Abstract
Many owners are able and willing to perform home monitoring of blood glucose concentrations in diabetic cats. Once owners are familiar with the technique, they appreciate its advantages and show long-term compliance. The success of home monitoring hinges greatly on careful preparation and instruction of the owner. Owners must have ready access to veterinary support if needed. Initially, most owners call for advice, and several of them need repeated explanation or demonstration of the procedure. The frequency of re-evaluations of the diabetic cats by veterinarians is not affected by home monitoring. One of its major advantages is that it enables frequent generation of blood glucose curves. In complicated cases, more than one curve can, therefore, be performed at home before a treatment decision is made. According to preliminary data cats managed with home monitoring may have better glycaemic control than those managed without. However, those results need to be confirmed in a large group of cats.
Diabetes mellitus is a frequently seen disease in cats. The reported incidence varies from one in 50 to one in 400 cats, depending on the population studied (Rand 2004). Diabetes is a chronic illness, that requires continuing medical care and owner education to prevent acute complications and to secure adequate life quality. Long-term management of diabetic cats relies on the owner's observation of clinical signs and on periodic evaluation by a veterinarian. The latter includes evaluation of owner's opinion, measurement of body weight and determination of blood glucose and serum fructosamine or glycated haemoglobin concentrations (Reusch et al 1993, Miller 1995, Rand and Martin 2001, Feldman and Nelson 2004). Although fructosamine or glycated haemoglobin is helpful to determine the quality of metabolic control, they do not help identify the underlying problem in poorly regulated cats.
Blood glucose curves (BGC) are necessary to assess time and duration of insulin effects, the nadir and degree of fluctuation in blood glucose concentration, and identify the Somogyi phenomenon (Feldman and Nelson 2004). Until recently, BGC in diabetic cats were almost always performed in veterinary hospitals because most owners cannot collect venous blood samples for evaluation. In hospitalised patients, the concentration of blood glucose can be markedly influenced by stress or refusal to eat. Cats in particular are sensitive to stress caused by an unfamiliar environment or veterinary manipulation. Consequently, the BGC may show a continual increase in the blood glucose concentrations, or it may be elevated from the start. When the latter occurs, it is not possible to differentiate among stress-associated hyperglycaemia, insulin under-dosage and the many causes of insulin ineffectiveness and resistance. Hospitalised diabetic cats frequently refuse to eat, which may result in a marked decrease in blood glucose concentration. It is then difficult for the veterinarian to decide whether anorexia or overdose of insulin is the cause of low glucose concentration.
Self-monitoring of blood glucose in human diabetics
In human medicine, those problems have been largely eradicated with the introduction of self-monitoring of blood glucose concentrations (SMBG). For SMBG, patients obtain a drop of capillary blood by pricking a fingertip with a lancing device. The drop is then placed on a test strip, and the glucose concentration is measured using a portable glucose meter (PGM). The introduction of SMBG is regarded as the most important advance in the management of diabetes mellitus since the discovery of insulin (Watts and Keffer 1989). In the recent position statement of the American Diabetes Association, SMBG is recommended as an integral component of diabetes therapy (ADA 2004).
Capillary blood sampling in cats
Until recently, SMBG in diabetic animals was not thought possible. However, during the last years several methods have been developed for obtaining capillary blood by means of lancing devices manufactured for humans. In 2000, two methods of capillary sampling from the ear were described by Wess and Reusch (2000a). One method, using a conventional lancing device after pre-warming the ear with a hair dryer could only be performed in dogs. The other method, which was called ‘Vaculance method’ was used very successfully in dogs and in cats. To obtain a blood drop a new type of lancing device, the Microlet Vaculance (Bayer Diagnostics, Switzerland), was introduced. After lancing the skin, this device creates a negative pressure, which facilitates collection of an adequate amount of blood. Since the year 2000 this method has been exclusively used in our clinic (Fig 1).
