Abstract
The aim of this paper is to define the endoscopic bronchial anatomy of the cat and to adapt these findings to the bronchoscopic nomenclature previously described for the dog by Amis and McKiernan (Amis TC, McKiernan BC (1986) Systematic identification of endobronchial anatomy during bronchoscopy in the dog. American Journal of Veterinary Research
Bronchoscopy using either rigid or flexible endoscope is an important diagnostic procedure for the evaluation of many respiratory diseases and occasionally is used for the removal of respiratory foreign bodies (Lotti and Niebauer 1992, Cucinotta et al 1998). This technique allows not only a direct visualization of the lumen of the trachea, principal bronchi and many segmental bronchi but also permits collection of diagnostic samples through bronchoalveolar lavage (BAL), biopsy or brushings (Venker-van Haagen 1985, Roudebush 1990, Padrid and McKiernan 1999, Rha and Mahony 1999, Jonhson 2001, Kuen and Hess 2004, McKiernan 2004).
When performing bronchoscopy, it is essential to be familiar with the normal structure of the bronchial tree because it is very easy to lose orientation during the examination. Understanding of the normal endobronchial anatomy is a necessary preliminary step to recognize abnormal anatomy and pathological changes that may be encountered by the bronchoscopist. An endoscopic atlas of the bronchial tree is useful because it can be consulted during the bronchoscopic examination and as an aid in evaluating chest radiographs in many instances. It is advantageous for clinicians to develop a consistent approach in the bronchoscopic examination, utilizing a routine method for the inspection of the airways by using a standardized anatomical nomenclature to ensure that the airways are completely inspected.
The endoscopic anatomy of the airways of the dog was first described in detail by Venker-van Haagen (1979). It was not, however, until the development of a standardized bronchial nomenclature for the dog by Amis and McKiernan (1986) that clarified the bronchoscopic anatomy. Using a standardized nomenclature, improves communication between endoscopists in defining a bronchial location, to relocate lesion sites and to correlate bronchoscopic findings with results of other diagnostic procedures (Amis and McKiernan 1986). Similar bronchoscopic mapping has been reported in other species but not in the cat (Nakamura et al 1986, Smith et al 1994, Harper et al 2001). Although the bronchial anatomy of the dog and cat is thought to be basically the same, a similar scheme and nomenclature has not been established. This paper describes the normal bronchoscopic anatomy of the cat with a photographic atlas of the feline airways.
Materials and methods
The endobronchial anatomy of the cat lung was determined through the combined use of videoendoscopy of normal anesthetized cats, euthanased cats and dried lung specimens. Silicone cast models were made from the dried lung specimens for further study. All the lungs were examined endoscopically in a systematic manner, moving through the bronchial tree in the same direction, from one lung lobe to the other.
Lung specimens
Ten air-dried feline lungs were prepared from euthanased cats using the methods previously described (Hildebrand 1968a, Markarian 1975, McKiernan and Kneller 1983, Weiss and Tweddal 1996). The bronchial anatomy of each lung was recorded with a rigid endoscope (Storz, Hopkins 7218BSA, 2.7 mm diameter, 18 cm length, 30° viewing angle; Karl Storz GmbH & Co. KG; Mittelstraße 8, Postfach 230, D-78532 Tuttlingen, Germany).
The bronchial anatomy was also determined in two cats immediately following euthanasia. These cats were placed in sternal recumbency and evaluated using a flexible bronchoscope (Olympus Bronchoscope, BF type P240, 5.2 mm diameter, 55 cm length: Olympus America Inc, Two Corporate Center Drive, Melville, New York 11747-9058). All the cats were euthanased for reasons unrelated to the study and were considered free of bronchial disease. The source of these cats was a local humane shelter.
Corrosion silicon casts from each air-dried lung were made. The technique for cast models of the trachea and bronchi was adapted from methods previously described for either ‘in situ’ or excised lungs (Adrian 1964, Hildebrand 1968b, Smith et al 1990). Household silicon caulking material was injected into the large airways and following hardening of the silicon caulk each lung was then digested using potassium hydroxide leaving a silicone model of the larger airways.
Normal cats
The airways of eight respiratory healthy cats weighing 2–4.8 kg were examined (age 10 months–10 years). All these cats underwent anesthesia for minor surgery (eg, ovariectomy), and all the bronchoscopies were performed with the Faculty's approval project and the owner's signed authorization. Each cat was premedicated with medetomidine (Domitor; Pfizer) (5–10 μg/kg) and butorphanol (Dolorex; Intervet) (0.2 mg/kg) IM or only with butorphanol (0.2 mg/kg) IV. The patients were preoxygenated for 5 min before starting the bronchoscopy, using 100% oxygen (2 l/min) through a face-mask. Patients were then anesthetized with propofol (Rapinovet; Schering-Plough) IV (3–4 mg/kg) followed by bolus injections (1–2 mg/kg) given as needed to maintain an anesthetic plane. A 2% lidocaine solution (Lidocaine 2%; Angelini) (0.2 ml total) was dropped on the glottis of each patient prior to starting the procedure to prevent laryngeal spasm. During the bronchoscopic procedure a flow of 100% oxygen (approximately 1 l/min) was delivered through the endoscope sheath. The flow of oxygen was reduced when moving the endoscope into smaller bronchi to avoid ‘air trapping’ and potential barotrauma. At the end of the procedure the oxygenation was given by face-mask or endotracheal intubation.
