Abstract
Spinal epidural empyema (SEE) represents a severe pyogenic infection of the epidural space. Clinical signs of the disease are non-specific – increased body temperature, intense neck pain, neurological signs of a transverse myelopathy – and can lead to severe and permanent neurological deficits. This report describes the diagnosis and successful surgical treatment of cervical SEE secondary to grass awn migration in a cat. Although it is uncommon, this disease should be suspected in cats with progressive myelopathy. Early diagnosis and emergency surgery combined with antibiotic therapy are required to allow a complete recovery.
A 2-year-old female neutered domestic shorthaired cat was referred to the National Veterinary School of Alfort with a 3-week history of intense neck pain that had progressed to tetraplegia 3 days before admission. Lateral cervical spinal radiographs taken by the referring veterinarian were normal and a course with meloxicam (Metacam; Boehringer; 0.1 mg/kg, PO, q24h) for a week was instructed. However, as the cat developed forelimb ataxia, the meloxicam was replaced with prednisone (Microsolone; Merial; 0.5 mg/kg, PO, q24h) for a week. Although the neck pain was partially controlled, the cat developed tetraparesis and was referred.
Initial neurological examination showed tetraplegia with rigidity of the forelimbs, absence of neck muscle tone and mild neck pain. Spinal and cranial nerve reflexes and deep pain perception were normal in all four limbs. A spinal cord lesion between the first and fifth cervical vertebrae (C1–C5) was suspected.
On the same day, the cat underwent a computed tomography (CT: Scanner Hispeed CT/e Plus, General Electric Medical Systems, Milwaukee, WI, USA) examination performed under general anaesthesia induced with propofol (Rapinovet; Schering-Plough; 6.5 mg/kg, IV) and maintained with inhaled halothane (Fluothane; Zeneca Pharma) and oxygen. Helical CT of the cervical spine (1 mm sections) pre- and post-contrast injection (sodium and meglumine ioxitalamate (Télébrix 35, sodium and meglumine ioxitalamate, 350 mg of iodine/ml, Laboratoire Guerbet, Roissy-Charles-de-Gaulle, France), 2 ml/kg, IV) was performed. The CT examination revealed a poorly defined and contrast-enhancing lesion extending into the left paravertebral musculature from the lateral aspect of C2 vertebral body. The lesion entered the spinal canal at the level of the left caudal articular facet of C3 and was causing lateral spinal cord compression (Fig 1a–d). The lesion was also localised along the left dorsal paravertebral musculature of C4 and on the lateral aspect of the C4 spinal process (Fig 1e and f). Magnetic resonance imaging (MRI) was performed under the same anaesthesia, using a low-field MRI unit (Signa Profile, General Electric Medical Systems, Milwaukee, WI, USA) which incorporated an open 0.2 T permanent magnet. Cervical sagittal (3 mm sections with a 0.3 mm gap) T2-weighted images and post contrast (gadoteric acid (Dotarem 0.5 mmol/ml, gadoteric acid, 27.93 g/100 ml, Laboratoire Guerbet, Roissy-Charles-de-Gaulle, France) 0.1 mmol/kg, IV) sagittal and transverse (4 mm sections with a 0.4 mm gap) T1-weighted images were acquired. Post contrast transverse T1-weighted images confirmed CT findings (Fig 2a and b). The spinal cord signal was hyperintense at the level of C3 on sagittal T2-weighted images (Fig 2d), and isointense on sagittal T1-weighted images, which was compatible with oedema of the spinal cord (Fig 2c and d). The para-sagittal images revealed two lesions extending from the C2 to C4 paravertebral musculature (Fig 2f). The centre of the lesions was hyperintense on T2-weighted images and slightly enhanced with gadolinium on sagittal T1-weighted images (Fig 2e and f).
