Abstract
This report describes an uncommon clinical case of cystic parathyroid adenocarcinoma. A 17-year-old male Persian cat was presented for evaluation of a ventral cervical mass. The cat was inappetent and showed weight loss, polydipsia and vomiting. Serum biochemistry and urinalysis revealed moderate hypercalcaemia, a mild increase of creatinine, isosthenuria and proteinuria. Sodium dodecyl sulphate-agarose gel electrophoresis showed a mixed tubular proteinuric pattern, in accordance with histological examination that revealed interstitial nephritis and glomerulonephritis. Diagnosis of parathyroid carcinoma was based on histopathological findings.
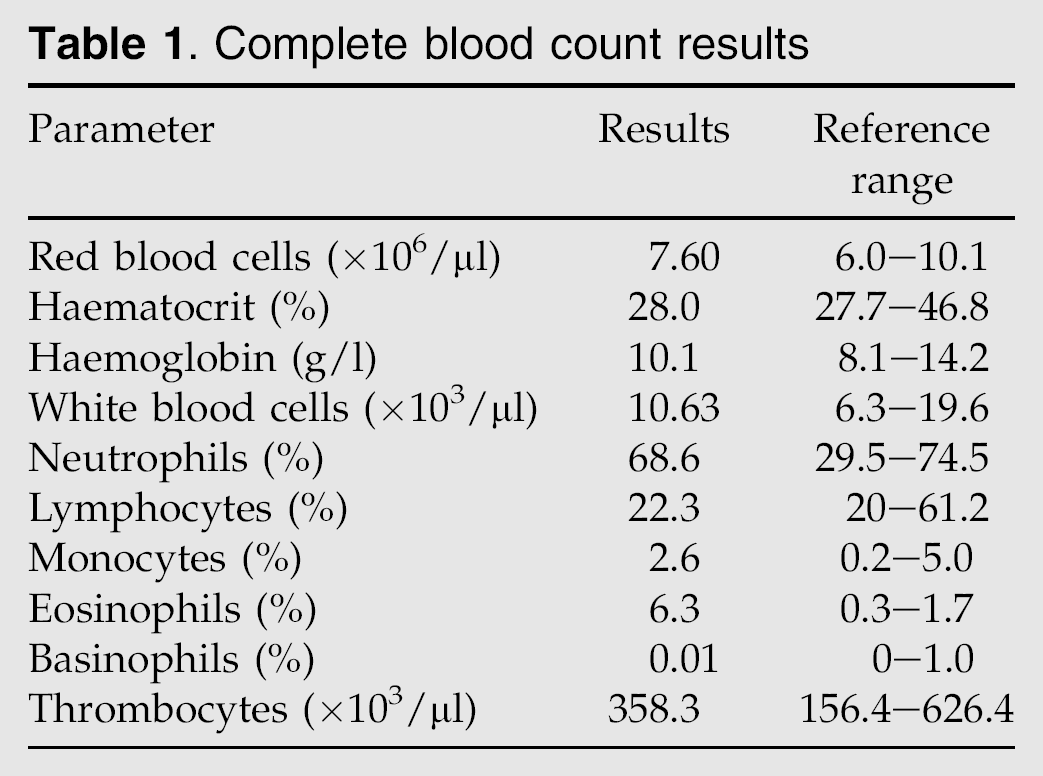
A 17-year-old, neutered male Persian cat, living in an apartment and fed with commercial cat food, was referred for a swelling in the ventral cervical region lasting approximately 4 months. The owner reported that the cat was inappetent and showed weight loss, weakness, polydipsia and vomiting lasting 1 month. Upon physical examination, a large (4.5×3.0 cm), firm, non-painful, single mass was present over the ventral to cervical trachea. The cat was moderately dehydrated. No other significant abnormalities were noted. The differential diagnosis for a cat presenting a ventral cervical mass should include branchial cyst, thyroglossal cyst, thyroid cyst, thyroid cyst adenoma, parathyroid cyst, parathyroid cyst adenoma, parathyroid carcinoma, thyroid carcinoma, salivary mucocele and abscess (Norsworthy et al 2002). Blood samples were taken for routine laboratory evaluations. Serum biochemistry, made at two different times, revealed total calcium moderately elevated, low-normal phosphate concentrations and a mild increase in creatinine (Table 1). Haematology and other serum biochemistry parameters were unremarkable (Table 1). The possible causes of hypercalcaemia include primary hyperparathyroidism, renal disease, granulomatous disease, malignancy with production of parathyroid hormone-related protein (PTHrP), hypoadrenocorticism, hypervitaminosis D or idiopathic hypercalcaemia.