The cats were placed in sternal recumbency and the airways were evaluated using a rigid endoscope (Storz, Hopkins 61324BA, 2.9 mm diameter, 36 cm length, 30° viewing angle). Images of the bronchoscopies performed in the normal anesthetized cats were systematically recorded (AIDA-dvd and AIDA-vet Digital Capture System; Karl Storz GmbH & Co. KG; Mittelstraße 8, Postfach 230, D-78532 Tuttlingen, Germany.) for the purpose of comparing the findings of each lung and to create a map and photographic atlas. No complications were encountered in any cat while performing bronchoscopy and all cats made an uneventful recovery.
Results
Feline endobronchial anatomy
The carina was easily identified in all lungs as a sharp wedge that divided the trachea into the two principal bronchi: the right principal bronchus is nearly co-linear with the trachea, while the left principal bronchus originates at a slightly more acute angle from the trachea. From the bifurcation of the trachea the right side of the bronchial tree was first evaluated. The right cranial, right middle, accessory and right caudal lobar bronchi could be observed. The endoscope was then withdrawn into the trachea and the left side was evaluated and the left cranial and left caudal lobar bronchi were observed. In all the lungs examined the lobar bronchi were always visualized. In many cases it was possible to distinguish segmental bronchi exiting lobar bronchi. In some areas subsegmental bronchi could also be observed.
After passing through the glottis, the trachea appeared as a round lumen and the tracheal cartilages were visible as C-shape rings connected dorsally by the tracheal membrane. The mucosa appeared as a very pale pink, smooth surface and submucosal vessels were readily visible along all the length of the trachea (Fig 1). The carina was visible as a sharp bifurcation with a V shape that divided the trachea into right and left principal bronchi (Figs 2 and 10). The lobar, segmental and subsegmental bronchi, that followed at regular intervals, appeared pale pink, round and smooth. The submucosal vessels were still visible, but with slightly less detail. Bronchial bifurcations at this level were more U-shaped compared with the carina. The reader should refer to the photographic atlas of the normal feline bronchoscopic anatomy (see Figs 1–17).

Trachea. The lumen is round and it is possible to distinguish the cartilage rings. The dorsal tracheal membrane is seen in a dorsal position. Under the pale pink mucosa a network of submucosal capillaries is visible.
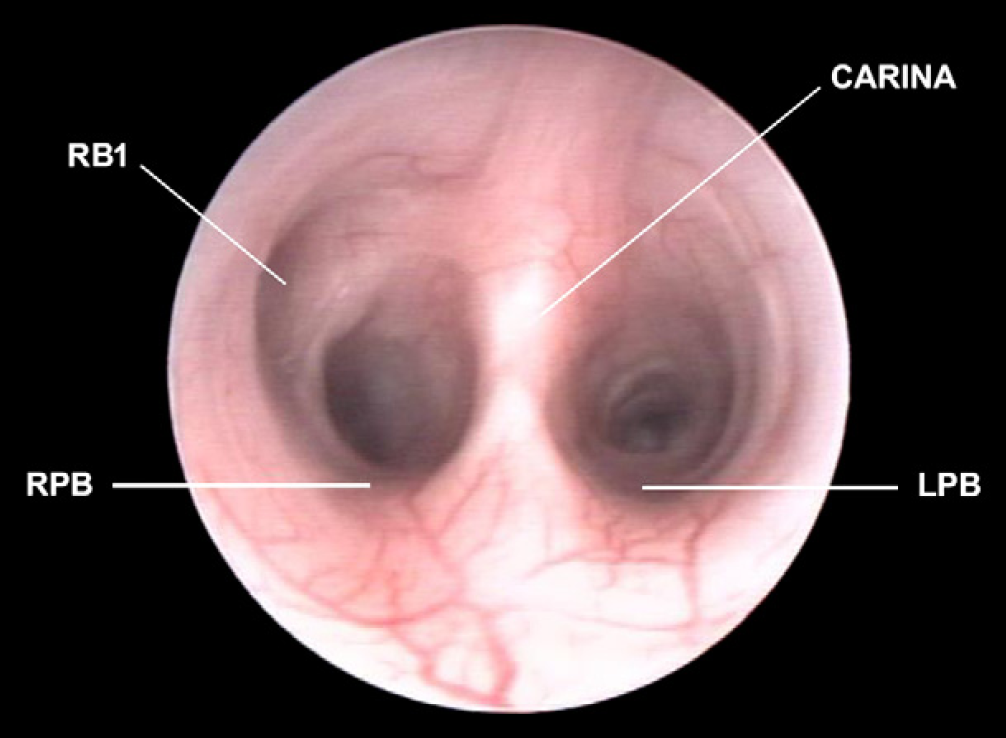
Carina and right and left principal bronchi (RPB, LPB). It is possible to see the origin of the right cranial lobar bronchus (RB1).

