Abstract
In-house tests for the identification of faecal parvovirus antigen are now available. The majority of these are licensed for canine parvovirus only; but anecdotal information suggests that they will detect feline panleukopenia virus (FPV) as well. This prospective study was designed to compare five commercially available test systems. In total, 200 faecal samples from randomly selected healthy cats (148) and cats with diarrhoea (52) were tested and compared with the results of examination by electron microscopy. Ten cats were positive for FPV and all of these had diarrhoea. In-house canine parvovirus tests can be used to detect FPV. All tests were suitable to screen cats for faecal parvovirus excretion (positive predictive values for the Witness Parvo, the Snap Parvo, the SAS Parvo, the Fastest Parvo Strip, and the Speed Parvo were 100.0, 100.0, 57.1, 38.9, and 100%, respectively, negative predictive values for the Witness Parvo, the Snap Parvo, the SAS Parvo, the Fastest Parvo Strip, and the Speed Parvo were 97.4, 97.9, 98.9, 98.4, and 97.4%, respectively). In-house parvovirus tests may be positive up to 2 weeks after vaccination, and therefore, in recently vaccinated cats positive results do not necessarily mean infection.
Feline panleukopenia virus (FPV) is a common viral organism infecting not only domestic cats but also other Felidae (including tigers, panthers, and leopards) as well as species of the families Mustelidae, Procyonidae, and Viverridae including raccoon, ring-tailed cat and mink (Scott 1987). The virus causes feline panleukopenia which is highly contagious and associated with significant morbidity and mortality. Not all cats infected will develop clinical signs, but if they do, the disease can be peracute and severe (mortality rate up to 100%; often without diarrhoea) or acute (25–90% mortality; fever, anorexia, vomitus, sometimes followed by diarrhoea) (Gillespie and Scott 1973, Addie et al 1996, Greene 1998). The virus is highly resistant and can survive up to 1 year in infected organic material (Poole 1972). Moreover, FPV can be transmitted to susceptible cats on clothes, shoes, or transport cages (Scott 1987). Cats can also be infected with canine parvoviruses (various antigenic types) (Mochizuki et al 1996, Truyen et al 1996, Nakamura et al 2001), and develop signs of panleukopenia.
Clinical signs in cats are variable and leukopenia is not always present at the time of presentation (Greene 1998). Early detection of the virus with reliable test systems is crucial to separate infected cats (especially in veterinary hospitals with other severely ill and immunocompromised animals), to treat the patients aggressively, and to inform the owners about prognosis and potential risks for other household cats.
Recommended confirmation tests to diagnose parvovirus infection in vivo are electron microscopy of faecal samples or polymerase chain reaction (PCR) of faeces, blood, or infected tissues (Eugster 1980, Schunk et al 1995, Schatzberg et al 2003). These tests, however, are time-consuming, labour-intensive, expensive, need the expertise of specialists, and are not readily available. Sending off samples to a commercial laboratory delays the diagnosis when time is crucial.
In recent years, in-house tests for the detection of faecal canine parvovirus and/or FPV antigen for use in veterinary practice have been introduced to the market. These tests are based on either enzyme-linked immunosorbent assays (ELISA) or immunochromatographic technology. The close structural and antigenic relation of FPV and canine parvoviruses (Mochizuki and Akaboshi 1988, Parrish 1991) offers the possibility to test cats for FPV (as well as canine parvoviruses) with the same test kit as dogs. This study was designed to evaluate the strength and weaknesses of five commercial tests and to assess their sensitivity, specificity, and predictive values.
Materials and Methods
Faecal Samples
The study was performed at the Medizinische Kleintierklinik of the Ludwig-Maximilians-Universität Munich, Germany. Faecal samples were obtained from 148 healthy cats and 52 cats with diarrhoea. The latter cats were patients of the Veterinary Teaching Hospital of the University; the healthy cats were either also from the hospital population (eg, cases presented for routine vaccination) or originated from two animal shelters around Munich. Ten of the cats with diarrhoea had clinical signs consistent with feline panleukopenia.
On each faecal sample, all in-house tests were performed simultaneously exactly following the instruction manuals. The samples were analysed immediately after sample collection (maximum 15 min after collection), to avoid storage or freeze–thawing effects. A portion of each sample was stored at room temperature and transferred to the Institute for Medical Microbiology and Infectious Epidemic Diseases for electron microscopy evaluation within 2 h. Samples were blinded for evaluation.