Complete blood count results
Serum parathormone (8.0 pg/dl, reference range 2.0–13.0 pg/dl), total thyroxine (2.7 μg/dl, reference range 0.6–3.6 μg/dl) and free thyroxine (20.1 pg/dl, reference range 7.0–24.0 pg/dl) were normal. Feline leukaemia and immunodeficiency virus tests were both negative as was urine culture. Urinalysis revealed isosthenuria and low proteinuria (49.87 mg/dl) with 0.4 protein-to-creatinine ratio (Table 2). Sodium dodecyl sulphate-agarose gel electrophoresis (SDS-AGE) showed a mixed tubular proteinuric pattern with some bands between 14 and 25 kDa, a band at 25 kDa, and an albumin band and protein traces around 150 kDa (Fig 1).
Serum biochemistry and urinalysis


SDS-AGE of urine proteins. Blue arrows, tubular proteins; *, albumin; and pink arrows, glomerular proteins.
Thoracic and abdominal radiography did not identify any abnormalities. Abdominal ultrasonography identified multiple, small, hypoechoic nodules within the liver and mild increased cortical echogenicity in the kidneys. The cervical mass was slightly hyperechoic to the surrounding tissue and contained several hypoechoic areas. Unfortunately, unrepresentative material mainly constituted by erythrocytes limited cytopathological evaluation of fine-needle aspirates (FNA) of the mass.
On the basis of these findings, the mass was considered to have a probable parathyroid origin. Surgical exploration of the cervical region was recommended, but was not carried out at the owner's request. Symptomatic care consisted of correction of dehydration with physiological saline solution 0.9% (15 ml/kg/h, IV) to promote calciuresis; ranitidine (2 mg/kg bid, IV, Zantac; GlaxoSmithKline) to reduce gastric acidity and a specific renal diet (feline k/d; Hill's), in consideration of the renal damage revealed by SDS-AGE and by mild increase in creatinine, although the cat was not azotaemic. Moreover, this diet was selected because hypercalcaemia associated with low or low-normal serum phosphorous levels can be less dangerous because the mineralisation of soft tissues, primarily kidney, is correlated with the calcium-phosphate product (Feldman and Nelson 2004).
On day 3, the cat appeared adequately hydrated, stronger, and was eating on its own. Considering the animal's age and the fact that sedatives were necessary for manipulation, the owners declined any further investigations or follow up. After discharge from the hospital, in addition to a specific diet and ranitidine (2 mg/kg bid, OS, Zantac; GlaxoSmithKline), the cat was administered with prednisone (0.5 mg/kg bid, PO, Deltacortene; Bruno Farmaceutici) for its effects on the calcium metabolism and to reduce the inflammatory component of the mass.
Two months later the cat was euthanased, because it was very weak, lethargic, thin, inappetent and showed signs of dysphagia and dyspnoea related to the increased mass that compressed the oesophagus and trachea.
Upon necroscopy a soft mass approximately 60×45 mm in diameter was observed in the left ventral cervical region (Fig 2A). The thyroid and the parathyroid were not found despite careful dissection. The ovoid mass was attached to the trachea that appeared to be compressed. On sectioning the mass had a thick and irregular capsule and a heterogenous brownish colour. Multiple bloody cysts were also present. Macroscopic lesions in other organs or metastatic calcifications were not identified. Only in the liver were multiple small cysts filled with a clear serous fluid observed.

A – Voluminous mass located in the ventral cervical region attached to the trachea; B – Neoplastic cells organised in sheets separated by thin fibrous tissue (haematoxylin and eosin; figure obtained using Nikon Coolscope objective 20×; bar 50 μm).
Histopathological examination of the mass using haematoxylin and eosin stained sections (Fig 2B) revealed the presence of a thick fibrous capsule that surrounded a multicystic neoplasia. Most of the mass was occupied by necrotic and haemorrhagic tissue infiltrated by a large number of neutrophils. Only small irregular islands of neoplastic cells were disseminated into necrotic areas. At the periphery, the mass was organised in large sheets separated by fibrous tissue sometimes oedematous and infiltrated by erythrocytes. In some areas the cells were distributed in pseudolobules. At the margin of the mass a few thyroid nodules that were largely necrotic were present. Generally the neoplastic cells were polygonal with defined cellular borders, round to ovoid hyperchromatic nuclei and small eosinophilic cytoplasms sometimes containing diffuse small vacuolisations. In some areas these cells were pleomorphic with nuclei of different size and colour and ill-defined cellular borders. No metastases were identified. Immunohistochemical staining of the tumour mass was performed with rabbit anti-human calcitonin (prediluted, Biogenex, Norris Canyon Road, San Ramon, CA, USA), rabbit anti-human thyroglobulin 1:3500 in PBS (Dako, Corp. Grostrup, Denmark) and mouse anti-human Ki-67 1:100 in PBS (Dako, Corp. Grostrup, Denmark) antibodies using the avidin–biotin method (Vectastain, ABC Vector Laboratories, Burlingame, CA, USA) and diaminobenzidine (Sigma Aldrich, St Louis, MO, USA) as the substrate. Thyroid and parathyroid tissues of a clinically normal cat were used as positive controls. No staining was observed in neoplastic cells with anti-calcitonin and anti-thyroglobulin antibodies. Rare mitoses were identified in the tumour mass using Ki-67 immunostaining.