Right principal bronchus (RPB), right cranial lobar bronchus (RB1) and origin of the first dorsal segmental bronchus (RB1D1).
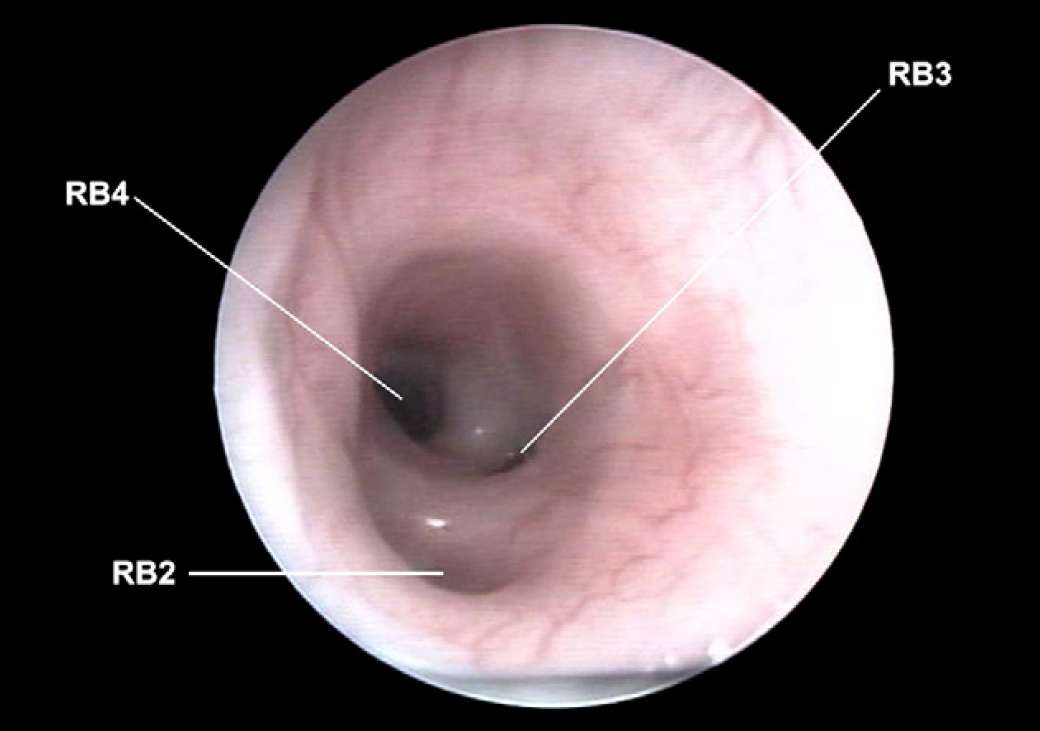
Right middle lobar bronchus (RB2), right accessory lobar bronchus (RB3) and right caudal lobar bronchus (RB4).

Right accessory lobar bronchus (RB3) and right caudal lobar bronchus (RB4) with the origin of the first dorsal segmental bronchus (RB4D1).

Right caudal lobar bronchus (RB4) with origin of the first dorsal segmental bronchus (RB4D1) and ventral (RB4V1) and second dorsal segmental bronchus (RB4D2). It is possible to make out the right accessory lobar bronchus (RB3).
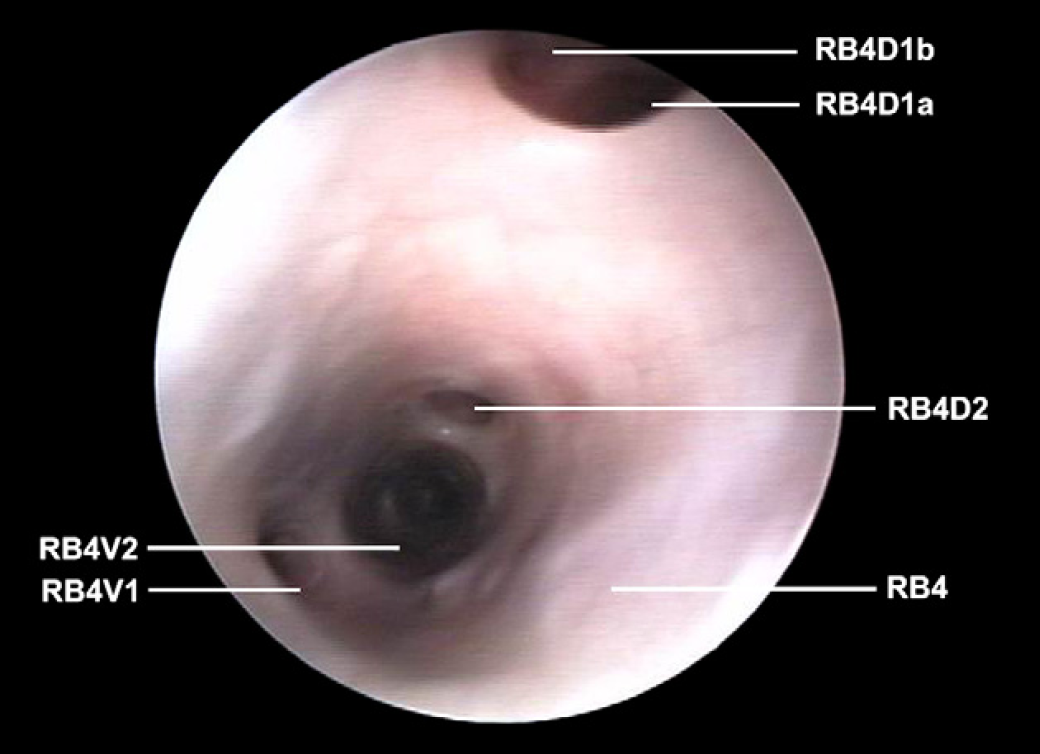
Right caudal lobar bronchus (RB4) with origin of the first dorsal segmental bronchus in which it is possible to make out the division into two subsegmental bronchi (RB4D1a and RB4D1b); first ventral segmental bronchus (RB4V1), second dorsal segmental bronchus (RB4D2) and ventral (RB4V2).

