Abstract
Faecal samples from 111 cats with diarrhoea that were living in the UK were submitted for the assessment of Tritrichomonas foetus infection by polymerase chain reaction (PCR). Sixteen (14.4%) samples were found to be positive. In agreement with studies from the USA, infected cats were predominantly of a year of age or less and of a pedigree breed, with Siamese and Bengal cats specifically over-represented in this population.
Tritrichomonas foetus is a flagellated protozoan parasite that has recently been identified as a natural and experimental cause of large bowel diarrhoea in domestic cats, particularly in the USA (Gookin et al 1999, 2001, 2004, Levy et al 2003, Foster et al 2004). Studies have demonstrated that T foetus can colonise the feline ileum, caecum and colon (Gookin et al 2001), and cause mild to moderate lymphoplasmacytic and neutrophilic colitis (Foster et al 2004, Yaeger and Gookin 2005). Clinical signs of infection can be variable, ranging from asymptomatic infection to prolonged and intractable diarrhoea which in some cases may wax and wane (Gookin et al 1999, 2001, 2004, Foster et al 2004). The predominantly large bowel diarrhoea typically presents with increased frequency of defecation, the passage of semi-formed to liquid often foul-smelling faeces, sometimes containing fresh blood and mucus (Gookin et al 1999). With severe diarrhoea the anus may become inflamed and painful (Gookin et al 1999, Mardell and Sparkes 2006), and faecal incontinence may be present (Gookin et al 1999). However, although the diarrhoea may be persistent and severe, most affected cats are otherwise well, and they rarely show significant weight loss (Gookin et al 1999).
Assessment of faeces for the presence of T foetus can be made using a number of different methods (Gookin et al 2004): (i) looking for motile trophozoites in fresh faecal smears with saline dilution, (ii) using the specific ‘In Pouch TF’ culture system (BioMed Diagnostics, Oregon, USA) (Gookin et al 2003a) or (iii) by detection of T foetus ribosomal DNA using polymerase chain reaction (PCR) (College of Veterinary Medicine, North Caroline State University, USA) (Gookin et al 2002). The different methods have differing sensitivities: in one study direct smears were positive in 5/36 cases, ‘In Pouch TF’ in 20/36, and PCR in 34/36 cases (Gookin et al 2004). These methods are needed because on routine faecal floatation examinations motile trichomonads are damaged by osmotic lysis and, therefore, are not observed.
Infection with T foetus is common in some groups of cats in the USA. While it has not been found in the faeces of feral cats or healthy indoor cats living in colonies that do not suffer from diarrhoea (Gookin et al 1999), it is frequently seen in households containing young pedigree cats, especially those where diarrhoea is common, in young cats from rescue centres, and in cats from other multi-cat households (Gookin et al 1999, 2004, Foster et al 2004). Indeed, in one study, 31% of cats at a cat show were identified as infected, suggesting that this organism may be an important and common cause of diarrhoea in cats in America (Gookin et al 2004). While cats of all ages can become infected, and may develop diarrhoea, clinical disease is seen most commonly in young cats and kittens, and the majority of cats with T foetus-associated diarrhoea are under 12 months of age (Gookin et al 1999).
Current information suggests that the long-term prognosis for infected cats is good, as most will eventually overcome the infection. However, this can be a slow process, taking from 2 months to 3 years, with a median of 9 months (Gookin et al 1999, Foster et al 2004). Most infected cats continue to shed low levels of the organism in their faeces for many months after the resolution of the diarrhoea (Gookin et al 1999, Foster et al 2004). These cats, and clinically affected cats, appear to be the major reservoir of infection to other cats. This is believed to occur via faecal–oral spread as there is no evidence of spread from other species, or spread via food or water (Gookin et al 2004). Currently, therapeutic interventions to reduce clinical signs and stop protozoal shedding are limited; many medications (including metronidazole and fenbendazole) are poorly effective (Gookin et al 1999, 2004, Foster et al 2004), and although recent work has indicated that treatment with ronidazole may be successful (Gookin et al 2006) there is currently no licensed form of this product in the UK, and its administration to cats can result in adverse reactions (typically lethargy, ataxia, tremors and, occasionally, seizures (Gookin personal communication 2006; author observations)).
To date, most reports of feline T foetus infection have come from the USA. However, it is likely that the organism has a much wider distribution. It has been found in the faeces of cats from Germany that attended an international cat show (Gookin et al 2003b), studies from the early 1900s indicated that the infection may have been present in Brazil, France, Yugoslavia, and China (summarised in Gookin et al 1999), and a single case report from the UK has recently been published (Mardell and Sparkes 2006). As there are no published studies on the prevalence of this infection in the UK the aims of the current study were to determine the frequency of occurrence of T foetus in the faeces of cats in this country, and to assess the age, sex or breed of infected cats.