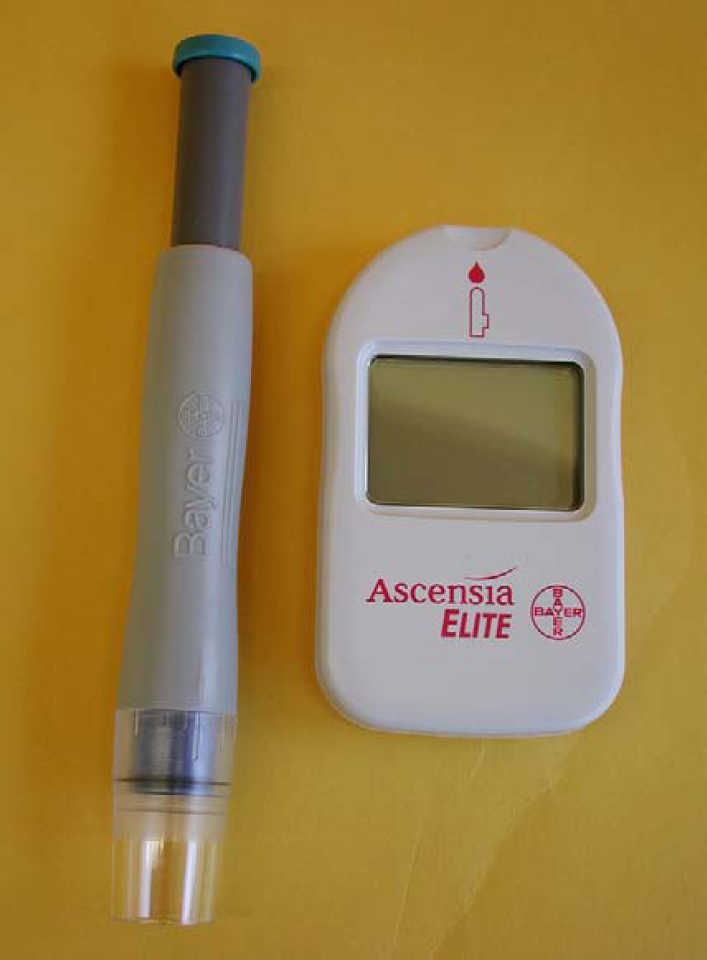
Equipment for the ‘Vaculance method’ of capillary blood sampling. The lancing device on the left is the Microlet Vaculance (Bayer Diagnostics, Switzerland). After lancing the skin, this device creates a negative pressure that facilitates collection of an adequate amount of blood. The portable glucometer on the right is the Elite (Bayer Diagnostics, Zurich). Since the end of 2002 the name has changed from Glucometer Elite to Ascensia Elite.
Briefly, the tip of the ear is held between the thumb and index finger and the entire surface of the outer pinna is held flat using the remaining fingers of the same hand. With the other hand, the lancing device is lightly placed on a non-haired area of the pinna so that an airtight seal is formed between the endcap of the device and the skin. When the plunger cap of the instrument is pressed, a lancet moves quickly once back and forth. Pressure between the endcap and the skin is maintained while the plunger is slowly released. The skin of the ear begins to slightly bulge up into the endcap, because of the developing negative pressure. The formation of a drop of blood is hastened by releasing the pressure, which is exerted on the surface of the pinna by the fingers. When an adequate amount of blood appears on the skin, the plunger is pressed down to release the negative pressure and the lancing device is removed. Then, the test strip in the PBGM is brought into contact with the blood drop and the required amount of blood is automatically absorbed on to the test strip (Fig 2).

Capillary blood sampling from the pinna using the ‘Vaculance method’. See text for details.
The buccal mucosa has been described as another site for capillary blood sampling in dogs by Fleeman and Rand (2000). We assume that this site is usually not feasible for cats. Alternatively to capillary sampling blood may be obtained by puncturing the marginal ear vein (Thompson et al 2002). This technique, called the marginal ear vein technique, has been successfully used in cats. However, so far it has only been used in the hospital by veterinarians and/or technicians and reports on its use by cat owners are lacking.