Right caudal lobar bronchus (RB4) with origin of the first ventral segmental bronchus (RB4V1) and second dorsal segmental bronchus (RB4D2).

Right caudal lobar bronchus (RB4) with closer vision of the second dorsal segmental bronchus (RB4D2).

Carina and right and left principal bronchi (RPB, LPB). It is possible to make out the origin of the left cranial lobar bronchus (LB1).
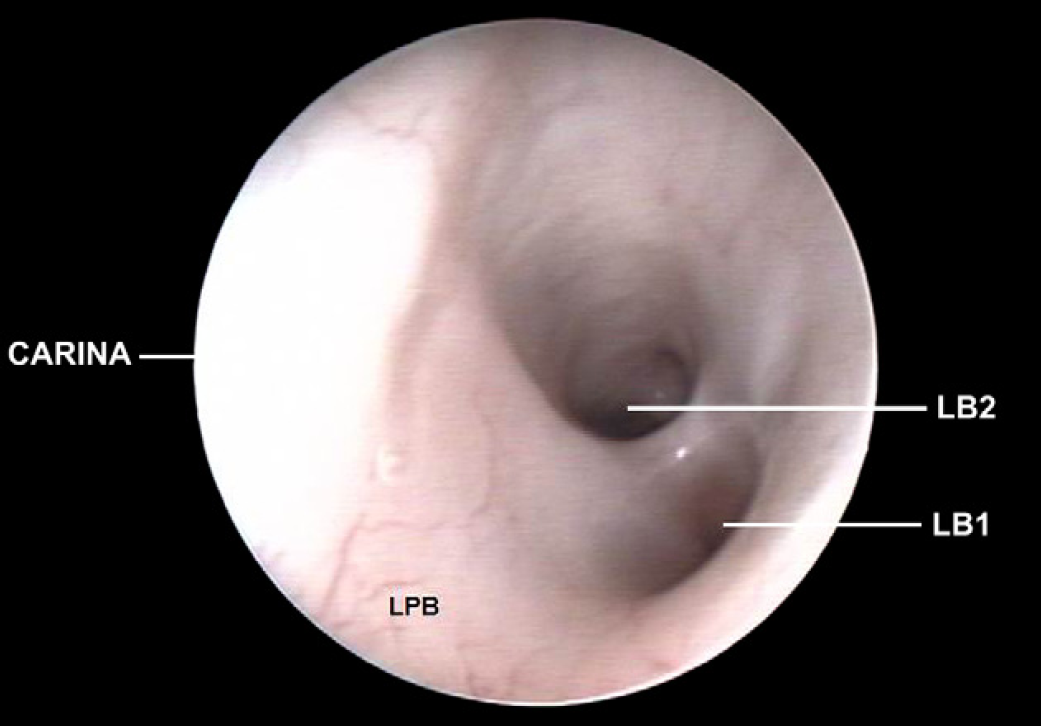
Left principal bronchus (LPB), left cranial lobar bronchus (LB1) and left caudal lobar bronchus (LB2).

Left cranial lobar bronchus (LB1) with origin of the first ventral segmental bronchus (LB1V1). Left caudal lobar bronchus (LB2) with origin of the first dorsal segmental bronchus (LB2D1).

Left principal bronchus (LPB) with closer vision of the left cranial lobar bronchus (LB1) with origin of the first ventral segmental bronchus (LB1V1). Left caudal lobar bronchus (LB2).