Test Systems Evaluated
Five commercially available test systems for the detection of FPV and/or canine parvovirus antigen in the faeces were evaluated. Tests used in this study included the Witness Parvo (Synbiotics, France), the Snap Parvo (Idexx, Germany), the SAS Parvo (SA Scientific, USA), the Fastest Parvo Strip (MegaCor, Austria) and the Speed Parvo (Bio Veto Test, France).
The Witness Parvo is based on immunomigration (RIM technology). Antibody/antigen complexes are migrating to a reactive band where they are visualised through a pink colour. The test has to be stored at room temperature. This test is licensed only to detect canine parvovirus antigen. The Snap Parvo is based on ELISA technology and has to be stored at +2 to +7°C. This test is licensed only to detect canine parvovirus antigen too. The SAS Parvo, the Fastest Parvo Strip, and the Speed Parvo are immunochromatographic tests (sandwich system with two antibodies) that are licensed to detect canine parvovirus and FPV antigen. They can be stored at room temperature. The SAS Parvo uses a colour-coded parvovirus-specific antibody that binds the virus and transports it to the reactive band, where a second fixed antibody binds to the antigen antibody complex and a colour indicates the positive test result. The Fastest Parvo Strip is based on two different monoclonal antibodies, one membrane-fixed and the other bound to movable gold particles. If there is parvovirus antigen in the sample it will bind to the gold particle antibody and will be visualised at the positive band through membrane-fixed antibodies. Unbound gold particle antibodies will pass the positive band and will bind to the negative band also through membrane-fixed antibodies. A colour-coded monoclonal antibody binds to the parvovirus antigen of the sample in the Speed Parvo. The resulting antigen antibody complexes will then be seen at the reactive band using a second polyclonal membrane-fixed antibody. All test systems mentioned above are designed for use in private practice.
Confirmation Test
For confirmation of the test results direct visualisation of parvoviral organisms was performed by electron microscopy. The electron microscope EM 10 CR (Zeiss, Germany) was used. The method is described elsewhere (Gedek et al 1993, Vieler and Herbst 1995).
Data Analysis
Parameters used to compare the tests quality were the percentage of invalid test results (eg, missing control band or coloured background), the percentage of results that were difficult to interpret (eg, weak control band or dirty result window), the diagnostic sensitivity (the proportion of positive test results in infected animals), the diagnostic specificity (the proportion of negative test results in uninfected animals), the positive predictive value (PPV) (the probability that a test-positive animal is infected), and the negative predictive value (NPV) (the probability that a test-negative animal is not infected). A positive test result in a sample confirmed with a positive electron microscopy result was considered a true-positive result. A positive test result in a sample with a negative electron microscopy result was considered a false-positive result. On the other hand, a negative test result confirmed with a negative electron microscopy result was considered a true-negative result and a negative test result in a sample with a positive electron microscopy result was considered a false-negative result. The calculations of sensitivity, specificity, positive, and NPVs are based on these categories.
Results
The Witness Parvo, the Snap Parvo, and the Speed Parvo had informative handling instructions, were easy to perform, and independent of the available amount of faeces. Results were given in 5, 8, and 5 min, respectively. The colour intensity of the reactive band correlated with the amount of antigen inside the samples when measured with the Snap Parvo and the Speed Parvo. In the SAS Parvo, valid results were available in 10 min. Using this test only small volumes of faeces (<30 μg) can be used, as higher volumes can lead to invalid test results due to the high viscosity of the samples. The Fastest Parvo Strip was easy to perform and valid results were available within 5 min. This test, however, can only be performed when larger volumes of faeces are available (≥pea size).
The presence of parvoviruses was confirmed by electron microscopy in 10 of 52 samples from cats with diarrhoea (prevalence 19.2%). This prevalence closely correlates to the prevalence of parvovirus infection in a larger population of cats of the same geographic region. Horlacher et al (2002) analysed 867 faecal samples of cats with clinical and/or laboratory signs of panleukopenia by electron microscopy; of these, 145 (16.7%) samples tested positive for parvovirus. Table 1 summarises the percentage of invalid test results, the percentage of results that were difficult to interpret, the diagnostic sensitivity and specificity, and the positive and NPVs.