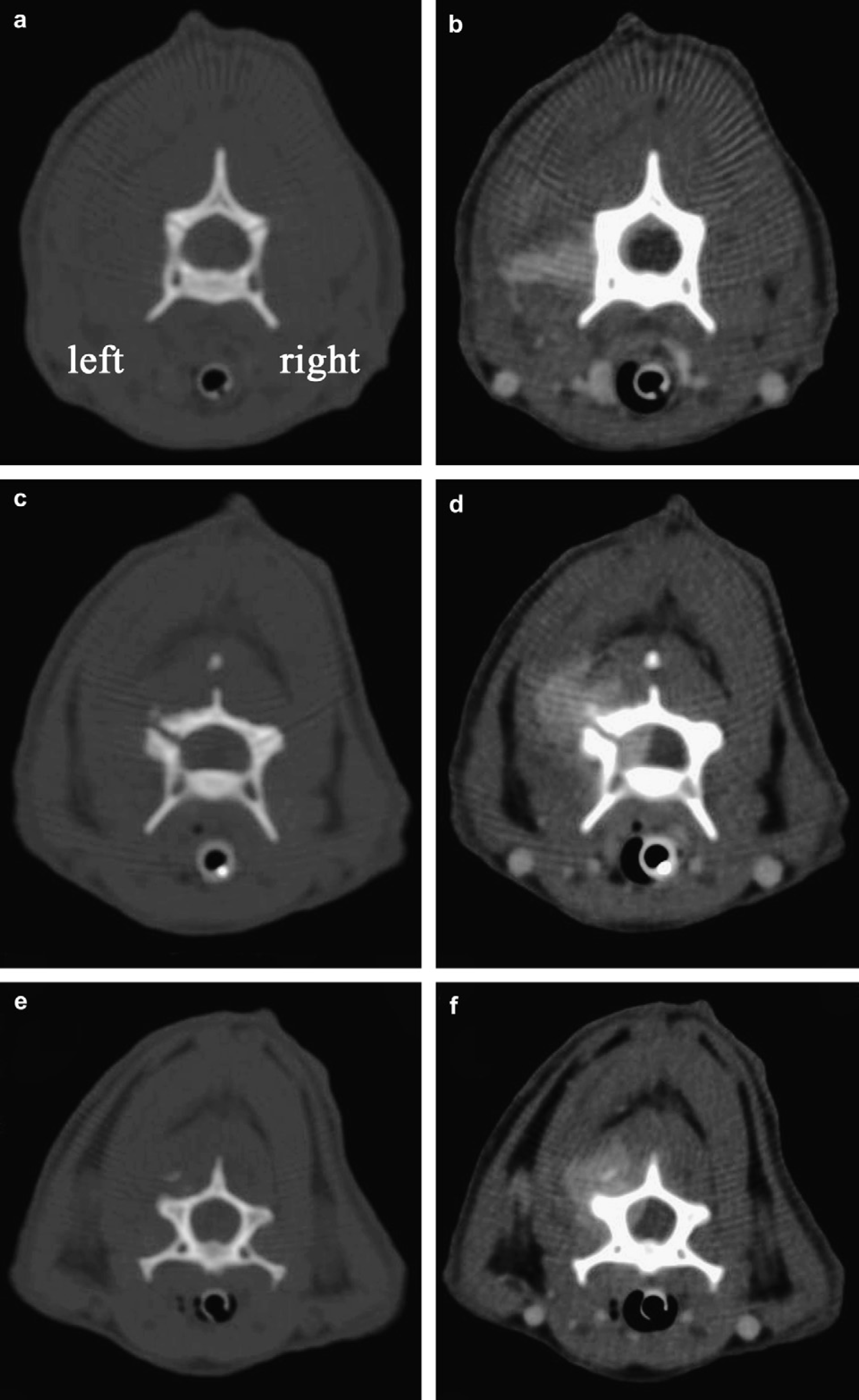
Transverse computed tomography images without contrast (left images, bone window) and with contrast (right images, soft tissue window), paired at the level of C2 (a, b), C3 (c, d) and C4 (e, f). A poorly defined and contrast-enhanced lesion extends into the left paravertebral musculature from the lateral aspect of C2 vertebral body (b) and enters the spinal canal at the level of C3 caudal articular facet which appeared distended (d). Spinal cord compression is obvious (d). The lesion is also localised along the dorsal paravertebral musculature of C4, on the lateral aspect of C4 spinous process (f).