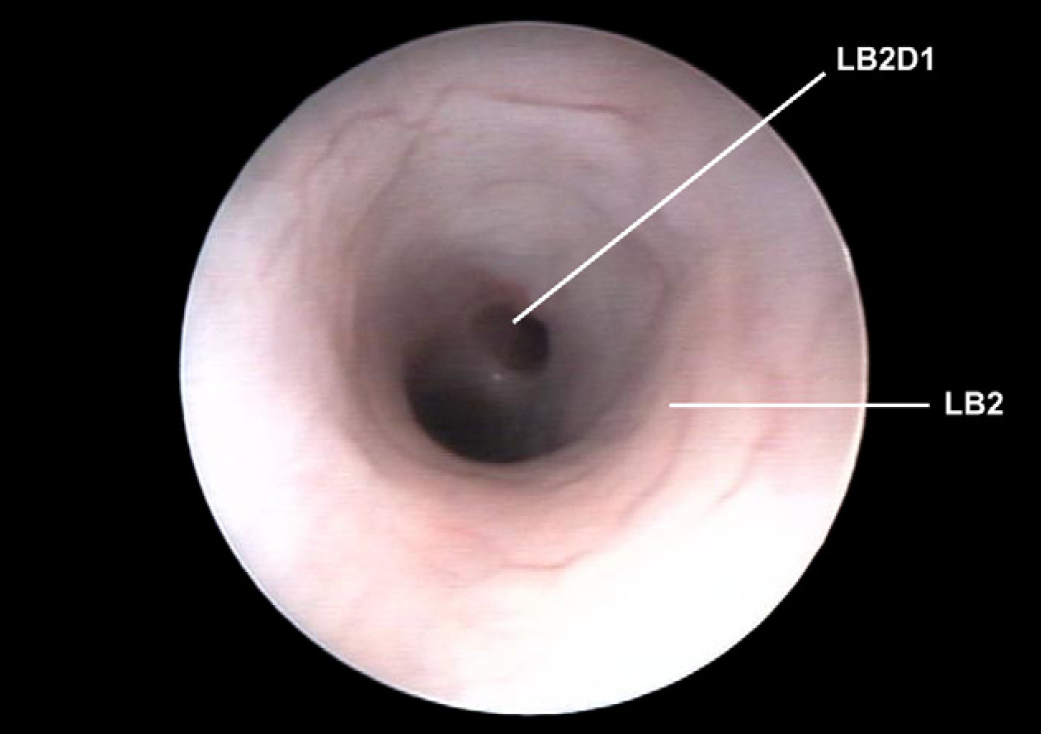
Left caudal lobar bronchus (LB2) with origin of the first dorsal segmental bronchus (LB2D1).

Left caudal lobar bronchus (LB2) with origin of the first ventral segmental bronchus (LB2V1) and second dorsal segmental bronchus (LB2D2).

Left caudal lobar bronchus (LB2) with origin of the second dorsal segmental bronchus (LB2D2) and ventral (LB2V2).
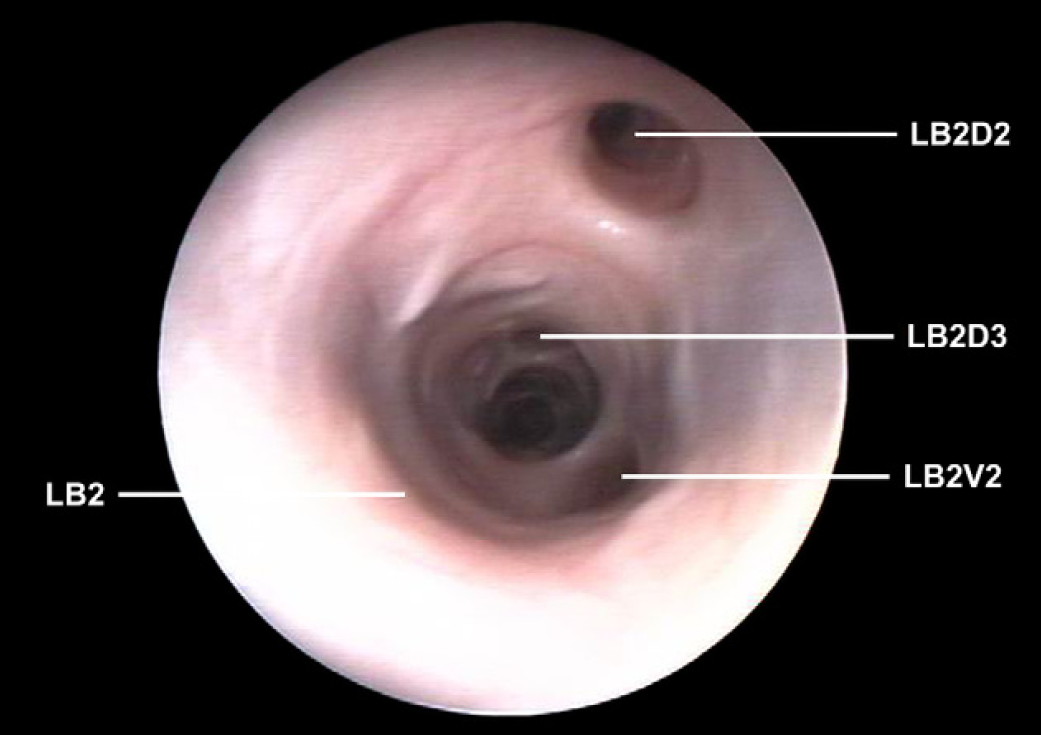
Left caudal lobar bronchus (LB2) with origin of the second dorsal segmental bronchus (LB2D2) and ventral (LB2V2). Origin of the third dorsal segmental bronchus (LB2D3).