Use of portable glucose meters
Since the introduction of portable glucose meters in the 1970s many new models have been developed and marketed, offering a variety of improvements. Current blood glucose meters use one of two principles of measuring blood glucose concentration: reflectance photometric systems and amperometric biosensor systems (for details see Reusch et al 2001). The quality control of portable blood glucose meters has been a frequent topic of discussion in human medicine. Factors with a possible effect on the results of glucose measurements include variation in haematocrit, altitude, environmental temperature and humidity, hypotension, hypoxia and triglyceride concentrations (Skyler and Cohen 1997). Overall performance of these systems depends on analytical performance of the meter, quality of the test strips and proficiency of the user. In contrast to human medicine, there are very few studies concerning the quality control of portable blood glucose meters in veterinary medicine, and only two of them evaluate their use in cats (Link et al 1997, Wess and Reusch 2000b). In the study by Wess and Reusch (2000b) the accuracy of Glucometer Elite (Bayer Diagnostics), Glucometer DEX (Bayer Diagnostics), SureStep (LifeScan), Precision QID (MediSense) and Accu-Chek Simplicity (Roche Diagnostics) were investigated by use of error grid analysis. With the exception of venous blood measurements in cats obtained with the Glucometer DEX, all results were in the clinically acceptable zones A and B. The accuracy of Glucometer Elite (Bayer Diagnostics) and Accu-Chek Simplicity (Roche Diagnostics Inc.) were also evaluated by error grid analysis using capillary blood from the ear of cats, and again all measurements were in the clinically acceptable zones A and B (Fig 3). In people, most errors in SMBG are associated with blood collection or the use of the portable blood glucose meter. For cat owners, home monitoring can constitute a challenge and, therefore, it is important to minimise technical difficulties. In our experience, of all meters on the market, the Glucometer Elite (Bayer Diagnostics, Zuerich, Switzerland) is the easiest to operate: it has no buttons to press, turns on automatically when the test strip is inserted and requires a very small amount of blood (2 μl), which is automatically aspirated into the reaction chamber after contacting the test strip. The measurement range is between 1.1 and 33.6 mmol/l, the result is displayed after 30 s and the last 20 measurements are stored. However, as with all portable blood glucose meters, problems may occur. It is important that the test strip chamber be filled to the mark. When too small a volume of blood (<2 μl) is used, the acoustic signal, that normally marks the end of the aspiration of blood, erroneously sounds, and measurement follows, producing an inaccurate low reading (Fig 4). Blood glucose concentrations that are measured by portable blood glucose meters almost always vary slightly from those measured by a reference method. Values obtained by the Glucometer Elite mostly underestimate true concentrations, whereas other glucometers may arbitrarily under- or overestimate true values, which was considered a disadvantage for clinical purposes. In the cat mean differences (underestimations) between the glucose concentration of capillary blood obtained by Glucometer Elite and that of venous blood obtained by the reference method (Autoanalyzer) were 0.4 mmol/l in the low glycaemic range (<4 mmol/l), 1.2 mmol/l in the normal range (4–8 mmol/l) and 1.9 mmol/l in the high glycaemic range (>8 mmol/l). Maximum difference was 4.6 mmol/l and occurred in the blood glucose range between 16 and 23 mmol/l (Wess and Reusch 2000a).

Error grid analysis for the Glucometer Elite using capillary blood. Values in zone A (clinically accurate) or zone B (benign estimate errors) are clinically acceptable. Values in zone C (unnecessary corrections), D (dangerous failure to detect) or E (erroneous treatment) are potentially dangerous and, therefore, not acceptable from a clinical point of view. For the Glucometer Elite all values were in zone A or B. (From Wess and Reusch, 2000a, with permission.)
