Abstract
Glutathione is an important intracellular tripeptide with multiple functions. Abnormal glutathione metabolism is thought to play an important role in various diseases of cats. However, no data regarding concentration of plasma glutathione are available for domestic cats. This study discusses the development of a rapid, simple high pressure liquid chromatography method for measurement of reduced glutathione (GSH), oxidized glutathione (GSSH) and total glutathione (GSHt) in plasma, for the purpose of establishing baseline data for future studies. The mean concentrations of GSH, GSSH and GSHt were 4.51±1 μM; 19.44±3.79 μM (expressed as GSH equivalent) and 23.59±3.89 μM, respectively. This is the first report of plasma glutathione concentrations in this species.
Glutathione is a water soluble tripeptide composed of glutamine, glycine and cysteine. Total glutathione (GSHt) can be free or bound to proteins. Free glutathione is present mainly in its reduced form [reduced glutathione (GSH)] which can be converted to the oxidized form [oxidized glutathione (GSSH)] during oxidative stress, in a reaction catalyzed by the enzyme glutathione peroxidase (Pastore et al 2003). GSH exists in most tissues and it is abundant intracellularly in erythrocytes, liver, spleen, kidney, leukocytes and cells of the ocular lens (Pastore et al 2003). Glutathione has evolved to serve diverse functions in biological systems including detoxification of xenobiotics, transport of amino acids, stabilization of cell membranes and synthesis of proteins and DNA (Cereser et al 2001). A deficiency of GSH puts the cell at risk of oxidative damage, therefore, maintaining an optimal intracellular GSH:GSSH ratio is critical for maintaining normal cell function and survival (Towsend et al 2003).
The cat is uniquely susceptible to oxidant injury (Webb et al 2006). Feline erythrocytes are sensitive to oxidative stress because their hemoglobin molecule contains 8–10 reactive sulfhydryl groups as opposed to four in the dog and other species. Therefore, the feline hemoglobin molecule is more susceptible to disruption following oxidative insult (Snow 1962, Harvey and Kaneko 1976, Christopher 1989, Christopher et al 1990). GSH is responsible for maintaining the sulfhydryl groups of hemoglobin in a reduced form, a requirement for their normal function.
During severe oxidative stress and detoxification reactions involving GSH, the concentration of GSH may decrease and the concentration of GSSH may increase in the affected cells. This results in a decreased export of GSH and an increased export of GSSH to plasma, thereby altering the GSH redox state of the plasma pool (Sakhi et al 2006). The altered plasma GSH and GSSH concentrations can, thus, reflect GSH/GSSH status and oxidative stress in other less accessible cells and tissues. Therefore, GSH and GSSH concentrations in plasma have been considered essential as an index of whole body glutathione and possibly a useful indicator of disease risk (Jones et al 2000). An imbalance of GSH is observed in various human diseases including cancer, neurodegenerative disorders, cystic fibrosis, human immunodeficiency virus infection, diabetes mellitus, renal failure, liver disease and aging (Towsend et al 2003, Webb et al 2006). There are few communications about the role of GSH in cat diseases. Feline immunodeficiency virus (FIV) infection was associated with significant and quite distinct alterations in intracellular GSH concentrations in leukocytes' populations from FIV infected cats (Webb et al 2006). Center et al (2002) discussed low liver GSH and GSHt concentrations in cats with naturally occurring liver disease. Webb et al (2003), in a feline acetaminophen model of oxidative injury, quantified GSH in whole blood and GSH, GSSH and GSHt in liver. Luvoni et al (2006) showed that the concentration of GSH is higher in mature oocytes as compared to immature ones. Hill et al (2005) quantified GSH in whole blood before and after oral acetaminophen doses in cats given dietary antioxidant supplementation.
The requirements for an accurate GSH determination are, respectively, specificity to discriminate GSH from other thiols and biotransformation products, and selectivity to separate GSH from possible interfering molecules in complex matrices (Camera and Picardo 2002). Methods for measuring GSH and GSSH in various biological samples have been recently reviewed (Camera and Picardo 2002, Pastore et al 2003). Some of these techniques such as spectrophotometry and fluorometry have good sensitivity and are inexpensive to run. They are non-separative techniques and have unsatisfactory specificity and selectivity. High sensitivity is an indispensable requirement for plasma GSH and GSSH quantification because both are present at low levels in extracellular compartments, especially when GSH is lowered due to oxidative stress and other conditions. High pressure liquid chromatography (HPLC) separation of GSH with fluorimetric detection has been extensively applied when a high sensitivity is usually required as in plasma. Because of the lack of strong chromophores and fluorophores in the chemical structure of GSH it is very often advantageous to derivatize the analyzed compounds introducing appropriate tags in the molecule in order to improve the detection limit (Camera and Picardo 2002). Ortho-phthalaldehyde (OPA) is widely employed to form fluorescent adducts of GSH. It reacts with both the sulfhydryl and primary amino group of GSH to form a highly fluorescence derivative (Camera and Picardo 2002, Pastore et al 2003).