Materials and methods
Between January and November 2006, 111 naturally voided diarrhoeic feline faecal samples were submitted by post to Capital Diagnostics (Capital Diagnostics, SAC Veterinary Science Division, Bush Estate, Penicuik, Midlothian EH26 0QE). The samples came from cats living throughout the UK and they were submitted specifically for the T foetus PCR assay. Information relating to the cat's age, gender and breed was obtained from their submission forms. The faecal samples were assessed for the presence of T foetus DNA using PCR methodology (processing, primers and controls as described by Gookin et al 2002). Categorical data were tested by construction of multiple two-by-two contingency tables and the use of an online two-tailed Fisher's exact test (http://www.exactoid.com/fisher/index.php).
Results
Of the 111 faecal samples, 16 (14%) were found to be positive for T foetus DNA. Table 1 shows the results in relation to the cats' age, sex and breed; the legend provides details of missing data.
The results of the study in relation to the cats' age, sex and breed
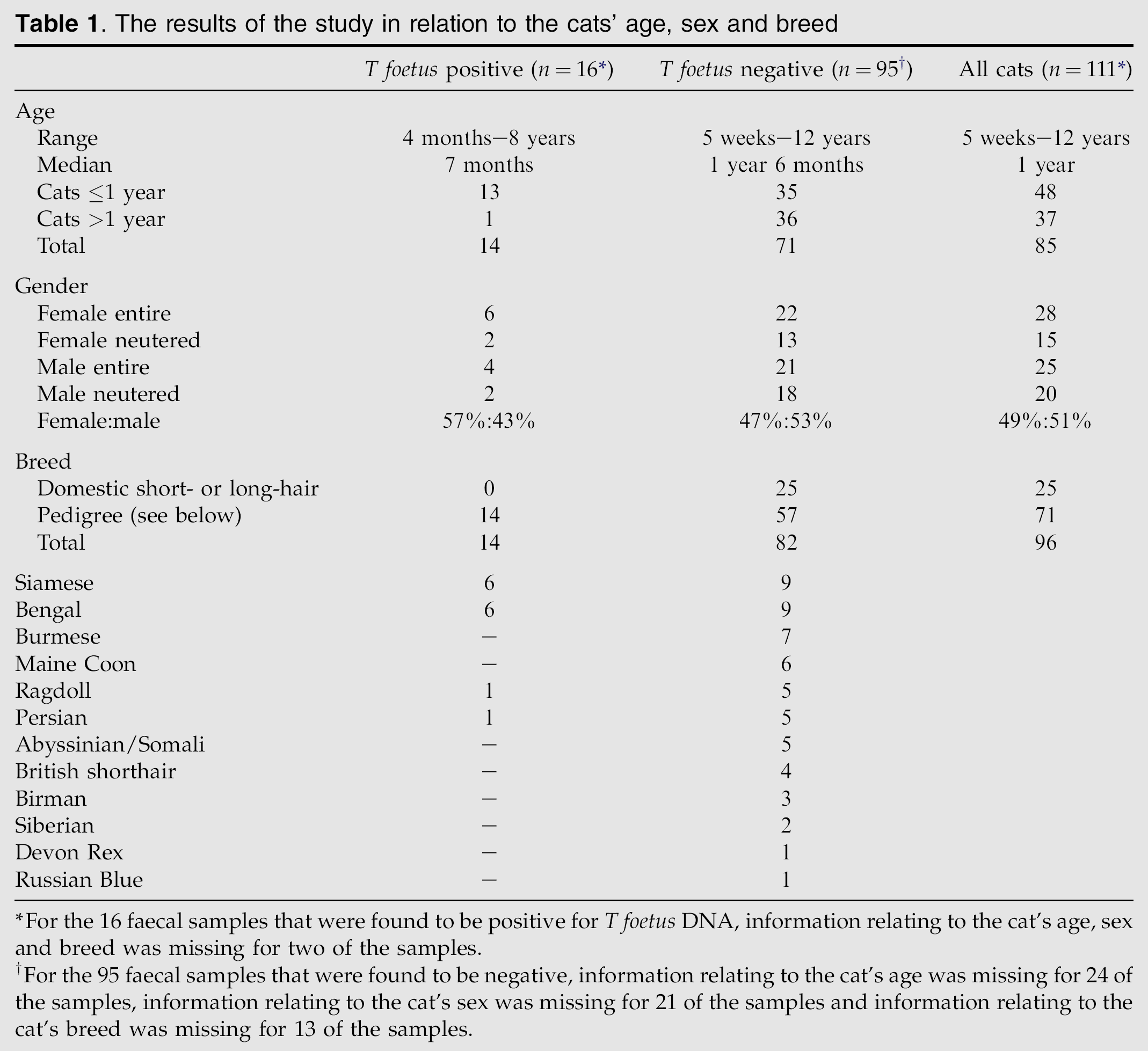
For the 16 faecal samples that were found to be positive for T foetus DNA, information relating to the cat's age, sex and breed was missing for two of the samples.
