Abstract
A congenital encephalopathy with spongiform degeneration and prominent neuronal apoptosis was observed in a 4-month-old Persian male cat with a history of depressed mental status and ataxia. On clinical examination, signs included right head tilt, ventroflexion of the head and neck, and tetraparesis. Histological examination of the central nervous system revealed multifocal, bilateral and symmetrical vacuolar degeneration of the neuropil, mainly involving the cerebellar and vestibular nuclei area, the caudal colliculi, the mesencephalic nuclei, the tegmental area and the deeper layer of the cerebral cortex. Accumulation of phosphorylated neurofilaments was detected in neuronal perikarya of the deep cortical layers, hippocampus and thalamus. Numerous pyknotic and apoptotic neurons were also observed in the cerebral cortex. These neuropathological changes differ from those observed in previous reports of spongiform degeneration of the grey matter in cats and were suggestive of a congenital neurodegenerative disease.
Spongiform change in the central nervous system (CNS) can involve the grey or white matter, or both, and is characterised by the presence of vacuoles in the myelin sheaths, in the dendrites or axons in the neuropil or by the swelling of astrocyte or oligodendrocyte cytoplasm (Summers et al 1995).
Congenital spongiform degeneration of the grey matter has been previously described in cats (Morita et al 2002, Vidal et al 2004) and a suspected inherited encephalomyelopathy characterised by spongy change in the grey matter and wallerian degeneration in the spinal cord has been described in five related Birman kittens (Jones et al 1992). Transmissible spongiform encephalopathies cause spongiosis in humans and animals and feline spongiform encephalopathy (FSE) has been recognised since 1990. Common clinical signs were ataxia, abnormal behaviour, hyperaesthesia, and altered grooming (Wyatt et al 1991). The presence of fibrils similar to those described in scrapie or bovine spongiform encephalopathy has also been described in FSE (Pearson et al 1992).
Spongiform degeneration of the white matter has been reported in two Egyptian Mau kittens with widespread vacuolation throughout the brain and spinal cord. The most severely affected areas were the cerebral subcortical, subependymal and cerebellar white matter, midbrain and brain stem (Kelly and Gaskell 1976). Similar lesions have also been described in cats with experimental bromethalin toxicosis (Dorman et al 1992). Grey and white matter spongiform degeneration has also been described in several dog breeds. Indeed, spongiform degeneration of the grey matter has been described in Bull Mastiff, Saluki, Cocker Spaniel and Malinois shepherd-cross puppies (Carmichael et al 1983, Luttgen and Storts 1987, Cachin and Vandevelde 1991, Summers et al 1995, Morales et al 2000, Johnson et al 2001) and white matter spongiform degeneration has been described in Labrador Retriever, Shetland Sheepdog, Samoyed, and Silky Terrier (Wood and Patterson 2001). In Shetland Sheepdogs, spongiform encephalopathy has been recently associated with a missense mutation in cytochrome b of the complex IV of the respiratory chain (Li et al 2006).
This report describes the clinical and pathological findings of an unusual form of spongiform encephalopathy in a kitten, mainly involving brain stem nuclei and associated with prominent neuronal apoptosis.
A 4-month-old Persian male cat was referred with a history of depressed mental status and progressive ataxia. The cat was the only abnormal kitten of a litter of three, but one kitten of the previous litter from the same queen, died with similar neurological signs. The cat lived indoors and was fed a dry commercial kitten diet. Vaccination and treatments against intestinal worms were up-to-date and no history of toxin exposure was reported.
Physical and neurological examination revealed depressed mental status, right head tilt, ventroflexion of the head and neck, and tetraparesis with absence of proprioception in both the fore and hind limbs. Examination of the spinal reflexes and cranial nerves revealed no abnormalities. The neurological examination was consistent with a brain stem anatomic diagnosis. Routine haemogram, urinalysis and serum biochemistry profiles revealed no abnormalities, except for a mild reduction of total iron concentration (69 μg/dl; reference range: 110–170) and a mild increase of α2 globulins (1.224 g/dl; reference range: 0.41–0.68). Cerebrospinal fluid analysis revealed a clear and colourless fluid with negative Pandy test. Differential diagnoses included congenital brain abnormalities, neurometabolic and neurodegenerative disorders. The owner refused further ancillary diagnostic tests and requested euthanasia because of poor general condition of the cat and poor prognosis.
A complete necropsy was performed and post-mortem examination revealed no gross abnormalities. Major organ and CNS samples were fixed in phosphate-buffered 4% formalin solution and then routinely processed for histology. Tissue sections were stained with haematoxylin and eosin, and nervous tissue sections were also stained with Luxol fast blue and Bielschowsky silver stain. Immunoperoxidase methods for glial fibrillary acidic protein (GFAP, Dako), phosphorylated neurofilament (clone 2F11, Dako) and PrP-res (L42, R-Biopharm) were performed on selected sections. Detection of apoptotic cells was performed in situ using a modified terminal transferase (TdT)-mediated dUTP nick end labelling (TUNEL) method on sections of the forebrain and cerebellum. The reaction was visualised using an anti-digoxigenin Fab fragment conjugated with alkaline phosphatase (Boehringer, GE) and the nitro blue tetrazolium (NBT)/5-bromo-3-indolyphosphate p-toluidine salt (BCIP) substrate.
