Abstract
A 3-year female-neutered domestic shorthair cat presenting with exophthalmos and an ipsilateral subzygomatic soft tissue mass lesion is described. Magnetic resonance imaging of the mass was performed followed by complete surgical excision. The mass was determined to be a retrobulbar teratoma and complete resection was curative. Teratomas are rare germ-cell tumours that uncommonly form in extragonadal sites. A retrobulbar location has not been previously reported in the cat and should be considered a rare cause of exophthalmos in this species.
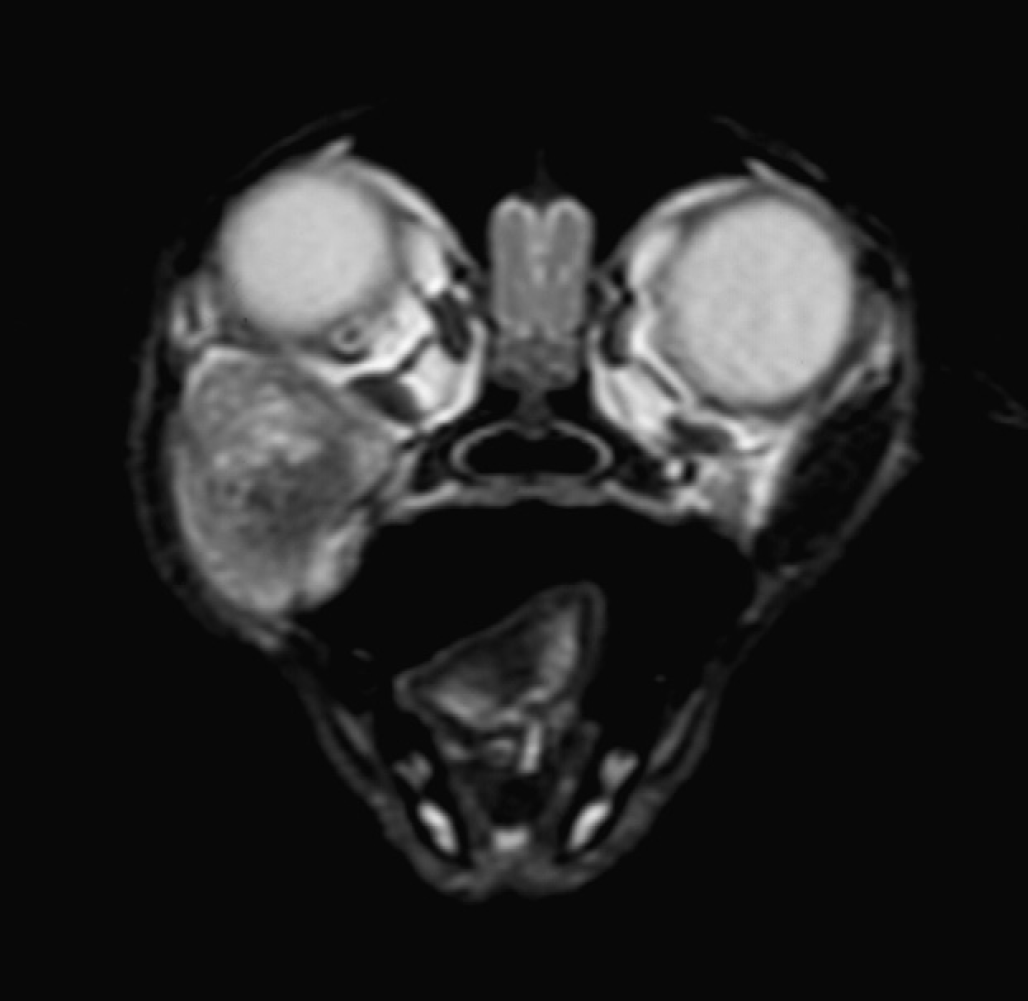
T2-weighted fast-spin echo transverse magnetic resonance image of the cat's head at the level of the mid-globes.
