Abstract
A 9-year-old Burmese cat was presented for investigation of a subacute onset of bilateral forelimb paresis. Magnetic resonance imaging of the cervico-thoracic vertebral column and brachial plexus revealed a bilaterally symmetrical, severe and diffuse swelling of the spinal nerves forming the caudal part of the brachial plexus. Histopathology of the abnormal nerve roots, spinal nerves and brachial plexi showed inflammatory and marked proliferative changes with similar features to that of hypertrophic neuritis of man. Hypertrophic neuritis in man is a rare, tumor-like, chronic inflammatory peripheral nerve disorder of unknown origin most frequently involving the brachial plexus.
A 9-year-old male neutered Burmese cat was presented to the Neurology Unit at the Animal Health Trust, Newmarket, UK, for investigation of a 6-day history of subacute onset, progressive, bilateral forelimb weakness. Prior to the onset of neurological signs, the cat was reported to have been intermittently lame on the right forelimb for 2 months, but no clinical abnormality had been found by the referring veterinarian. The cat had no previous medical history, exposure to toxins or trauma. The cat was not on any treatment at the time of referral and routine vaccinations were overdue. On presentation, the only abnormality noted on physical examination was moderate atrophy of the distal muscles of both forelimbs. Neurological examination revealed a severe, bilateral, forelimb paresis, worse on the left side. Severe postural reaction deficits were present in both forelimbs whilst postural reaction testing on the hindlimbs was normal. No abnormality was detected on cranial nerve examination. Segmental spinal cord reflexes were intact in the hindlimbs but severely reduced in the forelimbs. Cutaneous trunci reflex was absent bilaterally and nociception was reduced in the dermatomes corresponding to the radial, ulnar and median nerves. The neuroanatomic diagnosis was considered consistent with a focal (C6–T2 spinal cord segments) or multifocal (bilateral brachial plexus) neurolocalization. Inflammatory or infectious nervous system diseases or a multifocal neoplastic process were considered as the most likely differentials for these anatomic diagnoses.
Complete blood count and serum biochemical profile were performed on admission and were both found to be within normal limits. The cat was seronegative for feline leukemia virus and feline immunodeficiency virus (Snap Combo Plus, Idexx).
Thoracic radiographs were normal. Electrodiagnostic testing was performed under general anesthesia; concentric needle electromyography (EMG) of the major muscle groups of the fore- and hindlimbs, epaxial muscles of the cervical, thoracic, lumbar and sacral areas as well as temporalis and masseter muscles was performed and showed moderate abnormal spontaneous electrical discharge activity in the triceps brachii, extensor carpi radialis, digital extensors, superficial and deep digital flexors, and carpal flexors on both sides. The motor nerve conduction velocity for the ulnar nerves and the muscle-evoked responses were within normal limits.
Magnetic resonance (MR) imaging of the cervico-thoracic vertebral column and brachial plexus area was performed with a 1.5 Tesla super conducting magnet (General Electric 1.5T Sigma MR scanner) (Fig 1). The following pulse sequences were used: T1-weighted scans pre- and post-contrast (following intravenous administration of 0.1 mmol/kg body weight of gadopentetate dimeglumine (Omniscan; Nycomed, Norway)) in the transverse plane and T2-weighted scans in the transverse, dorsal and sagittal planes. Abnormalities detected on the MR images consisted of a bilaterally symmetrical, severe and diffuse swelling of the spinal nerves exiting at the C6–7, C7–T1 and T1–2 intervertebral foraminae. The origin of the right C6–7 nerve root created a large, lateral extramedullary mass resulting in moderate lateral spinal cord compression. The diffuse swelling of the spinal nerves was followed on these scans for several centimeters well into the axillae. The swollen nerves were hyperintense to skeletal muscle on T2-weighted and isointense on T1-weighted images. Post-contrast T1-weighted images revealed mild enhancement, especially of the periphery of the nerves. Cerebrospinal fluid (CSF) collected from the caudal lumbar site showed a mild increase in nucleated cell count (15 cells/μl – reference value <6 cells/μl) with 61% small lymphocytes, 33% neutrophils and 6% monocytes on a differential cell count. CSF protein concentration was markedly elevated (2.61 g/l – reference value <0.45 g/l). Bilateral brachial plexus neuritis, or a multifocal neoplastic process affecting the brachial plexi, were considered as the most likely differential diagnoses. Treatment with prednisolone 1 mg/kg orally twice daily was instituted, and after 5 days of treatment, the cat was showing moderate recovery of motor function on the right forelimb although failed to improve on the left forelimb. Chlorambucil 0.5 mg/kg orally every other day was then added to the prednisolone. Unfortunately, the cat's neurological condition did not improve further after 20 days of treatment. The owner elected for euthanasia and consented for post-mortem evaluation.