Histological lesions were confined to the CNS and were characterised by multifocal, bilateral and symmetrical vacuolar degeneration of the neuropil. Vacuoles, which were apparently empty, varied in size approximately from 40 to 100 μm and, in the most severely affected areas, they frequently merged into large confluent vacuoles up to 300 μm in diameter. The vacuoles were mainly distributed within the grey matter of cerebellar and vestibular nuclei area, caudal colliculi, mesencephalic nuclei (red, oculomotor, and trigeminal), tegmental area and the deeper layer of the cerebral cortex, especially in the frontal cortex and lesser in the parietal and occipital cortex (Fig 1). Randomly, tiny neuropil vacuolation was observed in the thalamic nuclear area and in the dorsal horns of the spinal cord. In the cerebellum, Purkinje cells were normal, whereas in the granule cell layer a few, unevenly scattered vacuoles were detected. Only rare vacuoles were observed within the molecular layer. Numerous dark and pyknotic neurons were observed in the superficial layers of the cerebral cortex, the gyrus dentatus of the hippocampus and the thalamic nuclear area. However, no vacuoles were detected in these neurons. White matter was normal throughout the CNS.

Cerebellar interpositus nucleus: diffuse neuropil vacuolation with single and confluent vacuoles. Neuronal cytoplasm and surrounding cerebellar white matter show no morphological abnormalities. Haematoxylin and eosin stain; ×320 magnification.
Immunohistochemistry for PrP-res yielded negative results and GFAP revealed no significant reactive astrogliosis in the areas in which the vacuolations were observed. Diffusely, neurons of the deep cortical layers, hippocampus and thalamus showed an intense perikarya accumulation of phosphorylated neurofilaments (Fig 2). Also scattered axonal spheroids were strongly immunolabelled with phosphorylated neurofilaments and numerous torpedoes and spheroids were evident within the molecular and granular layers of the cerebellar cortex.
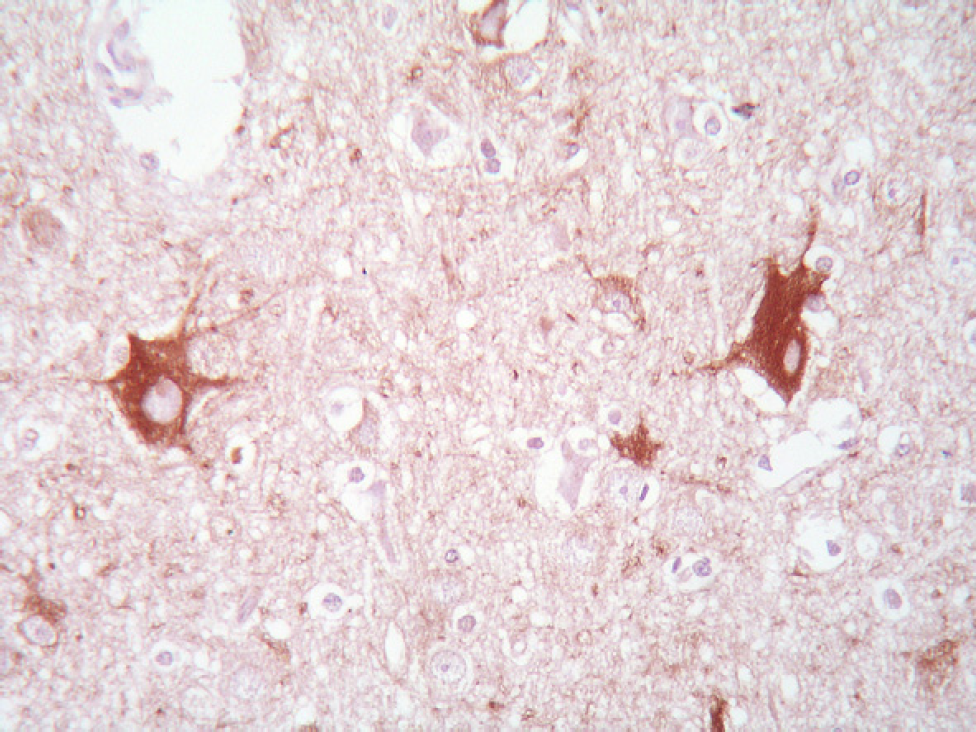
Deep cerebrocortical layer: two neurons show a striking perikaryal accumulation of phosphorylated neurofilaments. Axons show normal shape and normal content of neurofilaments. 2F11 antibody, avidin–biotin–peroxidase complex method, haematoxylin counterstain; ×500 magnification.