A 3-year female-neutered domestic shorthair weighing 4 kg was referred with an 8-week history of right-sided exophthalmos and an ipsilateral subzygomatic soft mass lesion of approximately 2 cm diameter. The cat was vaccinated annually against feline calicivirus, feline herpesvirus, feline panleukopenia virus and feline leukaemia virus and was predominantly an indoor cat with occasional reported fight wounds from other cats. The owners reported that since being a kitten, the cat had demonstrated intermittent ‘sucking’ sounds appearing to come from the mouth, unrelated to eating and to take more than usual time over eating, favouring the left side of the mouth and avoiding chewing soft food. No dysphagia, ptyalism, halitosis or head tilt had been noted. Prior to referral, therapy with clindamycin (6.25 mg/kg, (2.8 mg/lb), PO, q 12 h) for 10 days and carprofen (4 mg/kg, (1.8 mg/lb), SC, once) had resulted in no improvement and attempted needle drainage of the retrobulbar space yielded a small amount of viscous fluid but no cytological diagnosis. Skull radiographs were normal. On examination at the referral centre the cat was found to be in good body condition and general physical examination was normal. Mild right-sided exophthalmos with deviation of the globe dorsad and rostrad was seen, and a 2 cm diameter soft, well defined mass lesion was palpated ventral to the ipsilateral zygomatic arch. Gentle pressure on the base of the lesion resulted in further globe protrusion and the mass could be palpated per os, as it impressed the pharyngeal mucosa caudal to the fauces ipsilaterally. Ophthalmic examination was normal and the cat was found to be visual in both eyes. Haematology and biochemical evaluation was normal. Under general anaesthesia, magnetic resonance imaging (MRI) of the head was performed with a 1.5 Tesla superconducting magnet (General Electric 1.5T sigma MRI) using a human extremity coil. T1-, T2-weighted (with and without fat suppression) and T2* gradient echo dorsal and transverse images of the head were obtained with 2–3 mm slice thickness and 0.3 mm interspace. T1-weighted images were obtained before and after the administration of the paramagnetic contrast medium gadobenate dimeglumine (Mulitihance; Bracco spa, Milan, Italy) (33.4 mg/kg (15.2 mg/lb) of the dimeglumine salt, IV). A large (15–20 mm diameter) globoid mass was found to be present within the ventral aspect of the right orbit extending ventrolaterally between the right zygoma, pterygoid process of the maxilla and vertical ramus of the mandible (Fig 1). No invasion of bone or adjacent soft tissue was seen and the mass appeared discrete and clearly marginated. The mass displaced the right globe dorsally and slightly laterally and the overlying soft tissue laterally. The mass was heterogenous in structure with mixed high–low signal on T2- and T1-weighted images. The mass was predominately mid–low signal but within the centre of the mass was a stippled area of high signal on T2- and low signal on T1-weighted image consistent with fluid. The mass was surrounded by a uniform thin rim of hyperintense tissue on T1- and T2-weighted images that did not suppress on fat saturation images (Fig 2). Post-contrast T1-weighted images showed a thin, uniform rim of marked contrast enhancement surrounding the mass. No enhancement of the centre of the mass was present (Fig 3). The mass was hyperintense on T2* gradient echo images ruling out significant haemorrhage or calcification (Fig 4). Based on the MRI appearance, location and prior history of viscous fluid being aspirated differential diagnoses of zygomatic salivary duct cyst, granuloma, dermoid, epidermoid, abscess or benign tumour with inspissated cystic contents were considered. Given the well-circumscribed/encapsulated MRI appearance of the mass, and envisaged difficulty of complete surgical excision were the integrity of this (likely cystic) structure to be compromised, ultrasound-guided aspiration or needle biopsy was not performed. The cat was prepared for surgery and after premedication with medetomidine (4 μg/kg (1.8 mg/lb), IM), methadone (0.2 mg/kg (0.09 mg/lb), IM) and acepromazine (10 μg/kg (4.5 mg/lb), IM) was induced with propofol (7.5 mg/kg (3.4 mg/lb), IV) and maintained with isofluorane and oxygen. The region of the mass was approached via a right-sided orbitotomy using a subzygomatic approach with the patient in left lateral recumbency. The mass was identified medial to the fascia of the masseter muscle and was bluntly dissected out; closure was routine. An incisional biopsy of the mass lesion was submitted for culture and the remaining tissue submitted for histopathology. On incising the mass it was noted to contain large amounts of hair-like material (Fig 5). The cat recovered uneventfully and was eating the day after surgery; ocular position and vision in the affected eye was normal postoperatively.

