Abstract
This study evaluated thermal threshold (TT) testing for investigation into NSAID analgesia in cats. Seven cats participated in two crossover studies. TTs were measured on thoracic skin using a device developed specifically for cats. Skin temperature was recorded, then the heater activated. At the behavioural end point heating was stopped and temperature (=TT) recorded. In part 1, TTs were measured following subcutaneous (SC) ketoprofen or saline. In part 2, the process was repeated after intradermal kaolin induced mild inflammation at the test site. TTs were measured before treatment and two hourly for 24 h. In part 1, skin temperature did not change but in part 2 it increased more after saline than ketoprofen. TT did not change significantly after any treatment. However, after ketoprofen TT fell below the 95% confidence interval (CI) in part 1 and increased above it in part 2. The method detected some NSAID effects but is unlikely to be sufficiently sensitive for study of NSAID analgesia.
Pain management in cats has been a neglected subject until relatively recently. However, there are now numerous laboratory and clinical studies evaluating a number of analgesics in this species, and this has undoubtedly led to improved care of cats after trauma, when undergoing surgery and in chronic pain (Robertson and Taylor 2004, Taylor and Robertson 2004). In devising the most appropriate analgesic protocols it is essential that both laboratory and clinical investigations are undertaken. There are a number of clinical investigations evaluating both non steroidal anti inflammatory drugs (NSAIDs) and opioids in cats (Lascelles et al 1995, Balmer et al 1998, Slingsby and Waterman-Pearson 1998, 2000, 2002). However, few of the clinical studies have placebo control or allow comparison of duration or dose; such investigation requires laboratory conditions. Opioid analgesia has been successfully studied using acute analgesiometry, and the thermal threshold (TT) testing device reported by Dixon et al (2002) has been widely used to evaluate a number of opioids in cats (Robertson et al 2003, 2005a,b, Lascelles and Robertson 2004a,b). Data from such studies have contributed towards appropriate use of buprenorphine, morphine and butorphanol in clinical pain management in cats (Robertson and Taylor 2004). However, it is well recognised that it is much more difficult to detect NSAID analgesia, as this requires the presence of inflammatory pain. Acute analgesiometric devices do not cause tissue trauma or inflammation and are unsuitable for NSAID investigations (Dobromylskyj et al 2000).
The aim of this study was to adapt the feline thermal analgesiometry method previously employed with opioids (Dixon et al 2002, Robertson et al 2003) for use with NSAIDs. Mild inflammatory pain was produced at the testing site by intradermal injection of an irritant, as previously described in the study of some other pharmacological effects of NSAIDs (Taylor et al 1996, Lees et al 2003). Ketoprofen was chosen as the test NSAID in this investigation as it holds UK Market Authorisation for use in cats, and is a COX1 inhibitor (Lees et al 2003). The investigation was carried out under Home Office Licence PPL/80/00083. Some of these data were presented to the Association of Veterinary Anaesthetists (Taylor et al 2003).
Materials and methods
Animals
Seven healthy female adult cats (three neutered, four entire) weighing 2.2–5.4 kg were studied. They were group housed and fed dry laboratory cat food ad libitum supplemented with tinned food once daily. Water was always available. The cats underwent routine health checks, including a full haematological and biochemical examination every 6 months. On the day preceding any test two cats were moved into cages in the laboratory where they were housed singly. The cat's thorax was clipped in preparation for thermal testing. The cages were equipped with a cat bed, litter tray, toys, wall mirrors, and ad libitum water. Dry food was supplied ad libitum after the first post-treatment testing, and tinned food was given as treats throughout the day after tests.
Treatment and testing
Two randomised crossover studies (part 1, six cats and part 2, seven cats) were carried out with at least 2 weeks between each treatment in any one cat. TTs were measured using the TT testing device previously developed for cats (Dixon et al 2002). A heater element and temperature sensor in a small probe were held at constant pressure against the cats' shaved thorax with an elasticated band. Skin temperature was recorded before each test, and then the heater was activated. When the cat responded by flinching, turning or jumping the heater was turned off and the temperature recorded as the TT. A safety cut-out occurred at 60°C if the heating had not already been stopped.
The side of the thorax used for placement of the thermal probe was selected randomly (coin toss) on the first occasion and the contralateral side used the second time.