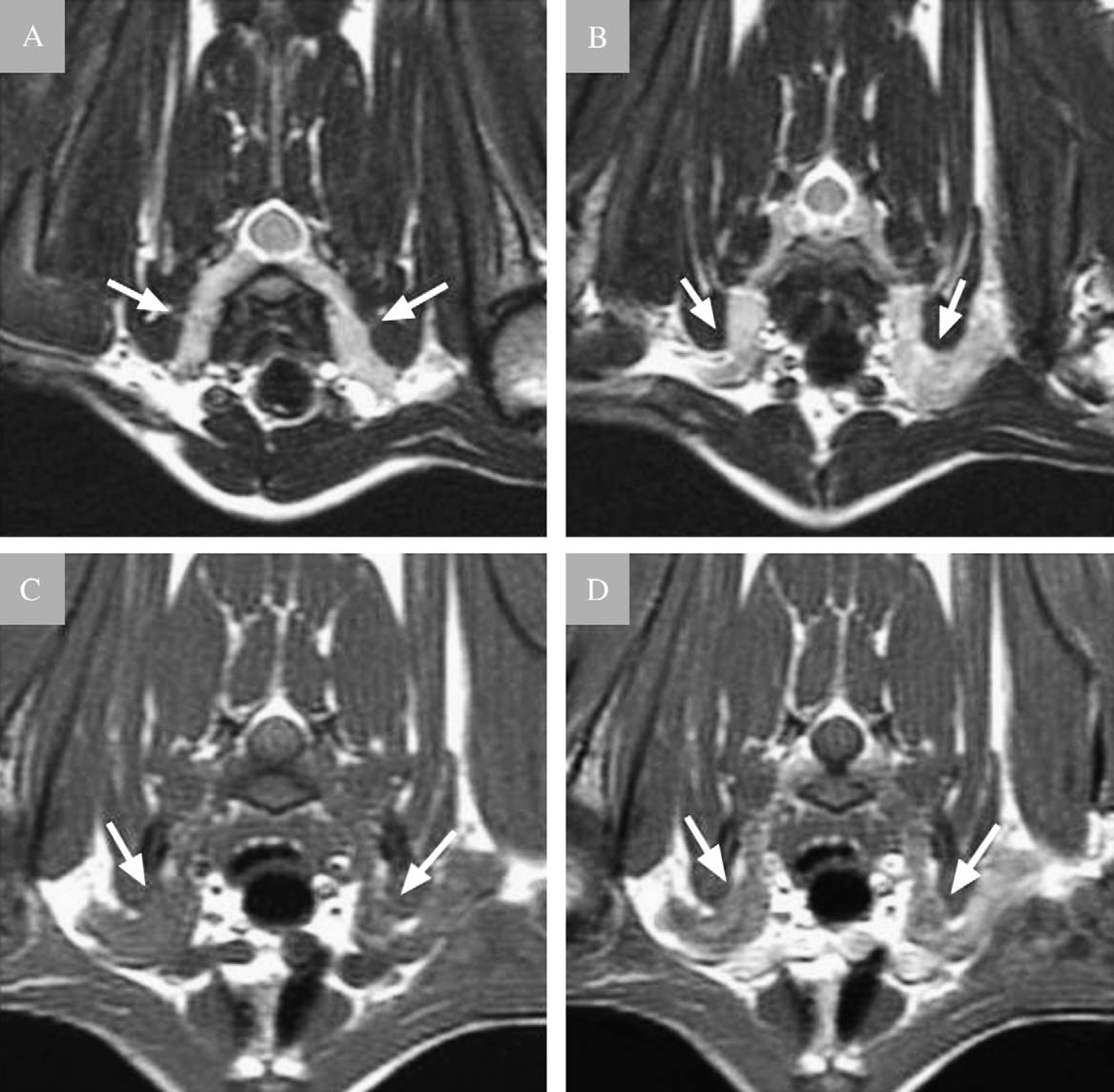
Transverse T2-weighted spin echo MR images (echo time (TE)=6000 ms, repetition time (TR)=89.4 ms, slice thickness (ST)=3 mm) of the cervico-thoracic region at the level of the C7–T1 (A) and T1–2 (B) intervertebral spaces, and transverse pre- (C) and post-contrast (D) T1-weighted spin echo MR images (TE=600 ms, TR=10 ms, ST=3 mm) at the level of the T1–2 intervertebral space. The C7–T1 spinal nerve and proximal part of the brachial plexus (arrows) are markedly swollen bilaterally and are hyperintense to skeletal muscle on T2-weighted image (A). At the level of the T1–2 intervertebral space, the mass (arrows) can be seen on both sides in the axillary region. The mass is markedly hyperintense to skeletal muscle on the T2-weighted image (B), isointense to skeletal muscle on the T1-weighted image (C) and enhances mildly following intravenous contrast medium (gadopentetate dimeglumine) injection (D).