Post contrast transverse T1-weighted magnetic resonance images (a, b) at the level of C2 and C3 caudal articular facet; paired sagittal T2-weighted and post contrast T1-weighted magnetic resonance images of the median cervical region (c, d) and left paravertebral musculature (e, f). A poorly defined gadolinium enhanced lesion is present in the left paravertebral musculature, lateral to C2 vertebral body (a), entering the spinal canal at the level of the C3 caudal articular facet and leading to spinal cord compression (b). The spinal cord signal is hyperintense at C3 level, T2-weighted image (d, arrowhead), and isointense on T1-weighted image (c). On para-sagittal images, two lesions extend from C2 to C4 paravertebral musculature (dorsal musculature: 1.8×0.6 cm and lateral musculature: 1.7×0.4 cm) (f, asterisk). The centre of the lesions is hyperintense on T2-weighted image and slightly enhanced with gadolinium on sagittal T1-weighted image (e).
Differential diagnosis included inflammatory, infectious or neoplastic diseases (lymphosarcoma, vertebral column or extradural non-vertebral column neoplasia).
Cerebrospinal fluid (CSF) was not examined. The following day, the cat underwent decompressive surgery.
Pre-operative physical examination showed elevated body temperature (39.3°C). While preparing the surgical field, a scar on the skin was found on the left lateral aspect of the neck near the C3–C4 region.
The lesion was surgically treated via a dorso-lateral approach to the cervical vertebrae. The dissection of the paravertebral muscles revealed a hard structure around the left C3 caudal articular facet. The fibrous structure was ruptured and revealed a purulent exudate. During aspiration, a grass awn fragment was found. Further dissection revealed that the inflammatory tissue adhered to the C3–C4 intervertebral foramen. Hemilaminectomy by pneumatic drilling was performed. Once in the epidural space, it was possible to visualise and remove necrotic fat and purulent material which were compressing the spinal cord. Macroscopic examination of the spinal cord showed an inflamed but unruptured dura mater. The surgical field was flushed with copious amounts of sterile saline solution and routinely closed.
As the cat's respiratory function was depressed, it was ventilated during the surgical procedure and for 1 h after withdrawal of anaesthetic gases. At recovery, the body temperature was 40.2°C. The cat was placed in the intensive care unit for complete recovery from anaesthesia.
Tissue samples collected, from around the left caudal articular facet of C3, from C3–C4 intervertebral foramen and from the epidural material, were submitted for histological examination and aero/anaerobic bacterial culture and sensitivity. Histopathology revealed necrotising suppurative myositis. Pasteurella multocida was isolated and showed sensitivity to ampicillin, cephalexin, trimethoprim and marbofloxacin.
Postoperative medical treatment consisted of intravenous cephalexin (Rilexine; Virbac; 30 mg/kg, q12h) and marbofloxacin (Marbocyl; Vétoquinol; 2 mg/kg, q12h) for 2 days continued orally for 3 weeks after bacterial culture results, prednisone (Microsolone; Merial; 0.25 mg/kg, q12h) for 5 days, transdermal fentanyl patch (Durogesic; Janssen-Cilag; 3 μg/kg) and supportive care.
Voluntary motor activity returned in the hindlimbs on the second day after surgery and in the forelimbs 3 days later. Ten days after surgery, the cat was able to support its weight with assistance. At 1 month, the cat was fully ambulatory.
Scratching erosive superficial dermatitis occurred 3 months after surgery. The lesion was surgically excised. Histopathology revealed a chronic, non-specific epidermal wound. At this time, neurological examination was normal.
Spinal epidural empyema (SEE) represents a severe pyogenic infection of the epidural space. The terminology ‘empyema’ describes the accumulation of purulent exudates in a natural cavity and was preferred over ‘abscess’ in the text. Baroni et al (1995) reported a case of cerebral abscess in a cat due to grass awn migration from the nasal cavity. To the authors' knowledge, SEE has never been reported in cats. There are several case reports of SEE in dogs, originating from foreign bodies (grass awns and wooden sticks), bite wounds, suspected haematogenous spread, iatrogenic contamination or of unknown origin (Brockman and Trout 1991, Jacob et al 1996, Remedios et al 1996, Dewey et al 1998, Jerram and Dewey 1998, Cherrone et al 2002, Leskovar et al 2003, Nykamp et al 2003, Naughton et al 2005, Schmiedt and Thomas 2005, Lavely et al 2006). In a retrospective study of 182 animals, only eight were cats with grass awn migration in the conjunctiva or nictitating membrane (seven cats) and urinary bladder lumen (one cat) (Brennan and Ihrke 1983). The nasopharyngeal region is an other described site of grass awn migration (Riley 1993).