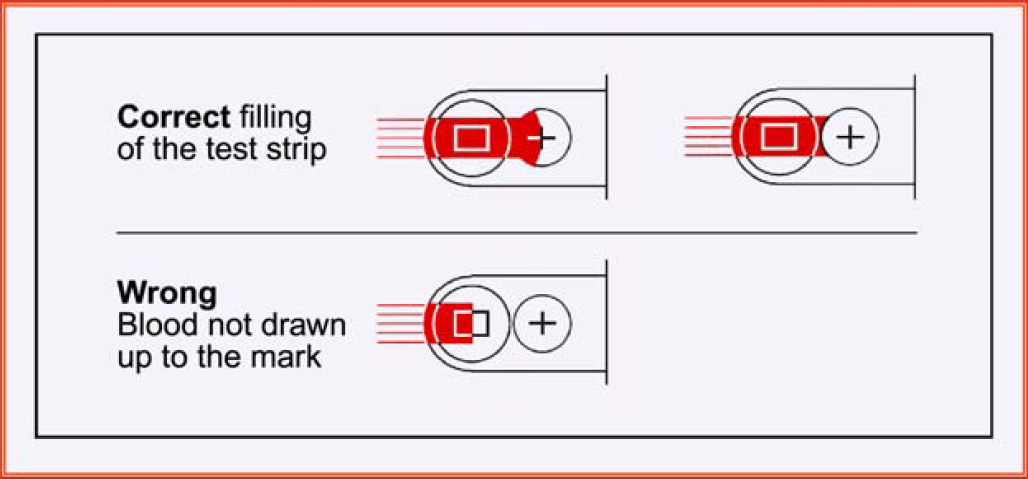
Correct and incorrect filling of the test strip. If the chamber is inadequately filled, the normal acoustic signal will still be heard, however, the blood glucose concentrations shown on the display will be inaccurately low.
In fall 2002 Bayer changed its brand name for their glucose monitor line. Since then Glucometer Elite is named Ascensia Elite, which has the same measuring conditions as described above for the Glucometer Elite (Fig 1). Recently a new Ascensia product has been launched called Ascensia Contour, which is currently the smallest meter on the market. It has several advantages over the Ascensia Elite: the blood volume needed is only 0.6 μl instead of 2 μl, measurement is not started when blood volume is too low, omitting the potential error described above and no coding is required. Results are given within 15 s, and the measurement range is 0.56–33.6 mmol/l. Preliminary evaluation results indicate that blood glucose concentrations obtained by the Ascensia Contour correspond very well with those measured by a reference method.
Measurement of capillary blood glucose concentrations by cat owners (home monitoring)
Home monitoring has been part of the routine protocol for long-term management of diabetic pets in our clinic since 1999. During an initial study with healthy cats it was investigated what types of technical problems may arise when owners who are unfamiliar with any kind of blood collection perform the procedure. Besides restraint of the cat, generation of insufficient blood volume due to false handling of the lancing device and failure to fill the test strip up to the mark were most frequently encountered (Casella et al 2002). From those results it is advisable to stress and clarify those parts of the procedure during explanation and demonstration of the technique. In diabetic cats a stepwise introduction to home monitoring of blood glucose has been described previously (Reusch et al 2001, Casella et al 2004). The first step is a definitive diagnosis of diabetes mellitus. Additional tests may be indicated at this point; determination of serum fructosamine concentration is routinely performed. Insulin therapy with an intermediate-acting insulin (Caninsulin; Intervet) is started, using 1 or 2 U/cat bid depending on body weight. The owner then receives detailed information on various aspects of diabetes mellitus including dietary recommendations and careful instruction on insulin injection technique, and the concept of home monitoring is introduced for the first time. This consultation takes approximately 45 min. The second step consists of re-evaluation of the cat after 1 week. Observations made by the owner are discussed, a clinical examination is performed and fructosamine concentration and an in-clinic BGC over 12 h (blood sampling every 2 h) are determined. Insulin dosage is adjusted if necessary in steps of 0.5–1.0 U/cat bid. At the time of discharge, the importance of the BGC in the control of the disease is emphasised. In addition, the advantages of home monitoring for owner and cat are discussed and she/he is informed that this procedure can be started after the next re-evaluation.