The goal of this study was to determine the GSH, GSSH and GSHt in plasma of healthy cats. A rapid, simple HPLC method with fluorimetric detection was used. This method may be useful for clinical or experimental studies in cats, in a variety of different diseases in which GSH imbalance is suspected.
Materials and Methods
Chemicals and Reagents
GSH, GSSH, OPA, dl-dithiothreitol (DTT), tris (hydroxymethyl) aminomethane and sodium tetraborate were obtained from Sigma–Aldrich. Sodium acetate was obtained from Farmitalia, Carlo Erba. Sodium dihydrogen phosphate monohydrate and clorhidric acid were obtained from JT Baker (Xalostos, Mexico). Methanol and acetonitrile HPLC grade were obtained from JT Baker (Phillipsburg, USA). Metaphosphoric acid (MPA) was obtained from Riedel-de Haën (D-3016 Seelze 1). All HPLC buffers were filtered through 0.45 μm Millipore filters prior to use.
Standard Solutions
Ten millimolar stock solutions of GSH and GSSH in 0.01 N HCl were prepared, aliquoted and stored at −20°C. Working standard solutions were prepared daily from each stock solution by dilution in buffer tris pH 8.5.
Derivatization Reagent
OPA solution (5 mg/ml) was used for derivatization. The solution was prepared by dissolving 50 mg of reagent grade OPA in 0.5 ml methanol and diluting this to a final volume of 10 ml with 0.1 M sodium tetraborate, pH 9.9. The solution was aliquoted and stored at −20°C.
Sample Collection and Preparation
Twenty-two healthy adult cats were used. The mean age was 24 months (20–28 months), and the average weight was 3.52 kg. The cats were fed with cat food for adult maintenance [Association of American Feed Control Officials (AAFCO)]. Cats were anesthetized with ketamine (15 mg/kg) and xylaxine (0.5 mg/kg), and then blood (2 ml) was collected by venepuncture of the jugular vein and placed quickly into cool tubes containing ethylenediaminetetraacetic acid as anticoagulant. The samples were immediately centrifuged at 6000 rpm for 5 min at 4°C to separate plasma from red blood cells (RBCs). Plasma (100 μl) was deproteinized by adding 400 μl of cold 6% MPA. Precipitated proteins were removed by centrifugation at 6000 rpm for 7 min at 4°C. The resulting supernatant was stored at −20°C for 24 h until analysis. For the derivatization procedure, 100 μl of MPA supernatant was neutralized by addition of 100 μl 0.1 M tris buffer, pH 8.5. Derivatization with OPA was performed by mixing 100 μl of this solution with 100 μl of OPA solution. After 5 min at room temperature, 20 μl was injected into HPLC system for GSH determination. GSHt was evaluated by performing a reduction step of plasmatic GSSH with DTT. Before protein precipitation, 100 μl of plasma was mixed with 100 μl of 25 mM DTT solution and 50 μl of 0.1 M tris buffer (pH 8.5). After 30 min incubation at 4°C, the proteins were precipitated by the addition of 800 μl 6% MPA and removed by centrifugation (6000 rpm for 7 min at 4°C). The MPA supernatant was stored at −20°C for 24 h and used for the derivatization procedure described previously for GSH.
Chromatographic Conditions
The HPLC separation of GSH–OPA was achieved on a Synergy Hydro-RP C-18 reversed-phase column, 150×4.6 mm; 4 μm particle size (Phenomenex) maintained at 30°C and followed by fluorimetric detection at 420 nm after excitation at 340 nm. Peak sensitivity was 2.5%. The HPLC system consisted of a Gilson Pump model 307 and a Gilson fluorometer detector model 129.
For GSH determination derivatives were eluted by isocratic elution A–B (30:70), where mobile phase A was 50 mM sodium acetate buffer, pH 6.2 and mobile phase B was acetonitrile:methanol (2:1). Mobile phase flow rate was 1 ml/min. The run time was 5 min.
For GHSt determination the gradient was from A–B (30:70) in the first 6 min, and then A–B (50:50) for 13 min, and then the column was re-equilibrated to initial conditions. The run time for GSHt was 25 min and mobile phase flow rate was 1 ml/min. The column was washed daily with ultrapure water (MilliQ) for 60 min and stored in 100% acetonitrile overnight. Integration of chromatograms was accomplished using Gilson 712 HPLC System controller.