Right lung
Entering the right principal bronchus the first lobar bronchus encountered was the right cranial that arose lateral to the right principal bronchus, directly opposite the carina. It was not possible to enter this bronchus with the rigid endoscope, but it was possible to visualize the dorsocaudal entrance of the first segmental bronchus in 15 of 20 lungs examined (Fig 3). In one air-dried lung it was also possible to visualize the origin of the second segmental bronchus that was in a dorsal position.
Moving caudally in the right principal bronchus the second bronchus encountered was the right middle lobar bronchus that was located in a ventral position (Fig 4). In two air-dried lungs it was possible to introduce the tip of the rigid endoscope in the right middle lobar bronchus and to view the first segmental bronchus which was oriented in a medial and dorsal direction.
Moving caudally the next or third lobar bronchus originated in a ventromedial position and was identified as the right accessory lobar bronchus (Fig 5). In two air-dried lungs it was possible to enter this bronchus and visualize the division into one ventral and one dorsal segmental bronchus.
The right principal bronchus then continued caudally to the origin of the accessory lobar bronchus as the right caudal lobar bronchus. At approximately the same level as the accessory bronchus, the origin of the first segmental bronchus of the right caudal lobar bronchus was noted located in a dorsal position (Fig 6). This segmental bronchus was visualized in all except in one case. In one anesthetized cat it was possible to visualize the bifurcation into two subsegmental bronchi having a lateral and medial direction (Fig 7). Advancing caudally the entrance of the second segmental bronchus was noted; it was located in a ventral position and was visualized in 14 cases (Fig 8). It was also possible to distinguish the division of this segmental bronchus into two subsegmental bronchi in two occasions (one air-dried lung and in one anesthetized cat). A third segmental bronchus originated from the dorsal wall of the right caudal lobar bronchus and was visualized in 10 cases (Fig 9). In five cases (two air-dried lungs and three anesthetized cats) it was possible to reach the entrance of the fourth segmental bronchus located in a ventral position.
Left lung
Entering the left principal bronchus the first bronchus encountered was the left cranial lung lobar bronchus arising laterally and slightly ventral in the wall of the left principal bronchus (Fig 11). It was not possible to enter this bronchus but it was possible to visualize its division into a ventrocaudal segment and the dorsocranial continuation of the first left lobar bronchus (Fig 12). The ventrocaudal segment arose immediately after the origin of the lobar bronchus and was always visualized (Fig 13).
The left caudal lobar bronchus was the continuation of the left principal bronchus caudal to the left cranial lobar bronchus origin. The first segmental bronchus branched in a dorsal location from the lobar bronchus and was identified in 12 of 20 cases (Fig 14). Caudal to the first segmental bronchus a second segmental bronchus originated from a ventral position and was observed in nine of 20 cases (Fig 15). In two air-dried lungs it was possible to enter this bronchus and to observe two subsegmental bronchi. The third segmental bronchus originating from the dorsal wall of the left caudal lobar bronchus, and was visualized in seven lungs and its further division into two subsegmental bronchi was seen in one air-dried lung. A fourth segmental lobar bronchus having a ventral origin was identified in four air-dried lungs and two anesthetized cats (Fig 16). In three air-dried lungs and one anesthetized cat it was possible to observe the opening to a third dorsal segmental lobar bronchus (Fig 17).
Cast models
Silicone cast models are replicas of the bronchial tree and were a useful guide to the anatomy evaluated by bronchoscopy. All lung casts demonstrated similar lobar bronchi that arose from the principal bronchi. The models confirmed the origin and direction of the lobar, segmental and subsegmental bronchi and in almost all cases corresponded with the findings of the bronchoscopic anatomy.
On the right side the first lobar bronchus (to the right cranial lobe) exits the right principal bronchus laterally and dorsally then turns cranially, running parallel to the trachea. The second lobar bronchus (to the right middle lobe) exits the right principal bronchus ventrolaterally and continued with a ventrocaudal and lateral direction. The third lobar bronchus (to the right accessory lobe) exits ventromedially and continues caudomedially. The right principal bronchus continues as the fourth lobar bronchus (to the right caudal lobe) with a caudolateral direction.
On the left side, the first lobar bronchus (to the left cranial lobe) exits laterally and ventrally from the left principal bronchus, and immediately divides into a larger dorsocranial segment that turns cranially and runs parallel to the trachea, as well as a smaller ventrocaudal segment with a caudoventral and lateral direction. The left principal bronchus becomes the second lobar branch (to the left caudal lobe) just after the origin of the cranial lobar bronchus and runs caudolaterally.