Comparison of five parvovirus test systems
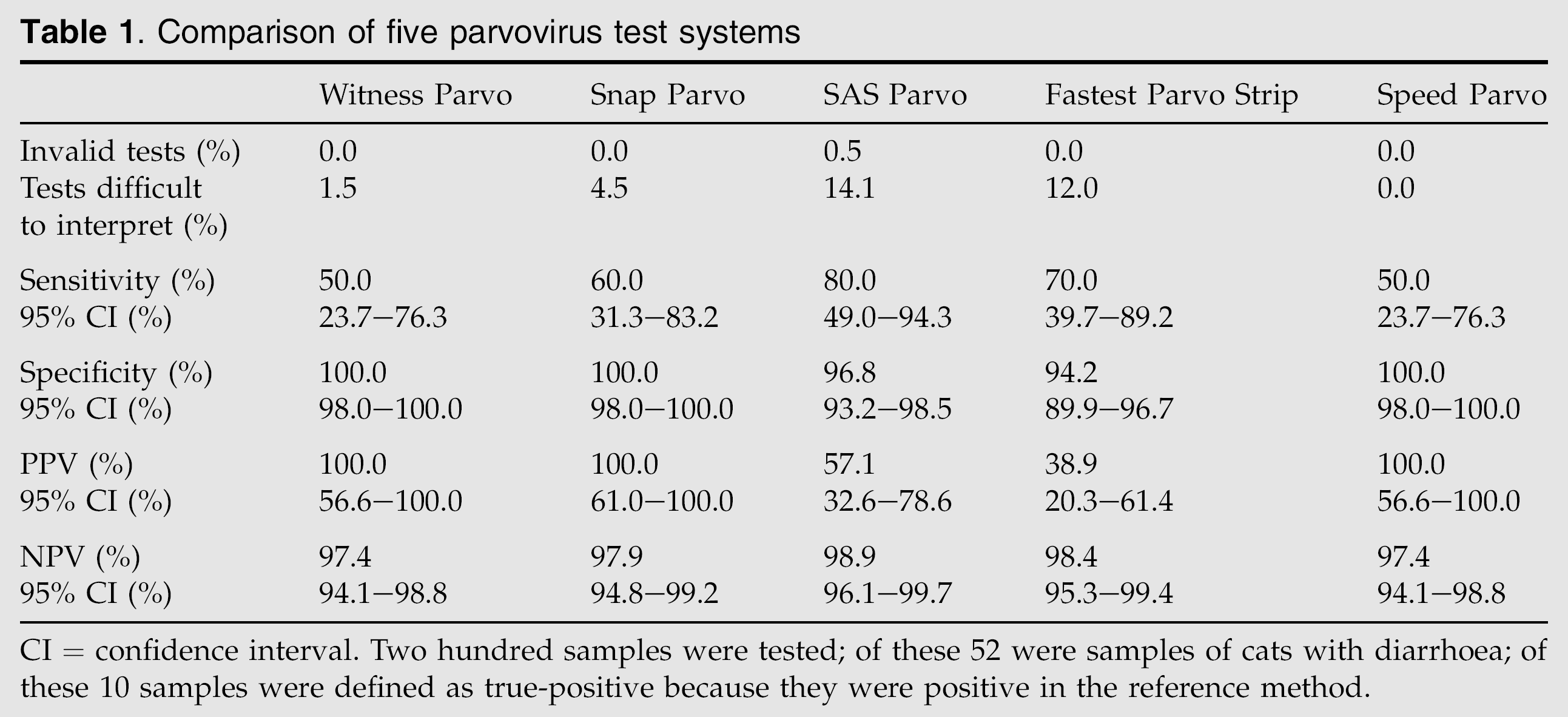
CI = confidence interval. Two hundred samples were tested; of these 52 were samples of cats with diarrhoea; of these 10 samples were defined as true-positive because they were positive in the reference method.
Discussion
Little information is available on the quality of different in-house tests to detect parvovirus in the faeces of cats. Only one paper published to date compared different tests for use in cats amongst other species (Esfandiari and Klingeborn 2000). However, results of this paper have to be interpreted carefully because three different ELISAs were used as ‘gold standard’ and no electron microscopy was performed.