Grass awns are covered with pointed spikelets and florets which easily stick to the animal's coat and can penetrate the skin (Horne 1981). Their barbed aspect allows migration facilitated by body motion. In this case report, a cervical cutaneous scar appeared to indicate the point of penetration of the foreign body.
Clinical signs resulting of SEE from grass awn migration are non-specific. Fever, pain and hyperaesthesia are characteristic findings in dogs and humans with SEE, but are not constant (Reihsaus et al 2000, Lavely et al 2006). In the presented case, cervical pain and hyperaesthesia were obvious. Only mild elevation of body temperature was noted before surgery.
In human medicine, the diagnosis of SEE is often delayed because of its rarity (Reihsaus et al 2000). Laboratory findings can show non-specific leukocytosis. CSF examination can reveal elevated protein concentration and neutrophilic pleiocytosis (Dewey et al 1998, Reihsaus et al 2000, Cherrone et al 2002, Schmiedt and Thomas 2005, Lavely et al 2006). Only four case reports of SEE in dogs had a CSF analysis (Dewey et al 1998, Cherrone et al 2002, Schmiedt and Thomas 2005). Microorganisms could not be isolated in these reports, probably because of the small amounts of bacteria present in the CSF and the localisation of the abscess, although CSF abnormalities were consistent with bacterial infection (pleocytosis with a predominance of neutrophils and elevated proteins) (Radaelli and Platt 2002). Lumbar puncture is no longer recommended in human medicine because of the risk of inducing bacterial meningitis by spread of the bacteria into the subarachnoid space (Reihsaus et al 2000).
Conventional radiography is not reliable in the diagnosis of SEE. Sclerosis and osteolysis of vertebral body end-plates may take several weeks to appear despite spinal pain, as described in diskospondylitis (Reihsaus et al 2000, Dewey and Coates 2003). In contrast, CT scan and MRI are the modalities of choice in the diagnosis of SEE (Reihsaus et al 2000, Cherrone et al 2002, Nykamp et al 2003, Naughton et al 2005). MRI, compared to CT scan, is able to form multiplanar images and scan deep structures without bone artefacts (Smith and Blaser 1991, Young et al 2004, Naughton et al 2005). In this case report, sagittal images allowed the delineation of the paraspinal lesions and led us to speculate spinal cord oedema.
Surgery is the treatment of choice for SEE because it allows decompression of the spine, drainage of the abscess and bacterial isolation. Pasteurella multocida, Streptococcus species, Staphylococcus species and Actinomyces species have been isolated from dogs and cats with foreign body migration (Brennan and Ihrke 1983, Dewey et al 1998, Nykamp et al 2003, Naughton et al 2005, Schmiedt and Thomas 2005).
Anaesthesia for cervical spinal canal surgeries carry a high risk of severe respiratory depression because the neurones in the medullary respiratory centre descend in the cervical spinal cord to project on motoneurones forming the phrenic nerves and the nerves controlling the intercostal muscles (Kapatkin and Vite 2000).
SEE requires early diagnosis and is a neurosurgical emergency (Kapatkin and Vite 2000). The severity and the duration of the neurological signs greatly influence survival and long term outcome. Consequently, the prognosis for human patients is considered guarded. However, between 1950 and 1990, a continual reduction in mortality from 34% to 16% has been observed (Reihsaus et al 2000). In dogs, recent reports have also demonstrated good survival and neurological outcome after decompressive surgery combined with antibiotic therapy (Brockman and Trout 1991, Jerram and Dewey 1998, Cherrone et al 2002, Schmiedt and Thomas 2005, Lavely et al 2006).
SEE is a challenging diagnosis for the clinician because it is an uncommon condition. Among the differential diagnosis, SEE was unlikely compared to inflammatory or neoplastic diseases of the spinal cord in cats (Mellanby et al 2003, Marioni-Henry et al 2004). In this case report, our aim was to enhance the recognition of SEE, which should be included in the differential diagnosis in cats with progressive myelopathy. Early diagnosis and surgical decompression, abscess drainage and antibiotic therapy can lead to complete recovery.