The third step follows approximately 3 weeks after the diagnosis of diabetes. Once again, owner observations are assessed, and a physical examination and BGC and fructosamine concentrations are determined. The owner is now provided the opportunity to learn the technique of home monitoring. This requires a minimum of 30 min and consists of repeated demonstrations of the use of the lancing device and the portable blood glucose meter. The owner then performs the technique several times with her/his cat. The owner is also taught how to calibrate the blood glucose meter, check its accuracy using the control strips and record the blood glucose values on prepared forms. Home monitoring is not started before the third week after a diagnosis of diabetes mellitus. This allows the owner to become familiar with the disease and to gain experience with the injection of insulin. However, introduction of home monitoring is delayed to a later date if the owner does not seem ready for it. It is important that owners perform the technique on a regular basis to stay familiar with it and that re-assessments are carried out frequently (Casella et al 2004). According to the protocol used in our hospital (Reusch et al 2001) fasting blood glucose concentration is determined twice weekly and a BGC for 12 h (blood sampling every 2 h) is performed at least once monthly. The former serves to detect morning hypoglycaemia, in which case the owner is instructed to call the hospital. With regard to BGC the owner sends the results, and appropriate changes in treatment are discussed, if necessary, over the phone. During the first few weeks of home monitoring, periodic re-assessment of the entire procedure is performed at the hospital. Our specific protocol is to have the owner measure the fasting blood glucose concentration, inject the insulin and feed the animal at home. In the hospital the owner takes another blood sample from the ear while a veterinarian is watching the procedure. Possible technical problems can be discussed with the owner, and the owner's technique for blood collection and operation of the blood glucose meter can be assessed. At each visit it is critically evaluated if judgement on glycaemic control made by home monitoring is correct. Judgement on glycaemic controls can be made quite reliably by means of clinical signs observed by the owner, physical examination and changes in body weight (Goossens et al 1998). Additionally a BGC is performed at each visit and fructosamine concentration is measured and compared to previous concentrations. After a few weeks (or months, depending on the reliability of the owner) the re-evaluation in the hospital may be reduced to consideration of clinical signs, body weight and fructosamine measurements. From time to time the owner should be watched during blood sampling. Regular veterinary re-evaluation of a diabetic cat is recommended, even when the owner is experienced with home monitoring. In human medicine, it has long been known that many errors can occur during SMBG. The National Steering Committee for Quality Assurance in Capillary Blood Glucose Monitoring (National Steering Committee 1993) emphasised the importance of patient education, which includes repeated demonstration of the procedure and observation of the patient during SBGM. It recommended re-assessment of the technique 30 and 180 days post-training and yearly thereafter or sooner when results indicate possible procedural errors. In the future these recommendations should also apply to diabetic cats. Because home monitoring has not been used widely in veterinary medicine, there are no studies to indicate how often re-evaluation should be performed. Currently, we feel that the owner should determine a BGC once monthly and the patient be re-assessed a minimum of twice a year by a veterinarian (Reusch et al 2001).
Problems encountered in home monitoring
In a recent study owners of diabetic cats were asked to perform one BGC per month over a 4-month period and to fill in a questionnaire, covering all aspects of the procedure, after each curve (Casella et al 2002, 2003, 2004).
Twelve of 15 owners were able to determine blood glucose concentrations. Three of 15 owners were unable to perform any blood glucose measurements because their cats did not tolerate the procedure.