Gross pathological changes were limited to the presence of bilateral brachial plexus enlargement with marked thickening of the spinal nerve roots exiting at the C6–7, C7–T1 and T1–2 intervertebral foraminae (Fig 2). The spinal cord was grossly normal apart from being moderately compressed on the right side by the thickened C6–7 nerve root. Microscopic examination of the abnormal nerve roots, spinal nerves and brachial plexi showed inflammatory and marked proliferative changes within the proximal spinal roots which to a variable degree effaced their normal structure. In some areas, accumulations of macrophages, lymphocytes and plasma cells and mild collagen deposits occurred in a subperineurial location and within the nerve fascicles among surviving nerve fibers (Fig 3). Remaining axons were identified with Bielschowski silver stains. In other areas, large accumulations of collagen and whorls of spindle-shaped cells (sometimes perivascular) had replaced the myelinated axons (Fig 4). While these whorls sometimes showed an arrangement around small capillaries, the silver stain did not suggest that they were ‘onion bulbs’. No organisms could be detected using Zeihl-Neelsen stains. Immunohistochemistry was performed using a leukocyte panel and an anti-S-100 antibody. A few scattered anti-CD3-positive T-cells were present throughout the inflammatory infiltrates. Labelling with anti-BLA36 (a B-cell and monocytic marker) was found within the large accumulations of macrophages and the whorls of spindle-shaped cells associated with the enlarged nerve roots. Anti-S-100 antibody labelled the nerve roots as expected while the whorled cells gave a non-specific pattern of staining with this antibody. These histological findings were suggestive of a hypertrophic neuritis involving both brachial plexi. Within the dorsal funiculi of the spinal cord at the level of the enlarged roots was a focus of reactive astrocytes, confirmed by an anti-GFAP (glial fibrillary acidic protein) immunostaining.

Gross appearance of the brachial plexi and contributing spinal nerves. This post-mortem specimen shows the swollen nerves (arrows) as they exit ventrally from the intervertebral foramina (arrowheads).

Histological section (haematoxylin and eosin) (magnification 200×) of the distal part of the C6/7 abnormal nerve root showing subperineurial inflammation.

Histological section (haematoxylin and eosin) (magnification 200×) of the proximal part of the C6/7 abnormal nerve root showing whorls of spindle-shaped cells.