The segmental lobar bronchi in the cast models arise in a more-or-less regular alternating sequence. However, in two casts the lobar segmental bronchi of the accessory lobe did not divide but gave off many subsegmental bronchi running in different directions.
In another cast an additional bronchus was observed arising from the ventral wall of the right caudal lobar bronchus behind the first dorsal segmental bronchus. This bronchus ran parallel to the right caudal bronchus in a ventrocaudal direction. This ‘additional’ bronchus divided in two branches of approximately the same diameter directed in a lateral and medial aspect giving off subsegmental bronchi. During bronchoscopy this ‘additional’ bronchus was not visualized.
Bronchial nomenclature
The 20 lungs endoscopically evaluated generally had a consistent anatomical structure showing similarity to the endobronchial anatomy and nomenclature described in the dog by Amis and McKiernan (1986).
The lobar bronchi were named on the basis of the principal bronchus (right or left) from which they originate and were labeled either R or L, respectively. Next a numeric sequence was then given for each exiting lobar bronchi in the order of their origin from the principal bronchus moving caudally. The right cranial bronchus was then designated as RB1 (right bronchus 1), the right middle bronchus was RB2 (right bronchus 2), the right accessory was RB3 (right bronchus 3) and the right caudal bronchus was RB4 (right bronchus 4). On the left side, the left cranial bronchus was LB1 (left bronchus 1) and the left caudal bronchus was LB2 (left bronchus 2).
The segmental bronchi were designated on the basis of their origin from the lobar bronchus, and were almost always in a dorsal or ventral aspect and identified with the capital letter D or V (dorsal, ventral). The identified segmental bronchi were then named in a numeric sequence in the order of origin from the lobar bronchus advancing caudally with the scope. Thus, for example, the first two segmental bronchi originating dorsally from the right caudal lobe were identified as RB4D1 and RB4D2, while the two segmental bronchi with a ventral origin were recognized as RB4V1 and RB4V2. On the left side, the first two dorsal segmental bronchi of the left caudal lobe were LB2D1 and LB2D2, and the same for the two first ventral segments that were LB2V1 and LB2V2. The bronchus to the ventrocaudal part of the left cranial lung lobe was recognized, from the endoscopic point of view, as a segmental bronchus (LB1V1), rather than a new lobar bronchus.
An exception was the right middle bronchus that had segmental divisions that originate in a cranial and caudal direction from the lobar bronchus and to avoid confusion they were defined as caudal (C) and rostral (R). Examination of two air-dried lungs identified the caudal origin of the first segmental bronchus of the middle lobar bronchus, and it was designed as RB2C1.
Although this is an ideal model for segmental lobar nomenclature in the cat, in most cases it was not possible to advance the endoscope into the segmental lobar bronchi.
The subsegmental bronchi were visualized in some situations in this study and when these were observed, the previously reported dog's nomenclature applied. Subsegmental lobar identification was proposed on the basis of the order in which they originate from the segmental bronchus using small letter in sequence (a, b, …), with no reference to their position and orientation. Thus, for example, the two first subsegmental bronchi that originate from LB2V1 are identified as LB2V1a and LB2V1b.
The results from each bronchoscopy recorded using the endoscopic nomenclature, indicate the principal, lobar, segmental and subsegmental bronchi visualized in each case. From these data it was possible to draw a map of the normal feline bronchial tree observed in this study (Fig 18).

Diagrammatic dorsal view of the feline bronchial tree, with the application of the proposed nomenclature system, based on Amis and McKiernan bronchoscopic nomenclature.
Discussion
Bronchoscopy is an important diagnostic and frequently therapeutic tool in small animal veterinary medicine. When performing bronchoscopy it is imperative that the operator has a complete understanding of the normal endoscopic anatomy for proper orientation, to locate lesions and to systematically describe the bronchoscopic findings. The bronchoscopic anatomy has been published for the dog but not for the cat. While many endoscopists assume that the endoscopic anatomy in the dog to be similar to the cat a detailed endobronchial map and adapted nomenclature is not yet available. The purpose of this study was to describe the normal endoscopic anatomy derived from examination of 20 cat lungs and 10 silicone corrosion cast models to develop an endoscopic anatomic nomenclature system and to draw a bronchial map (Fig 18).
Nomenclature systems have been developed for the human bronchial tree but are not suitable for dogs and cats because of their monopodial branching pattern of the segmental and subsegmental lobar bronchi. This is quite different from that of the symmetrical dichotomous pattern observed in humans (Schlesinger and McFadden 1981, Mortesen et al 1983). The monopodial branching system aids endoscopic examination because the parent bronchus is the larger of the two branches seen through the endoscope. In contrast in the dichotomous branching pattern of humans the parent bronchus divides into two daughter segments of similar size making it difficult to maintain orientation during the bronchoscopic examination. For human bronchoscopy extensive mapping systems have been developed identifying airway diameters, lengths and branches (Phalen et al 1978).