Problems encountered initially included producing negative pressure with the lancing device, producing a blood drop, restraining the cat, absorption of the blood drop and correct use of the test strips and the PBGM. At the end of the study, problems occurred only occasionally involving restraint of the cat, production of a blood drop and the generation of negative pressure using the lancing device. At the end of the study, home monitoring was considered difficult by only one owner, mostly straightforward by six and straightforward by five. Ten of the 12 owners performed blood glucose measurements without help and two owners required another person for help (Casella et al 2004).
It is important that owners have ready access to veterinary support, if required. The majority of our clients call for advice one or more times, especially after the start of home monitoring. Some have specific questions regarding the procedure, while others want reassurance that they are performing the procedure correctly. Sometimes support via telephone is not sufficient and additional explanation or demonstration of the technique must be provided. By watching an owner perform the procedure, a veterinarian can immediately identify and correct errors. The prerequisite for good owner instruction is a knowledgeable veterinarian, who is able to perform the procedure correctly and is aware of all potential sources of error. Usually, owners feel that their technique improves quickly, which leads to increased confidence in the test results. Many owners devise strategies for easier restraint of their cat and report that the cats tolerate blood collection better when placed in a favourite spot, such as a windowsill or bed or in a confined area such as a sink. The skin puncture does not seem to be painful, and the puncture sites are barely visible, even after numerous blood collections (Reusch et al 2001, Casella et al 2002).
Comparison between blood glucose concentrations measured at home and in the hospital
Up till now only a single study is available comparing glucose concentrations measured at home with those measured in hospital (Casella et al 2004). Twelve cat owners were asked to generate a BGC at home within 1 week before the next re-evaluation in the hospital, where another BGC was generated. Blood glucose concentrations were determined before and every 2 h after administration of insulin for a 12-h period. Insulin dosage was not changed between the two corresponding curves, and the ‘Vaculance method’ was used at home as well as in the hospital. On four occasions over a 4-month period, BGC were generated at home and in the hospital. Insulin dosage was adjusted based on the glucose nadir: when the nadir was <5 mmol/l, from 5 to <9 mmol/l, or ≥9 mmol/l, the insulin dosage was decreased, remained unchanged or was increased, respectively.
Interestingly, glucose concentrations measured in the hospital had the tendency to be lower than those measured at home. The nadir in the third- and fourth-curve comparisons and the mean glucose concentration in the fourth-curve comparison were significantly lower for hospital curves (Fig 5). In fact, we had assumed that due to stress caused by an unfamiliar environment, veterinary manipulation and reduced physical activity during the stay in the clinic, the blood glucose concentrations would be elevated. There are several possible explanations for this finding. First, the differences could be due to technical errors made by owners at home. However, this appears to be unlikely because an inadequate amount of blood would have resulted in an erroneous low blood glucose measurement (Wess and Reusch 2000b). Second, one has to consider the possibility that the values at home were higher than those in the hospital because the cats were actually more stressed when untrained owners collected capillary blood. To investigate this possibility, tolerance of the procedure at home was classified for each BGC as good, moderate and poor. Statistically no association was found between blood glucose levels and tolerance of the procedure. Third, it has to be considered that the glucose levels at home reflected the true glucose concentrations and that, however, levels in the hospital were ‘false low’. A plausible reason could be that hospitalised cats frequently refused to eat. In 10 pairs of BGC obtained from seven cats during the third- and fourth-curve comparisons the nadir was lower in the hospital than at home and at least five of the seven cats refused to eat. In a similar study of diabetic dogs, a decrease in food intake was believed to be the most likely explanation for lower blood glucose concentrations in hospitalised dogs compared to dogs evaluated at home (Casella et al 2003). However, over the years we also experienced the opposite findings in cats with diabetes mellitus, eg, ‘false’ high glucose levels in the hospital caused by stress (Fig 7). In addition to comparing home and hospital curves statistically, the potential treatment decisions based on the BGC of each cat were also compared. In 62% of cases (case=one pair of BGC) treatment decisions would have been the same. In 38% of the cases, treatment decisions based on the hospital curves differed from those of the home curves; in three cases, the treatment decisions would have been completely contrary (increase vs decrease of insulin dosage) and in the remaining 11 cases, they would have differed, although probably with little clinical consequence (Fig 6). So far no data are available about the reproducibility of BGC generated at home. BGC are known to vary from day to day (Feldman and Nelson 2004). Similarly, Fleeman and Rand (2003) reported that in diabetic dogs, BGCs determined in the clinic on successive days varied widely. Studies are currently underway in our clinic to compare the reproducibility of BGC performed at home and in the clinic.