In situ analysis of DNA fragmentation with the TUNEL method revealed TUNEL positive labelling in cerebral cortex where the nuclei of large-sized pyramidal neurons in layers III and V were intensely stained (Fig 3).

Deep frontal cortical layer: numerous neurons show shrunken cytoplasm and TUNEL positive nuclei. TUNEL method, nitro blue tetrazolium; ×450 magnification.
Pathological findings in our cat were consistent with a spongiform encephalopathy mainly involving the cerebellar and vestibular nuclei area, some brain stem nuclei and the deeper layer of the cerebral cortex. Vacuolation involved the neuropil and was associated with a striking neuronal degeneration and apoptosis. Indeed, neurofilaments undergo a progressive phosphorylation along the axon. Neurofilament hyperphosphorylation in the perikarya and in the axons suggests an impairment of axonal transport. These features are common pathological changes of several neurodegenerative conditions (Nixon and Sihag 1991). Neuronal apoptosis has been described in human neurodegenerative diseases and the abundance of TUNEL positive nuclei in cortical areas suggests that apoptosis may be involved in the mechanism of neuronal death in this cat.
Spongiform changes in the CNS of animals are frequently associated with prion diseases, metabolic deficits or derangements as in lysosomal storage disorders, hepatic encephalopathy, thiamine deficiency, and disorders of aminoacids' metabolism (Summers et al 1995). The features and distribution of lesions in our cat can be differentiated from FSE, which mainly consists of spongiform changes involving the medial geniculate nucleus, thalamus and corpus striatum and neuronal vacuolation mainly involving the raphe nucleus, dorsal nucleus of the vagus nerve, and red nucleus. Moreover, FSE has a later onset, affecting adult or aged animals (Wyatt et al 1991) and PrP-res immunolabelling in our case was negative.
Bilateral and symmetrical foci of neuropil spongiform change have been described in five related Birman kittens affected by hind limb paralysis with exaggerated spinal reflexes, and deficits of postural reactions (Jones et al 1992). Lesions mainly involved the cerebral cortex of the piriform lobe, the inferior collicular nuclei of the midbrain, thalamus, cerebellar peduncles, oculomotor nucleus, and medulla oblongata. Cerebral spongiform degeneration was associated with wallerian degeneration in sensory and motor white matter tracts of the spinal cord. A case of neuropil vacuolation of the substantia nigra and some brain stem nuclei has been described in a 9-month-old female cat showing generalised ataxia and normal spinal reflexes (Morita et al 2002). Clinical signs of our cat resembled a case reported by Vidal et al (2004) in which lethargy, head tilt, tetraparesis and conscious proprioceptive deficits were described, but lesions were different. Indeed, in that case a severe neuropil vacuolar degeneration associated with dendritic processes of neurons and astrocyte prolongations involved all brain stem nuclei, cerebellar nuclei, cerebral and cerebellar cortices.
Thiamine deficiency in cats causes axonal and neuronal degeneration with vacuolar changes but is typically associated with bilateral and symmetrical petechial haemorrhages and oedema involving caudal colliculi, lateral geniculate and vestibular nuclei (Summers et al 1995).
In dogs, grey matter spongiosis with neuropil vacuolation has been reported in Bull Mastiff (Carmichael et al 1983, Johnson et al 2001), Saluki (Luttgen and Storts 1987, Summers et al 1995), Malinois shepherd-cross puppies (Cachin and Vandevelde 1991) and Cocker Spaniel dogs (Morales et al 2000). As in our cat, lesions were prominent in the cerebellar nuclei, but bilateral and symmetrical spongiform degeneration was associated with apparently normal neurons (Summers et al 1995, Morales et al 2000, Johnson et al 2001). Spherical or ovoid vacuoles were accompanied by gliosis (Cachin and Vandevelde 1991, Summers et al 1995), axonal spheroids (Carmichael et al 1983, Morales et al 2000, Johnson et al 2001), and demyelination (Morales et al 2000). The aetiology of those diseases remained undetermined and a genetic basis has been suggested (Morales et al 2000, Johnson et al 2001). Spongiform degeneration with neuronal vacuolation and spinocerebellar degeneration has been also described in several Rottweiler puppies. Affected dogs showed an early onset of laryngeal paralysis and gait abnormalities with single or multiple neuronal vacuolation mainly in the cerebellar and vestibular nuclei and also in some brain stem nuclei (Davies and Irwin 2003). No involvement of the apoptotic pathway in vacuolated neurons was found (Pumarola et al 1999) and a possible genetic basis was suspected (van den Ingh et al 1998).
The age of the animal, the early onset of neurological signs and the characteristics of the lesions in our cat are suggestive of a congenital form of encephalopathy.
Footnotes
Acknowledgements
We wish to thank the Italian Reference Centre for the Spongiform Transmissible Encephalopathies (CEA, Turin) for the PrP immunohistochemistry.