For the 95 faecal samples that were found to be negative, information relating to the cat's age was missing for 24 of the samples, information relating to the cat's sex was missing for 21 of the samples and information relating to the cat's breed was missing for 13 of the samples.
Significantly more of the T foetus-infected (positive) cats were of a year of age or younger (13/48 cats ≤1 year vs 1/37 cats >1 year; P=0.0026). Pedigree cats were significantly more likely to be PCR-positive than domestic crossbred cats (14/71 vs 0/25; P=0.018). Furthermore, when the infected cohort of cats was compared to the negative cats both Siamese and Bengal breeds of cat were over-represented (6/15 of that respective breed vs 8/96; P=0.0077). Most of the cats were of a pedigree breed, including all of the infected ones. The gender ratio of the two groups was similar.
Discussion
The current study shows that T foetus infection is common in cats with refractory diarrhoea in the UK, with 14.4% of samples submitted for assessment being found to be positive. This finding is similar to those from the USA, although in the USA the percentage of cats infected may be even higher, as 31% of faecal samples collected at a cat show were found to be positive using the same PCR (Gookin et al 2004). However, as those samples were not collected in the same manner to the samples in the current study, it is not possible to determine whether the prevalence of infection gained from the cat show was particularly high because it only contained pedigree cats or if it could actually be low as it only contained samples from cats that were well enough to attend the show, as such cats were most unlikely to have refractory diarrhoea. In comparison, the cats in the current study were mainly, but not entirely of pedigree breeds and all had significant diarrhoea. If the frequency of infection in the current study is calculated for only the pedigree cats (Table 1: n=14 infected cats and 57 negative ones) it rises to 19.7%, supporting the American finding that pedigree cats are more often infected than non-pedigree cats (Gookin et al 1999, 2004).
While it is possible that some cat breeds may have genetic factors that predispose them to this infection, it is probably more likely that it is the way in which the cats are managed that predisposes their colony to the introduction of the parasite and exposes the cats to a high level of infection. This is consistent with the knowledge that high population density is the most important risk factor for this infection because it predisposes to high levels of faecal–oral spread (Gookin et al 1999, 2004, Foster et al 2004). There may be many reasons why cats in the UK may have a lower frequency of infection than in the USA, and this could result, at least in part, from the lower indoor pedigree cat density that is seen in the UK compared to the USA (Julia May and Susan Little, personal communications 2006). It may also vary between different cat breeds as management practices vary and some breeds contain significant numbers of cats imported from the USA.
It is notable that over 70% of the faecal samples in the current study came from pedigree cats. This is in marked contrast to the general UK data that suggest that only 8–12% of domestic pet cats are of pedigree breeds (PFMA 2004, Tiffin 2006). This indicates that faecal samples from pedigree cats are much more likely to be submitted for T foetus PCR than those from non-pedigree cats. This sampling bias probably arose because breeders and owners of pedigree cats are more likely to be motivated to undertake the investigation. In addition, they may be financially more able to pay for the assay, and through international cat breeding magazines they may have had an increased awareness of the infection. The prevalence of the infection may have been subject to sampling bias: Only submitting samples after most other easily diagnosed causes of diarrhoea had been ruled out may have biased towards the likelihood of finding trichomonad infection. However, it is also possible that recent antibiotic use could have suppressed the infection, and as this information was not present on the admission forms this could not be assessed.
The current study agrees with the USA data in relation to the age of the T foetus-infected cats. The infected cats in the current study had a median age of 7 months, compared to a median age of 9 months in the USA (Gookin et al 1999, 2004, Foster et al 2004). All of these studies have found that the majority of infected cats are of a year of age or younger (Gookin et al 1999, 2004, Foster et al 2004). Unfortunately, the relatively low number of positive cats in the current study meant that because of low power the study was unable to show that younger cats were statistically more likely to be infected. In addition, as the samples were submitted specifically for T foetus assessment it is possible that they were biased towards young cats as information from the USA has suggested that T foetus is particularly common in this age group.
The current study indicates that T foetus infection may be found frequently in diarrhoea from cats in the UK, suggesting that it may be an important, common, and previously unrecognised cause of diarrhoea in cats in this country. This protozoan is, therefore, an important differential diagnosis to consider when investigating cats with persistent large bowel diarrhoea, particularly when the cats are young, and/or come from a pedigree household or another multi-cat establishment.
Information
More information about T foetus infection in cats is available on the FAB website www.fabcats.org, the PCR and ‘In Pouch TF’ tests are available in the UK from Capital Diagnostics in Edinburgh (0131 535 3145) and in the USA from North Caroline State University, USA, www.cvm.ncsu.edu/mbs/gookin_jody.htm. Information on the use of ronidazole for the treatment of this infection in cats in the UK is available from
Footnotes
Acknowledgements
The authors wish to thank Jenny Thacker for her technological assistance. KS and NR wish to thank the Feline Advisory Bureau (FAB) for the support of their Lectureship and Residency, respectively, and TM wishes to thank Hill's Pet Nutrition for the support of her Residency.