Dorsal plane T2-weighted fast-spin echo with fat saturation (a) and T1-weighted fast-spin echo (b) images of the cat's head at the level of the hard palate. Note the hyperintense rim to the lesion on both the T2- and T1-weighted sequences and the failure of the rim to suppress (turn hypointense) on the fat-suppressed T2-weighted image.

Dorsal plane T1-weighted fast-spin echo fat-suppressed images post-administration of contrast (a) and digital subtraction image (b). Note the thin, uniformly enhancing rim.

Transverse plane T2* GRE image at the level of the mid-orbit. The uniform hyperintensity rules out haemorrhage within the mass.
Resected material was prepared for histopathology and stained with haematoxylin and eosin and immunohistochemistry performed. The lesion was found to consist partly of a large cystic cavity lined by skin with an inner layer of hyperplastic epidermis (Fig 6a). This showed marked hyperkeratosis with plentiful hair follicles and adnexal gland structures within the dermal tissue. The cystic cavity was filled with multiple hair fragments and amorphous eosinophilic material. More peripheral to the cyst the skin layer was found to extend into a zone of adipose tissue which then merged with an area of haphazardly arranged nervous tissue composed of nerve fibres and clusters of neurones (Fig 6b). Further staining of this tissue with synaptophysin (Polyclonal Rabbit Anti-Human Synaptophysin, DakoCytomation ref A 0010) demonstrated positive stain uptake consistent with nervous tissue. Surrounding this was found an area of fibrous tissue (Fig 6c) and striated muscle. The histopathological findings were consistent with a teratoma.

Excised mass sectioned to demonstrate tightly packed hair content.

(a) Photomicrograph of haematoxylin and eosin-stained section of cystic wall of excised mass. The cystic wall (A) is seen to comprise normal skin with defined epidermal and dermal layers containing follicle and sebaceous units. The cyst contents (B) comprises central keratinous debris interspersed with hair fragments brown pigmented sebum. (b) Haematoxylin and eosin-stained section of the mass adjacent to the cystic wall. Nervous elements comprising plump neurone bodies interspersed with supportive glial cells are seen. Subsequent positive staining with synaptophysin was demonstrated. (c) Haematoxylin and eosin-stained section of the mass adjacent to the nervous tissue of (b). A variably orientated arrangement of wavy collagen fibres and nucleated fibroblasts is seen.
Teratomas are differentiated tumours that contain elements arising from more than one germ-cell layer, are foreign to the organ or location in which they arise and show independent growth (Nielsen and Kennedy 1990). Teratomas with immature (poorly differentiated) histological components are often referred to as teratocarcinoma (Klein 2001). Teratomas are part of a family of germ-cell tumours that originate from malignant transformation of embryological tissues, teratomas being derived from totipotential germ cells that have undergone some somatic differentiation (MacLachlan and Kennedy 2002). They are characterised by the presence of tissue from more than one somatic cell type in contrast to dermoid cysts in which a single somatic cell type predominates (Rochat et al 1996). Germ cells give rise to gonadal cells (spermatogonia in testes and oogonia in ovaries) and consequently most teratomas originate from these gonads, especially ovaries in man and domestic animals (Norris et al 1969, Dehner et al 1970, Clark 1975, Gelberg and McEntee 1985, Greenlee and Patnaik 1985, Patnaik and Greenlee 1987, MacLachlan and Kennedy 2002, Sato et al 2003). Extragonadal teratomas are considered rare and usually occur in midline of the neck, brain and spine or retroperitoneum (Patnaik and Nafe 1980, Valentine et al 1988, Chenier et al 1998, Nagashima et al 2000, Lambrechts and Pearson 2001, Ferreira et al 2003). A condition of temporal teratoma has been reported in the horse, though the terms heterotropic polyodontia or dentigerous cyst are preferred (Gaughan and DeBowes 1993, DeBowes and Gaughan 1998).