Comparison of glucose nadirs of home and hospital blood glucose curves of 12 cats with diabetes mellitus. (From: Casella, Hässig, Reusch 2005.)

Graphic representation of the glucose nadirs for all blood glucose comparisons. The x-axis represents values from hospital curves and the y-axis represents values from home curves. In 23 of 37 blood glucose pairs, the decisions regarding treatment were in agreement (zone A). In 14 of 37 pairs, the decision regarding treatment differed in one of five different ways. Zone B – hospital curve: decreased insulin; home curve: insulin unchanged. Zone C – hospital curve: decreased insulin; home curve: increased insulin. Zone D – hospital curve: insulin unchanged; home curve: decreased insulin. Zone E – hospital curve: insulin unchanged; home curve: increased insulin. Zone F – home curve: increased insulin; home curve: insulin unchanged.
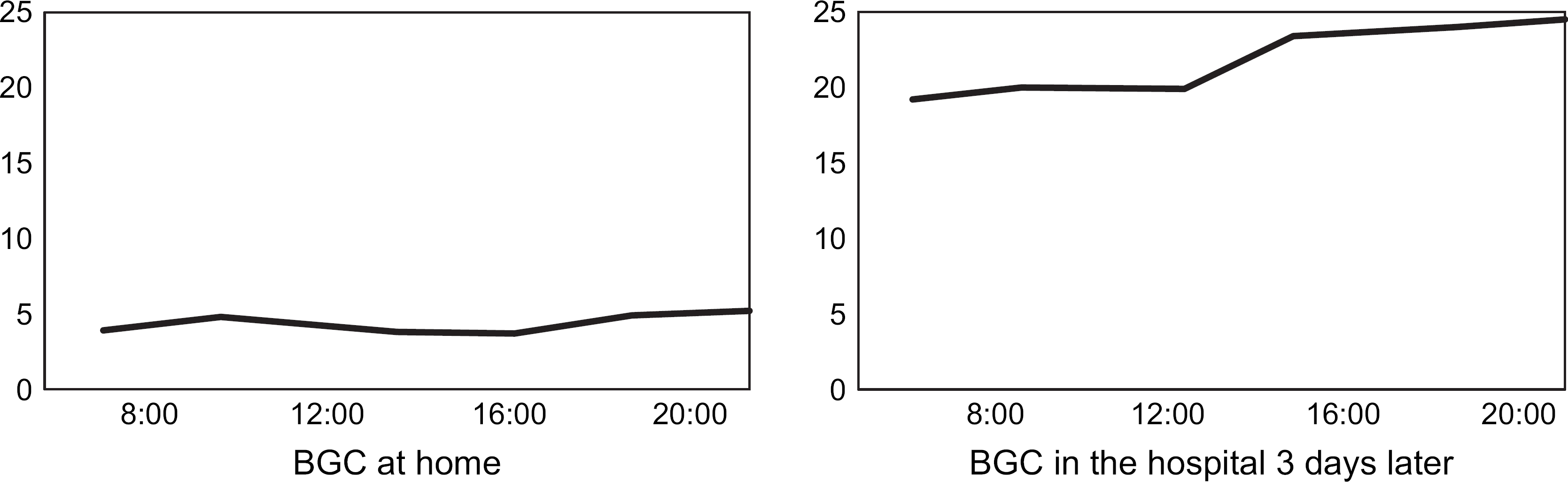
Blood glucose curve at home and in the hospital 3 days later in a cat with diabetes mellitus (spayed female domestic shorthair, 4.7 kg). The cat was diabetic for 1 year and received 0.5 U/cat Caninsulin bid at the time when the two curves were generated. According to the owner the cat was doing well and had no signs of diabetes mellitus. The high blood glucose levels measured in the hospital were assumed to be caused by stress. Insulin dosage was not changed and the cat continued to do well.