Before any treatments were given, the band was placed around the thorax and the cat was left quietly in the cage for 15–30 min whilst the probe temperature equilibrated with skin temperature. Four baseline pre-treatment TTs were recorded at 15 min intervals, then the treatment was given at time 0. In part 1, TTs were measured following subcutaneous (SC) injection of ketoprofen (ketofen 1%; Merial Animal Health UK) (2 mg/kg) or a similar volume of saline. In part 2, after the baseline readings had been taken, the cats were anaesthetised with isoflurane in oxygen using an induction chamber followed by a mask and T-piece breathing system. During anaesthesia, a 1.5 cm2 was marked on the cat's thorax with indelible ink at the site of the thermal probe. Mild inflammation was produced in the marked area using five intradermal injections of 5 mg kaolin in 0.1 ml saline at each corner and in the centre of the square. The band with the thermal probe was then placed around the thorax, positioning the probe directly over the injected square area. Saline or ketoprofen as in the first study was injected immediately after band placement (time 0). The cats were then returned to their cage and allowed to recover from anaesthesia. All were awake and behaving normally within 20 min. TTs were measured at 2 h and then two hourly for a further 24 h. All testing was performed by one individual who did not know whether saline or ketoprofen had been given.
Statistical analysis
Data within each of the four treatment groups (ketoprofen and saline in part 1 and part 2) were analysed for changes with time by one-way analysis of variance (ANOVA) for repeated measures followed by Dunnett's test, if appropriate. In both part 1 and part 2 the effects of saline and ketoprofen treatment were compared using repeated measures two-way ANOVA and a Bonferroni post-test (Graphpad Prism software, version 4.00, Graphpad Software Inc., San Diego, California, USA). P<0.05 was regarded as significant. TTs were further analysed by comparison of post-treatment thresholds with a reference range taken from the saline group in part 1 and from the pre-treatment control baselines in part 2 (Dixon et al 2002, 2007). The reference range was generated by taking the mean TT for each cat and using these means to generate 95% confidence intervals (CI) for TT in untreated cats. Thresholds after any treatment lying above the upper 95% CI were considered to indicate hypoalgesia (decreased pain), and those below to indicate hyperalgesia (increased pain).
Results
In the part 1 group mean skin temperature did not change significantly in either the saline or ketoprofen group throughout the study, remaining between 36 and 37°C, and there were no significant differences between the groups (Fig 1). Mean TT did not change significantly in either group, remaining around 39–41°C, and the groups were not significantly different. However, after ketoprofen, TT fell below the lower 95% CI after 8 h (Fig 2).
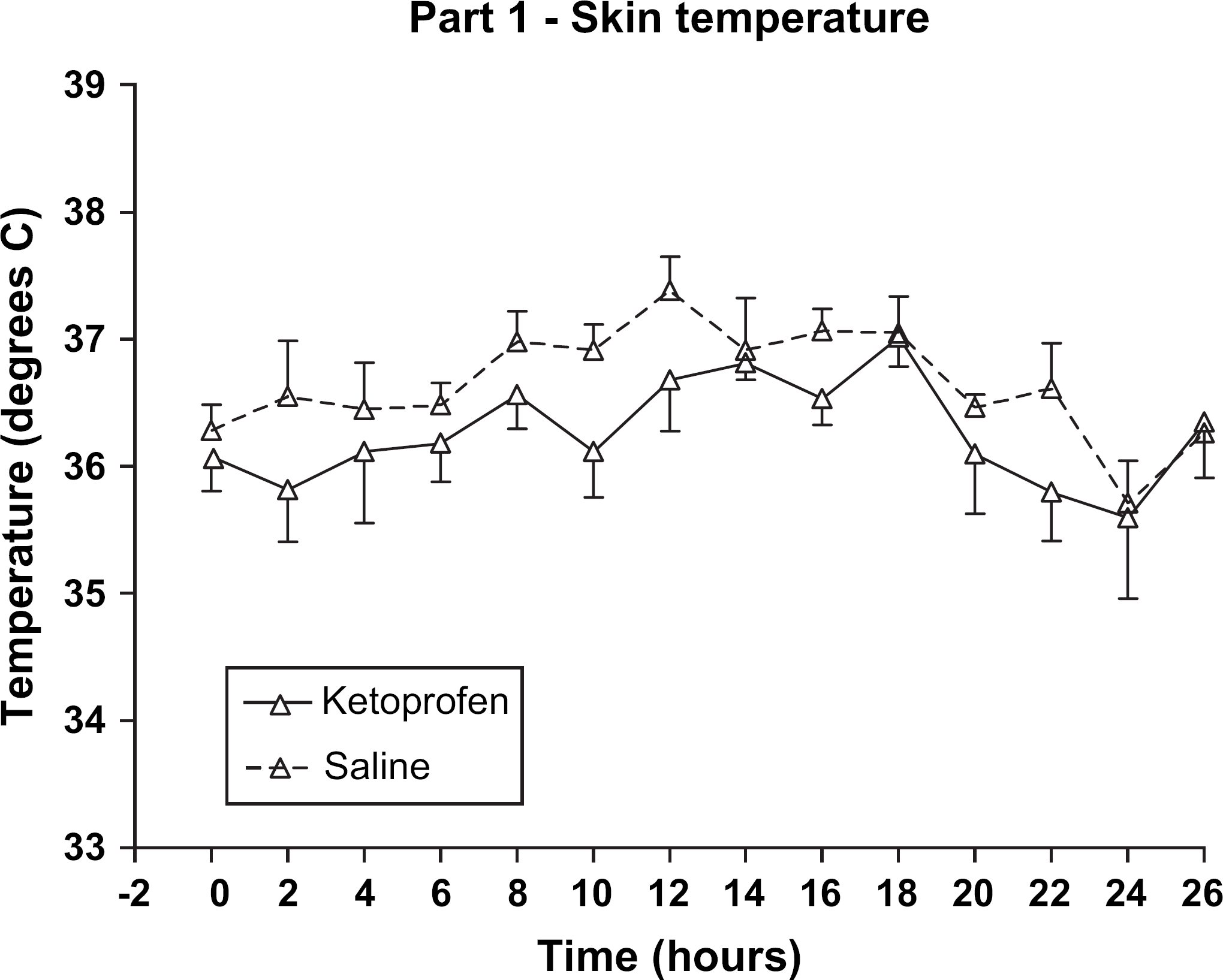
Part 1 (without inflammation). Skin temperature after subcutaneous saline or ketoprofen in six cats. Treatment given at time 0. Reading at time 0=mean of four pre-treatment measurements. No significant difference between saline and ketoprofen groups. No significant changes with time.