In the dog and cat, classical anatomic descriptions (Crouch and Lackey 1969, Gilbert 1975, Hare 1975, Evans and Christensen 1979, Light 1993), comparative anatomical studies (Nakakuki 1980, 1994) and morphometric analyses of airways (Adrian 1964, Horsfield and Cumming 1976, Phalen et al 1978, Thurlbeck and Horsfield 1980, Horsfield et al 1982, Phalen and Oldham 1983, Aslan and Tacki 1994) have been published. This information is not readily useful to the bronchoscopist and is often confusing and difficult to follow. The first endobronchial nomenclature system for the dog was adopted by Ishaq (1980) who described the lobar bronchi of the dog extending monopodially throughout the principal lobe and identified the main series of segmental bronchi with letters and sequential numbers in order of their origin and anatomical orientation. The terms given for the lung lobes (right and left cranial, accessory, right and left caudal) were accepted from this nomenclature system (Nomina Anatomica Veterinaria 1983). Subsequently the endoscopic nomenclature system was modified by Amis and McKiernan (1986) for the dog. In this paper we have adapted their nomenclature system for the cat.
Extensive bronchoscopic examination of the feline bronchial tree is more difficult than of dogs due to the small lobar and segmental bronchial size and the available endoscopes used in this study. A rigid 30° viewing angle, 2.9 mm diameter endoscope used permitted a more extensive evaluation of the feline airways than the flexible 5.2 mm diameter human bronchoscope because of the smaller endoscope diameter. Rigid endoscopes however do provide superior photographic documentation compared to flexible endoscopes. A limitation in the rigid endoscopes in this study was the inability to enter the segmental and subsegmental bronchi as they exit from their parent bronchi. The flexible bronchoscope (5.2 mm diameter) used in this study also did not permit entry further than the lobar bronchi and occasionally a few segmental bronchi. Possibly a smaller diameter flexible endoscope with acute bending angle of the distal tip would have allowed a more detailed exploration of the lobar bronchi and their bifurcations into segmental and subsegmental bronchi but a smaller bronchoscope was not available for this study. Also, if an endoscope is too large in diameter it may obstruct the airway impeding airflow or result in laryngeal, tracheal or bronchial trauma. Thus, in cats the extent of respiratory tree examination is limited more by airway diameter than by the length of the endoscope. Although it was difficult to visualize the smaller airways in the cat routine diagnostic procedures such as biopsy, airway brushing and BAL (Andreasen 2003, Hawkins, 2004) can still be performed for microbiological and cytological examination.
Dried cat lungs were also endoscopically studied and subsequently used for silicone cast models. The dried specimens were useful to compare the endoscopic anatomy of the major airways by examination of the respective silicone corrosion cast of the airways. Endoscopic evaluation of dried lungs was only performed using rigid endoscopy because the airway diameter became smaller following drying and the airway surface became rigid, irregular and difficult to pass by using the flexible endoscope. The cast models allowed us to determine the location of the segmental and subsegmental bronchi and to compare our findings with those reported in the dog. The bronchial tree casts used in this study were not used to study airway length, airway diameter, branching angles and gravitational inclination or anatomical variations as reported in other studies in cats (Adrian 1964, Nakakuki 1980, Aslan and Tacki 1994) but rather to confirm our bronchoscopic interpretation.
During our examination it was never possible to enter the segmental bronchi with the bronchoscope used but only to determine the location of their origin. In some cases we were able to observe the origin of subsegmental bronchi. Both the segmental and subsegmental bronchi were drawn in the bronchial map (Fig 18) when they were visualized. In most cases the lobar bronchi were often too small to enter with the endoscopes we used with the exception of the right and left caudal lobar bronchi. It was in these bronchi that the largest number of segmental bronchi was identified. We also found some differences in the location of segmental bronchi in the cat compared to the dog. For example in one cast model an ‘additional right caudal bronchial lobe’ was identified but during endoscopy was not recognized and misinterpreted to be the first ventral segmental bronchus of the right caudal bronchus (RB4V1) rather than an additional right caudal bronchial lobe.
Knowledge of the normal endoscopic anatomy of the feline bronchial tree is important to know the exact location of the endoscope during the procedure. A standard examination procedure should be followed to evaluate all accessible airways. We recommend evaluating the entire right side (RPB, RB1, RB2, RB3, RB4) first and then the entire left side (LPB, LB1, LB2). Each lobar bronchus should be entered when possible. When the operator is uncertain as to their location in the airway the endoscope should be withdrawn to the carina to regain orientation. Understanding of the normal endobronchial anatomy is a necessary preliminary step to recognize abnormal anatomy and pathological changes that may be encountered by the bronchoscopist. The endoscopic map developed from this study should improve bronchoscopic examination in the cat.
Footnotes
Acknowledgements
A special thanks to Dr Robert Lee for his precious help in the anatomical pr1eparations and to Maura Green, Kriss Obssuth and Jo Ann Harper for their technical support. Thanks to Karl Storz Veterinary Endoscopy America for all the equipment necessary to complete this work.