Results
Typical chromatograms obtained are shown in Figs 1 and 2. Peak was identified by direct comparison of retention time with an analytical standard and adding GSH to plasma sampled and observing an increase in the corresponding symmetrical peak. The retention time of glutathione was 1.86 min. Chromatograms showed appropriate separation of GSH without interfering peaks.
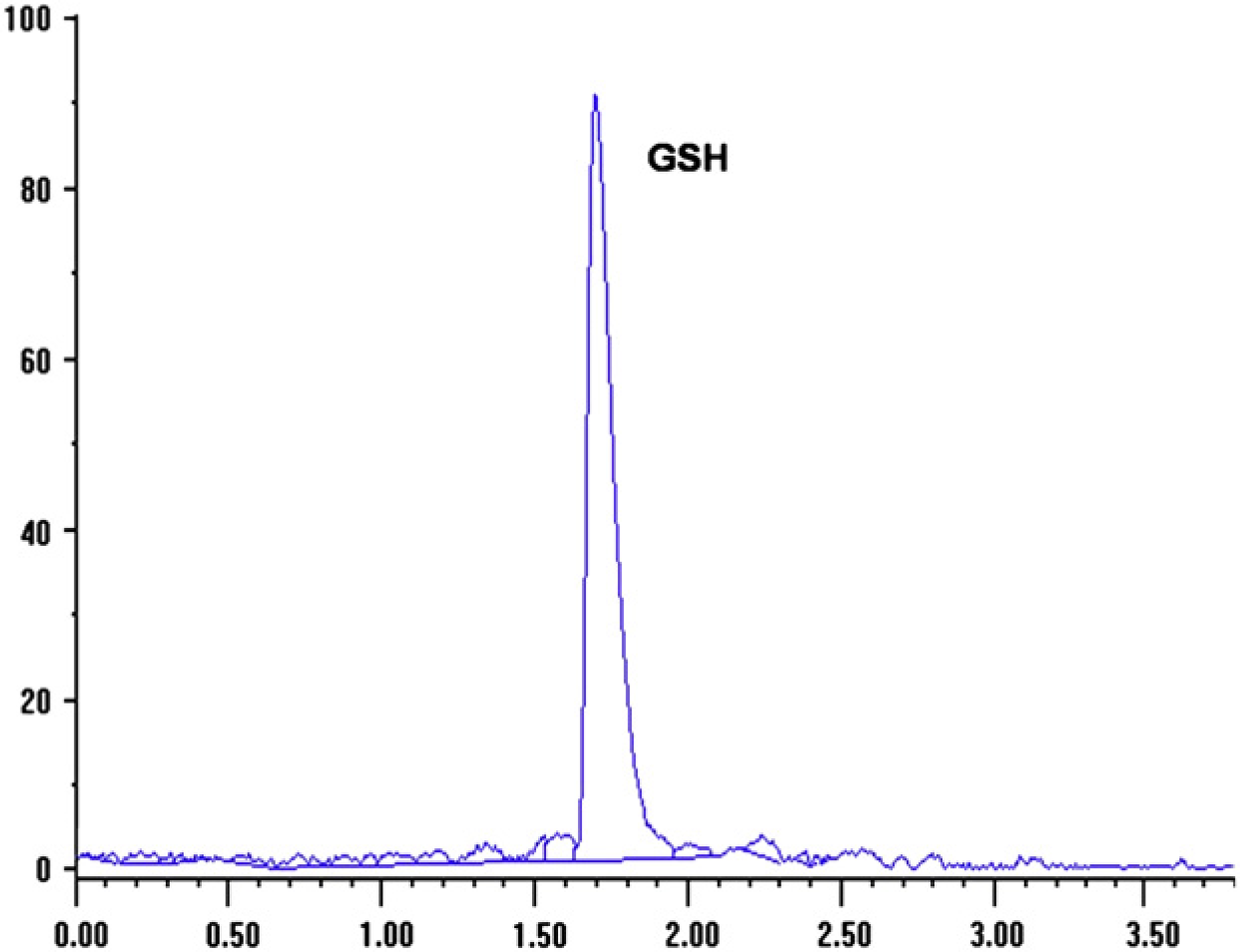
HPLC profiles of GSH for domestic cat plasma.

HPLC profiles of total GSH (GSHt) for domestic cat plasma following reduction with dithiotreitol.
The calibration curve for GSH showed a correlation coefficient (r) of 0.990 ensuring a linear response from 1.5 to 10 nM. The lower quantification limit was 0.95 nM. The recovery of GSH from plasma was 96.7% and of GSSH was 95%.