Part 1 (without inflammation). Thermal threshold temperature after subcutaneous saline or ketoprofen in six cats. Treatment given at time 0. Reading at time 0=mean of four pre-treatment measurements. 95% CI from means from each cat after saline. No significant difference between saline and ketoprofen groups. No significant changes with time.
In part 2 mean skin temperature increased 12, 24 and 26 h in the saline group (37.3±0.5–38.1±0.6, 38.0±0.8 and 38.1±0.8°C, respectively, P<0.05) and at 24 and 26 h in the ketoprofen group (37.4±0.3–38.5±1.1 and 38.5±1.2°C, respectively, P<0.05) (Fig 3). TT did not change significantly from pre-treatment baseline values in either the saline or ketoprofen group, remaining between 41 and 45°C. However, in the ketoprofen group, TT increased above the upper 95% CI after 18 h (Fig 4).

Part 2 (with inflammation). Skin temperature after subcutaneous saline or ketoprofen in six cats. Treatment given at time 0 immediately after kaolin injection. Reading at time 0=mean of four measurements before kaolin injection. No significant difference between saline and ketoprofen groups. *Indicates significant increase above pre-kaolin temperature (as at time 0).

Part 1 (with inflammation). Thermal threshold temperature after subcutaneous saline or ketoprofen in six cats. Treatment given at time 0 immediately after kaolin injection. Reading at time 0=mean of four measurements before kaolin injection. 95% CI from means of each cat's baseline readings before kaolin injection. No significant difference between saline and ketoprofen groups. No significant changes with time.
There were clear signs of inflammation at the treated site, with reddened skin, mild oedema and sensitivity to digital pressure for up to 36 h after the kaolin injection.
Discussion
In this investigation, intradermal kaolin clearly produced mild inflammation for 1–2 days. However, there was no clear increase in sensitivity to thermal stimulation. The method detected some effects of a COX1 selective NSAID in that ketoprofen produced a degree of hypoalgesia from 18 h after treatment. However, the effect was limited, and no hyperalgesia was detected in the saline placebo group. Our goal of producing a mild inflammatory focus which did not cause distress or any lasting harm to the cats was achieved. However, thermal stimulus proved unsuitable for detection of hypo- or hyperalgesia to an extent that may be useful for future investigation into NSAID analgesia in cats.
Steagall et al (2006b) reported that carprofen prevented hyperalgesia to a pressure stimulus in a similar feline inflammatory model. However, it did not produce hypoalgesia, in contrast with the limited effect of ketoprofen in the present study. This may reflect activation of different receptors, or the clearer COX1 inhibitory action of ketoprofen compared with carprofen (Lees et al 2003).
The decrease in TT in the cats given ketoprofen in part 1, with no inflammatory focus, is unexplained. Although there were no obvious signs of irritation, it is conceivable that the ketoprofen injection vehicle caused a slight effect in comparison with saline. The SC injection was a few centimetres anterior to the dorsal part of the thoracic band which may conceivably have exacerbated any irritant effect.