Studies in mice have indicated that germ cells originate from the upper cell layers of the blastocyst and migrate to the caudal yolk sac endoderm (Glenn and Barkovich 1996). It is thought that failure of germ cells to migrate or their becoming entrapped with mesodermal cells during migration/formation of primitive neuroaxis results in the extragonadal location of some tumours (Glenn and Barkovich 1996). The contents of teratomas may be bizarre, reflecting their heterogeneity of germ-cell origin, and neural tissue, bone and cartilage, lens, hair follicles, sebaceous and apocrine glands, endocrine and adipose tissue have been reported in veterinary species (Chenier et al 1998, Miyoshi et al 2001, Patterson-Kane et al., 2001, Sato et al 2003, Yamaguchi et al 2004). The very densely packed hair clearly visible on gross sectioning of the mass in the present case made a dermoid cyst the principal macroscopic differential diagnosis. The presence of multiple somatic cell types on histological examination precluded this, however. Teratomas in domestic species are usually considered benign tumours though 32% of canine ovarian teratomas are reported to metastasise (Greenlee and Patnaik 1985).
The MRI changes, in conjunction with the history, were thought to represent a benign cystic mass. The lesion was well marginated with no invasion of adjacent structures and no changes in the adjacent tissues, both of which might be expected with a malignant mass or an infectious/inflammatory lesion. The thin uniform enhancing rim to the lesion and lack of enhancement of the central area are typical of a cystic lesion such as salivary mucocoele, lacrimal gland cyst or benign cystic tumour. The thin uniform rim would be unlikely to occur with a granuloma or abscess both of which might be expected to be more ill-defined and irregular. A heterogeneous centre within a cystic mass is helpful in differentiating complex cystic masses such as teratomas from clear cystic masses such as simple lacrimal gland cysts, epidermoid cysts and mucocoeles. The MR changes were similar to those reported for orbital teratomas and in man, however, there is overlap in the appearance of dermoids and teratomas (Smirniotopolos and Chiechi 1995). Dermoid cysts are unilocular and often contain significant amounts of lipid from sebaceous secretions, with a distinct fat/fluid level. Teratomas also often contain fatty tissue but are multiloculated with solid areas and complex intermingling of lipid and calcification (Smirniotopolos and Chiechi 1995). There are often areas of calcification or haemorrhage within teratomas but calcification of the rim of dermoids and epidermoids may be seen. The presence of significant amounts of fat within the lesion helps differentiate teratomas and dermoid cysts from epidermoid cysts which have water-like content (Smirniotopolos and Chiechi 1995, Ueno et al 2004). Adipose tissue was seen on histopathological examination of the mass but was not recognised on the MR images. The thin hyperintense rim on the T1W and T2W images was isointense to fat on both sequences and could have been interpreted as fat. However, the failure of the hyperintense rim to suppress on the fat-suppressed images and the absence of a chemical shift artefact (seen at fat/fluid interfaces) suggested that the hyperintense tissue was not solely fat. The T2* GRE sequence showed no evidence of haemorrhage or calcification. The MRI findings in this teratoma were unusual in that neither fat nor areas of calcification or haemorrhage were seen.
Retrobulbar teratoma should be considered an extremely rare cause of exophthalmos in cats and, to the authors' knowledge this is the first reported case of retrobulbar teratoma in this species.