To assess the reliability of a new diagnostic test, it has to be compared with a reference method. This reference or confirmation test should be performed by an independent and blinded person, and samples of the patients tested should be representative of those in which the test will be applied in clinical practice (Polzin et al 1999). In this study, electron microscopy was chosen as the reference method for verification of parvovirus infection, although this test has some limitations. The test is very specific if performed by experts, but can be false-negative because of intermittent excretion or excretion of too few viral organisms. In addition, the test can be performed too early in the course of the infection where the virus is not yet excreted with the faeces (Krauss and Arens 1981). PCR of blood might be more accurate in the early phase of infection when there is viraemia but not yet excretion of the virus in the faeces. This method, however, can be false-negative in a later phase when viraemia is not present anymore. Moreover, this test would be unsuitable as a reference method for tests measuring the excretion of a virus. PCR of faeces suffers from the same limitation of intermittent shedding as electron microscopy, although it might be more sensitive (Schunk et al 1995). It was, however, not readily available to us at the time when the study was performed. Measurement of antibody titres is not useful to diagnose parvovirosis because many cats have antibodies due to vaccination or previous clinically inapparent infection. A four-fold rise in titre taken 2 weeks apart may be indicative of acute infection (Greene 1998) but delays the clinical diagnosis unacceptably. The confirmation test chosen for this study was performed by an independent specialist, who had no information about the history of the samples.
Another limitation of the study is that the test systems were evaluated to the point that data have been generated by performing the tests according to the manufacturers' instructions with no attempt to validate the precision or accuracy of the exact test procedures. There is a difference in evaluating a method for its performance and evaluating a method to see if it corresponds to the clinical disease and to the ‘gold standard’. The evaluation performed here is mainly of clinical usefulness while the other would be a total assessment of the methods performance and precision.
Comparing general aspects of the test systems evaluated in this study, all but the SAS Parvo had clear, easily understandable handling instructions. The SAS Parvo had only one invalid test result (0.5%). There were no invalid results in all the other test systems, which is excellent. The SAS Parvo and the Fastest Parvo Strip had high numbers of tests that were difficult to interpret because of background colour (14 and 12%, respectively). In this regard, the Speed Parvo performed best. It had no invalid tests and no results that were difficult to interpret.
With regard to the sample size, the Witness Parvo and the Snap Parvo could be performed with the smallest faecal volume; according to the user manual a cotton swab of material is enough to receive valid results. This is especially important for patients with very liquid faeces and those without any defecation. The SAS Parvo had problems when amounts of faeces were only a little over size. This may lead to higher viscosity of the samples and to invalid test results. The handling instructions of this test point out that a volume of 30–50 μg should be taken, which is difficult to determine. In consequence, this test can only be used by experienced personnel; otherwise, the number of invalid test results will be too high.
As far as the different quality criteria of the tests are concerned, the NPV is most important for a screening test because it predicts the likelihood that a cat is not infected with parvovirus and thus will not potentially shed the organism and infect other cats. All tests were quite similar in this regard with NPVs ranging from 97.4 to 98.9%.
The SAS Parvo and the Fastest Parvo Strip were the two test systems with the highest NPV (98.9% and 98.4%, respectively). Both tests, however, had a relatively high number of false-positive results (6 of 199 and 12 of 200, respectively). The instruction manual for the Fastest Parvo Strip states that the test can be positive up to 12 days after vaccination of cats with a modified life vaccine. Interestingly, all six samples that tested ‘false-positive’ with the SAS Parvo were taken from recently vaccinated cats. The same held true for 11 of 12 samples that tested ‘false-positive’ with the Fastest Parvo Strip. Moreover, the 12th ‘false-positive’ sample tested with the Fastest Parvo Strip was drawn from a cat that had a positive parvovirus PCR in the blood. Whether all these tests where truly false-positive cannot be completely ruled out, but it rather can be assumed that both tests were probably more sensitive than the reference method, the electron microscopy, to detect minimal amounts of viral antigen. Therefore, a more sensitive reference method is needed, and likely PCR of faecal samples, which might be more sensitive, should be the method of choice in the future (Schunk et al 1995).
In summary, all tests were suitable to screen cats for faecal parvovirus excretion. The Fastest Parvo Strip was the test with the best overall performance. It had the second highest NPV (98.4%), was easy to perform, had no invalid test result, was fast, and seemed to have a higher sensitivity than electron microscopy. The SAS Parvo had the highest NPV (98.9%), but was more difficult to perform. It had one invalid test result even after a ‘learning phase’.
In general, the recommendation for a diagnostic screening to detect cats infected with parvovirus would be to use an in-house test first, followed by a faecal PCR for confirmation in questionable cases.
Footnotes
Acknowledgements
This work was undertaken at The Clinic of Small Animal Medicine, Ludwig Maximilian University, Munich, Germany without additional funding.