Baseline skin temperature and TT were higher in part 2 than part 1. However, delta T, the difference between skin temperature and TT, was similar. The difference between parts 1 and 2 probably reflects the use of a different group of cats, with the inclusion of an extra cat in part 2. However, part 1 and part 2 took place during different months of the year, with different ambient temperature, which may have affected skin and threshold temperatures. As each of part 1 and part 2 were crossover studies, the differences in baseline do not affect the results.
A humane system for evaluation of NSAID analgesics in cats requires mild inflammation causing little distress and no lasting harm. In addition, any method for nociceptive evaluation for use in the cat requires considerable attention to the unique temperament of this species, and we consider it particularly important to avoid any physical restraint so that behaviour is unaffected (Dixon et al 2002). Giraudel et al (2005a,b) described a technique for examining NSAID effects in cats, where a much greater degree of inflammation was induced with kaolin. However, this method caused considerably more tissue damage and, although providing excellent conditions for study of the anti-inflammatory effects of an NSAID, still did not allow objective assessment of pain in the unrestrained cat. Both these techniques described in cats are considerably more acceptable than older methods employing Freund's adjuvant-induced or septic arthritis which have been used in rats and horses, respectively (Lloyd et al 1990, Levy et al 2006).
In view of the obvious signs of inflammation and increased sensitivity to touch reported here, it is clear that intradermal kaolin was an effective means of producing a site of mild inflammation. Kaolin was chosen over the other most commonly used option, carageenin, as this produced insufficient effect at 0.033% and 0.100% (Taylor et al 1996). Higher concentrations of carageenin produced a hard nodule at the site of injection, and topical phorbol 12-myristate 13-acetate caused too much tissue damage (unpublished observations), so were also considered unsuitable. Giraudel et al (2005a) successfully used 500 mg SC kaolin injection to inflame the whole paw in their investigations into NSAID pharmacodynamics. Kaolin appeared to be a suitable irritant in cats and much smaller doses (25 mg) were sufficient for our model.
The TT testing system has proved robust for studies of opioid analgesia in cats (Robertson et al 2003, 2005a,b, Lascelles and Robertson 2004a,b) and it was a logical step to attempt to adapt the system for study of inflammatory pain and NSAID analgesia. However, neural processing of nociceptive stimuli may not necessarily be the same for all nociceptors, which include mechanical, thermal and chemical receptors. Most nociceptors respond to pressure and thermal stimulation but have different thresholds of activation (Raffe 1997). Ley et al (1989) reported mechanical but not thermal sensitisation in sheep with footrot. They considered that this may have been due to processing of thermal stimuli at a supraspinal level, whereas mechanical processing occurred at spinal and peripheral levels. Supraspinal processing of thermal stimuli in the sheep was due at least to the site of the thermal test on the pinna of the ear, from which nociceptive input travels to the facial nucleus. However, Omote et al (2005) also demonstrated a difference between thermal and mechanical sensitisation in their model of postoperative pain in rats. They showed that intrathecal prostaglandin (PG)E2 subtype EP1 receptor antagonism prevented mechanical but not thermal sensitisation, again indicating that mechanical and thermal sensitisation is produced through different mechanisms. Hence, it is not surprising that, where inflammation and hyperalgesia is required, as in a system for study of NSAIDs, one nociceptive stimulus may be better than another. It is likely that a mechanical test would be more appropriate.
A further possible reason for the limited ability of thermal testing to detect inflammatory hyperalgesia may be a result of oedema at the test site effectively insulating the nociceptors from the heat stimulus. We have previously noted how inadequate contact between the skin and thermal probe prevents accurate detection of a TT (Dixon et al 2002). This has generally been due to inadequate shaving of the hair or to low pressure in the bladder that holds the probe against the skin under the thoracic band. It is conceivable that the oedema which developed might have a similar effect.
In conclusion, the method described in this paper appears to fulfil the role of a humane means of producing a site of mild inflammation of a few days duration in cats. The test site was subjectively sensitive to digital pressure without causing undue distress or lasting harm to the cats. However, thermal sensitisation was not detected, making this inappropriate as a means of investigating NSAID analgesia. The apparent sensitivity to touch as well as reports such as that by Ley et al (1989), demonstrating mechanical but not thermal sensitisation in sheep with footrot, suggest that a mechanical test should be evaluated in cats. This is the subject of further investigations reported elsewhere (Steagall et al in press, 2006, Dixon et al 2007).
Footnotes
Acknowledgement
Thanks go to Feline Advisory Bureau who funded the project.