At the moment we are unable to prove whether hospital or home curves reflect the ‘true’ blood glucose concentrations. In a study of Goossens et al (1998) glycaemic control based on owners’ opinion was good in 57%, moderate in 39% and poor in 4% of diabetic cats; home monitoring was not performed. In the study with home monitoring (Casella et al 2004) glycaemic control based on owners’ opinion was good in 67% and moderate in 33% of the cats, in none of the cats was glycaemic control thought to be poor. As the treatment decisions were exclusively based on the BGC generated at home, and glycaemic control was better than in the study of Goossens et al (1998) without home monitoring, we strongly believe that home monitoring will improve metabolic control in diabetic cats.
Long-term compliance with home monitoring in cats with diabetes mellitus
In a recent study the objective was to determine whether home monitoring of diabetic cats with all its practical demands is feasible long term. Additionally, the influence of home monitoring on the frequency of re-evaluation at the hospital was assessed (Kley et al 2004). Seventeen of 26 cat owners (65%) who were taught the procedure used home monitoring on a regular basis. Home monitoring was performed for more than 1 year by 13 of the 17 owners. At the time of the study, one owner had been performing home monitoring for almost 4 years and many of the cats were still undergoing home monitoring. Therefore, a general statement on the length of time that cat owners are willing to perform the procedure cannot be made at present. However, the results clearly indicate that many owners are willing to perform home monitoring long term. Eight of 26 cat owners were not able to perform home monitoring because of technical difficulties or the fractious nature of the cat, and six of the eight individuals ceased home monitoring after the first attempts. It is possible that some of the cats were indeed uncooperative, although the owners themselves may have had psychological reasons for such rapid discontinuation of home monitoring. One cat was euthanased a few days after home monitoring was started (ie, 3 weeks after diagnosis) because the owner found management of the disease to be too difficult. Possibly, the additional burden of home monitoring overwhelmed the owner and prompted a decision to proceed with euthanasia of the cat. Therefore, veterinarians must carefully determine whether and when an owner is psychologically prepared to start home monitoring of a diabetic cat. One of the major advantages of home monitoring is that it facilitates frequent generation of BGCs. All cat owners in the study of Kley et al (2004) believed that home monitoring had raised their self-confidence with regard to their ability to manage the disease in their pets. Owners were reassured to know that if clinical changes developed in their cat, they were able to measure blood glucose concentration to determine whether hyper- or hypoglycaemia was a problem. Most cat owners complied with the home monitoring protocol and performed a BGC every 2–4 weeks. Human diabetic patients quickly lose interest in self-monitoring of blood glucose if they are not taught how to interpret and act on test results and if physicians do not view the test results during patient re-evaluation (Burritt et al 1991). This is probably applicable to owners of diabetic pets as well. Therefore, it is advisable to inform all owners of diabetic cats who undertake home monitoring of the optimal range of blood glucose concentration and the most frequently detected patterns of glucose response. However, it should be recommended that adjustments in the insulin dosage be made after consultation with a veterinarian. Fifteen of the 17 owners mentioned above complied with that recommendation and only two changed their cat's dosage of insulin without consultation. The frequency of re-evaluations of diabetic cats was not substantially affected by home monitoring. Although the number of re-evaluations did not differ significantly between cats managed with and without home monitoring, cats managed with home monitoring were more frequently re-evaluated in general. It is not known whether this was a result of the home monitoring programme itself or because of a stronger commitment to the care of their cats by the owners using it (Kley et al 2004).