The neoplasm was classified as a carcinoma of the parathyroid gland on the basis of the characteristic of the tumour cells, the size of the mass, the diffuse presence of necrosis and haemorrhages, and the destruction of the thyroid tissue (Capen 2002). The examination of routinely stained histological sections of the kidneys revealed multifocal, moderate, chronic occasionally severe, non-suppurative interstitial nephritis. Diffuse glomerulonephritis characterised by mesangial proliferations, multifocal thickening of the Bowman's capsule, and focal flocculo-capsular adhesions were the most important microscopic findings, observed in both kidneys. Segmental or global hyalinosis/sclerosis were also seen in some glomeruli. Moderate multifocal tubular degeneration, interstitial hyalinosis and diffuse hyaline casts were also seen.
Parathyroid tumours in cats are uncommon and have generally been reported in older animals. Siamese cats may be predisposed, while no sex predilection has been documented. Most parathyroid lesions are classified as adenomas, while parathyroid carcinoma is only rarely found (Ogilvie and Moore 2001). The most common clinical signs reported in feline patients with parathyroid adenocarcinoma are anorexia and lethargy. Vomiting, polyuria, polydipsia, weight loss, weakness, constipation, reluctance to move, muscle tremors, and ataxia are seen occasionally (Kaplan 2002).
The diagnostic workup should include a complete blood count and serum biochemistry, urinalysis and selective analysis for thyroid hormones and PTH. Mild to marked hypercalcaemia with low to normal serum phosphorus is the most significant abnormality reported (Kallet et al 1991). Urinalysis, urea and creatinine concentrations should be monitored to evaluate possible renal damage related to hypercalcaemia with reduction of glomerular filtration and parenchymal mineralisation. Characterisation of proteinuria using SDS-AGE, in which the urinary proteins migrate based on molecular mass, can help to assess renal damage (Zini et al 2004). This technique may be considered a valid, non-invasive, and useful option for diagnosis of renal pathologies in both dogs and cats (Abate et al 2003). In our case there was a good correlation between SDS-AGE and histological exams for localisation of renal lesions. Additional findings may include isosthenuria, haematuria, bacteriuria, pyuria and crystalluria with or without calcium oxalate uroliths (Savary et al 2000). Parathormone levels are increased or normal. This implies that multiple samples should be analysed if a parathyroid tumour is suspected (Feldman and Nelson 2004; Mooney et al 2004). Abnormal PTH levels with negative PTHrP, a tumour product that has the ability to active PTH receptors, aid in distinguishing primary hyperparathyroidism from hypercalcaemia of malignancy (Feldman and Nelson 2004). In this case PTHrP was not determined as the mass was evident and the assay was not available.
A neck mass is often palpable, but ultrasonography may detect non-palpable masses and can aid in the location of the tissue of origin as well as provide assistance in FNA (Sueda and Stefanacci 2000). Cytopathological evaluation and selective hormonal analyses of cystic fluid should be performed whenever possible. Potential additional diagnostic methods include nuclear scintigraphy using technetium-labeled sestamibi that has commonly been used to identify parathyroid masses in human and, more recently, in dogs, but has not been formally evaluated in cats (Lurye and Behrend 2001).
Cervical exploratory surgery remains the only definitive diagnostic tool to confirm parathyroid masses; surgical excision of adenomas can result in rapid clinical improvement and good long-term prognosis. Surgery was reported to be curative in one cat with parathyroid carcinoma: 6 months after presentation the cat had no evidence of either local recurrence or metastasis and remained normocalcemic (Phillips et al 2003). Postoperative hypocalcaemia is uncommon, but serum calcium levels should be monitored within 24 h to 6 days after surgery. Supportive medical cure for parathyroid tumours may include the use of bisphosphonates, calcitonin and glucocorticoids to reduce severe hypercalcaemia (Feldman and Nelson 2004). In this case considering the mild hypercalcaemia and the impossibility of monitoring serum calcium levels, bisphosphonates and calcitonin were not used, but only a low dosage glucocorticoids.
The definitive diagnosis of parathyroid carcinoma was based on the anatomic and pathological findings. The report of multiple cysts in a cat parathyroid carcinoma has been reported in only two other instances (Marquez et al 1995; Phillips et al 2003). Renal histopathological findings and the proteinuric pattern show the presence of tubulo-interstitial damage in agreement with the presence of primary hyperparathyroidism (Kruger and Osborne 1995). The nephrocalcinosis can be absent for the moderate hypercalcaemia and normal phosphoremia (Kruger and Osborne 1995). The presence of membrane proliferative glomerulopathy, glomerulosclerosis and glomerular proteinuria may be correlated with neoplasm or merely a coincidence. In human medicine glomerulonephritis is known to be associated with malignant tumours, but in veterinary medicine such an association remains unclear (Loi et al 2004; Luyckx et al 2002).
