later human study, Tiffee and Aufdemorte (1997) suggested that giant cell granuloma might originate from mononuclear cells that express markers for both osteoclasts and macrophages. In the case reported here, positive results from TRAP and PCNA staining indicated the osteoclast-like lineage of cells involved and also suggested neoplastic characteristics in the MGCs.
Perhaps the most important differential diagnosis in this case is to giant cell osteosarcoma, which is definitively diagnosed by the production of osteoid by the neoplastic cells and presentation of irregular trabeculae of woven bone (Negrin et al 2006). However, in the present case, no obvious osteo-like material or irregular trabeculae of woven bone was found microscopically. Additionally, the fact that MGCs scatter throughout all the tumour in the current case, made it different from a feline giant cell osteosarcoma, which is characterised by scattered nests of MGCs separated by small bundles of pleomorphic and polygonal cells (Negrin et al 2006). Other differential diagnosis of epulis in the cat usually includes malignant fibrous histiocytoma and fibrous epulis. Malignant fibrous histiocytoma shares some cellular similarities with euplis, for instances, MGCs, histiocytic-like cells and fibroblasts-like cells (Goldschmidt and Shofer 1992), however, the tumour behaviour reported in this case was benign instead of malignant. This was supported by the fact that the epulis did not recur during a 3-year follow-up, which was different from a malignant fibrous histiocytoma which tends to recur at surgical site. On the other hand, the differentiation from fibrous epulis was made based on cellular composition and the location of the tumour in which microscopically, a feline fibrous epulis showed densely compacted fibro-collagenous core covered by slightly thickened squamous epithelium (Clogin et al 2001) while GCE is often darker red in colour and occurs in the gingiva. Finally, as jawbone was not involved in the lesion, the current epulis was deemed different from the bone-originated osteoclastoma.
The origin of epulis remained to be discussed. GCE has been considered as a consequence of trauma resulting from extraction of teeth in feline species (Schneck 1975), but this was apparently not in accordance with the current case. GCE is generally accepted as benign with little tendency to recur (Schneck 1975, Valentine and Eckhaus 1986, Gardner 1996), although recurrence after surgical removal has been reported in a suspected case (Rothwell et al 1988), it was eventually categorised as an osteoclastoma. Nevertheless, the fact that GCEs may present with similar histology but different cell behaviour cannot be overlooked and might suggest a different spectrum of diseases or different process. Although the original lesion site of osteoclast-like GCE remains obscure, it is obviously not consistent with osteoclastoma. The evident osteoclast nature without infiltrating osteolysis, partially supported that periosteal tissue could be a possible origin for this epulis similar to the one that was reported previously (Schneck 1975).
Footnotes
Acknowledgements
The authors would like to thank Prof Dr Chi-Chung Chou for his critical reading of this manuscript.