Table 1 shows the glutathione level in plasma of 22 adult healthy cats. The mean GSHt and GSH concentrations were 23.59±3.89 μM and 4.51±1 μM. The calculated oxidized fraction was 19.44±3.79 (expressed as GSH equivalents).
Concentration (μM) of different glutathione forms in plasma from 22 healthy cats

Discussion
A rapid, simple HPLC method for measurement of GSH, GSSH and GSHt in cat plasma through modifications of the Cereser et al (2001) technique was used to establish baseline values in cat plasma. This is the first report of GSH, GSSH and GSHt concentrations in plasma of domestic cats. Several methods are available for the determination of glutathione in biological samples. The most frequently used assay is based on the enzymatic recycling reaction discovered by Owens and Belcher and developed by Tietze (Kand'ár et al 2007). Several HPLC methods with ultraviolet, fluorescence or electrochemical detection have been successfully applied to the determination of GSH and GSSH (Camera and Picardo 2002, Pastore et al 2003). In the present study an HPLC technique based on the derivatization of GSH with OPA was used. The result is the formation of a highly fluorescent product. This technique has high sensitivity. This is important because GSH and GSSH are present in micromolar concentrations in plasma.
GSH and GSSH in plasma are considered an index of whole body glutathione and a useful indicator of disease (Jones, 2002). Changes in oxidative stress in tissues can be expected to alter the redox state of the plasma pool (Sakhi et al 2006). Cats are uniquely susceptible to oxidant injury and feline erythrocytes are much susceptible to oxidant injury as compared with other species. Up to 5% of RBCs from healthy cats demonstrate Heinz bodies as a result of this unique susceptibility to oxidant damage and relatively inefficient Heinz body removal (Blue and Weiss 1981, Christopher et al 1990). Moreover, cats have greater requirements for methionine and cysteine compared with other species and a relative inability to regulate their rate of amino acid metabolism (Stipanuk et al 1994, Kirk et al 2000). These amino acids are both precursors of GSH synthesis.
Previous studies of GSH regulation in cats have focused primarily on the role of erythrocytes and measure GSHt concentration in lysates of whole blood or in erythrocytes lysates (Center et al 2002, Hill et al 2005, Webb et al 2006). As intracellular GSH often varies widely from one cell type to another, the results of whole blood lysate assays can be influenced significantly by changes in the relative proportion of different cells types (Scott et al 1990). Webb et al (2006) found that feline erythrocytes contained significantly less GSH than feline lymphocytes or neutrophils using flow cytometry for quantification. These results illustrate clearly that there are normally pronounced differences in erythrocyte and leukocyte GSH content in cats and suggest that assessment of GSH content using blood samples may be significantly influenced by the total white blood count (Webb et al 2006). In the same way, concentrations of erythrocyte GSH may be influenced by erythrocyte age, because studies suggest higher GSH concentrations in young compared with aged erythrocytes and a greater ability for young RBC to synthesize GSH (Smith and Agar 1975).
Values for total, GSH and GSSH in human plasma differ from one laboratory to another. The variability of the results may be related to different methodologies, differences in sample processing or the selection of subjects that are under the influence of various factors affecting the plasma glutathione concentration. In Ono et al (2001), plasma GSHt concentration was 8.1 μM for a human adult group (53 subjects). Jones et al (2000) report 2.8±0.9 μM for GSH and 0.14±0.04 μM for GSSH in 24 adult human subjects. Levels of GSHt in healthy rat plasma were 22–27 μM and the 85% of the whole were GSH (Anderson and Meister 1980). Kand'ár et al (2007) found 1.82±0.55 μM for GSH and 0.154±0.044 μM for GSSH in a group of 70 human healthy donors. According to these data the concentrations of GSHt in cats are higher than the concentration reported for humans and similar to rats. GSSH was present in a higher percentage than GSH. This does not occur in either human or rat plasma. The increase in the GSSH:GSH ratio may be due to oxidation of GSH during sample preparation; but samples were processed one at a time. Blood was collected into cold tubes, immediately centrifuged at 4°C and then cold 6% MPA were added. The MPA supernatant was stored at −20°C for 24 h. Under these conditions GSH was stable for 3 months at −20°C (Cereser et al 2001). All aspects were considered in order to prevent the formation of oxidized forms during the sample preparation. Thus, the high GSSH:GSH ratio in plasma found in this study may be a consequence of high erythrocytes GSH requirements due to the special hemoglobin molecule. Erythrocytes have the capacity to transport GSSH into the plasma. This happens when the intracellular concentration is high, as, for example, during oxidative stress. The high susceptibility to oxidative stress in cats may be the cause of the high concentration of the GSHt and the high GSSH:GSH ratio in plasma compared to man and rats. More studies about the metabolism of GSH and its role in diverse pathologies of the cats are needed.