The brachial plexus is a technically and anatomically challenging area of the peripheral nervous system for diagnostic imaging, and investigation of suspected brachial plexus disorders thus it can represent a real challenge for the clinician. Before advanced imaging modalities such as computed tomography (CT) and magnetic resonance imaging (MRI), electromyography was the only consistently useful non-surgical modality in the diagnosis of these disorders (Carmichael and Griffiths 1981, Targett et al 1993). Survey radiography is usually unremarkable (Carmichael and Griffiths 1981, Wheeler et al 1986) and myelography is only useful when there is vertebral canal involvement (Wheeler et al 1986, Targett et al 1993). Ultrasonography has been proposed in dogs to recognize large nerve sheath tumors (Platt et al 1999). However, this modality is very much operator-dependent and it is difficult to image the whole proximal to distal course of the brachial plexus due to the limited access. Imaging techniques such as CT and MRI are now being used in humans and animals to assist with early diagnosis of disease affecting the brachial plexus and contributing nerve roots (McCarthy et al 1993, Platt et al 1999, Simpson et al 1999, van Es 2001, Rudich et al 2004). Contrast-enhanced CT examination has been used successfully in dogs with brachial plexus tumors and has been reported to be able to depict brachial plexus masses as small as 1.0 cm (McCarthy et al 1993, Simpson et al 1999, Rudich et al 2004). Reports of the use of ultrasound or CT in cats with brachial plexopathy are lacking. MR is the imaging method of first choice in human patients with suspected brachial plexus disease or for studying the extension of tumors in this region (Blair et al 1987, Rapoport et al 1988, Bowen et al 2004). As well as offering multiplanar capabilities, it shows more anatomical detail and better contrast between the nerves and the surrounding fat compared with CT (Mellanby et al 2003). Varying imaging planes have been proposed in the human literature to depict the brachial plexus (Rapoport et al 1988, Bowen et al 2004). The sagittal plane demonstrates the brachial plexus most consistently because the nerves and related vessels are seen in cross section. The axial plane is particularly useful for delineating the nerve roots as they exit the foramina, because the imaging plane is then parallel to the orientation of the nerve roots. The axial and coronal planes permit left-to-right comparisons (Bowen et al 2004). However, translation of these imaging strategies to the dog or cat is not appropriate because of the different orientation of the brachial plexus in relation to the spinal cord (parallel to the sagittal plane). In our experience, transverse images parallel to the intervertebral space using a large field of view centered on the vertebral column and extending to the distal third of the brachium are particularly useful to permit left-to-right comparisons and to detect any asymmetry that might indicate disease. On the sagittal view, the imaging plane is parallel to the orientation of most of the brachial plexus and contributing spinal nerves and can in most cases allow visualization of these neural structures in a single image. However, a detailed knowledge of the normal anatomy is required, as symmetry is harder to assess in the sagittal plane and small vessels can mimic or obscure plexus components. The dorsal plane view can show the spinal cord and part of the ventral rami in one image but visualization of all five rami in a single image is not possible due to cervical lordosis.
The MRI signal characteristics of a variety of disorders that can involve the brachial plexus in man have been reviewed (Rapoport et al 1988, Bowen et al 2004). However, signal changes of an enlarged brachial plexus on MRI are rarely characteristic of a specific disease process (Bowen et al 2004). Tissue biopsy using percutaneous needle sampling or surgical exploration is therefore necessary for histological diagnosis (Carmichael and Griffiths 1981, Sharp 1988, Platt et al 1999, Stumpo et al 1999, van Es 2001).
Differential diagnoses of peripheral nerve enlargement in cats based on imaging or gross pathological findings should include idiopathic (suspected immune-mediated) neuritis, infectious neuritis (Paulsen et al 2000), metabolic disease (Jones et al 1986) and neoplastic neuropathy (most frequently peripheral nervous system lymphoma) (Zaki and Hurvitz 1976, Spodnick et al 1992, Fox and Gutnick 1994, Lane et al 1994, Jones et al 1995, Summers et al 1995, van Es 2001). Infectious neuritis caused by a Mycobacteria species has been reported in a cat with enlargement of sciatic, common peroneal, and tibial nerves in one limb (Paulsen et al 2000). Histological findings in that case report bore some similarities with our cat, with a severe inflammatory infiltrate composed of lymphocytes, plasma cells and macrophages along the demyelinated nerve fibers. However, acid-fast organisms were not detected in our cat and the significant component of whorled, proliferating cells would not be consistent with mycobacteriosis. Unfortunately, culture or polymerase chain reaction was not performed in our case to fully rule-out infectious causes.
Enlargement of spinal nerves apparently compressed the spinal cord, resulting in the focal astrocytosis seen in the dorsal columns. Macroscopic enlargement of peripheral nerves has also been reported in cats with inherited primary hyperchylomicronemia (Jones et al 1986). This enlargement was attributed to an extraneural compression from lipid granuloma, a histological feature absent in our cat. Additionally, the cats with hyperchylomicronemia were much younger than the cat presented here.
Immunohistochemical staining may be helpful in the diagnosis of peripheral nerve tumors and in differentiation between neoplastic and non-neoplastic lesions. In our cat, the chronic inflammatory changes with fibrosis and a mixture of lymphocytes (some T-cells), plasma cells and macrophages were consistent with a chronic neuritis. The unusual features were that the whorls were often angiocentric, BLA36-positive spindle-shaped cells within nerve fascicles. These whorls were shown not to contain axons following evaluation with Bielschowski silver stain and, therefore, were not considered to represent areas of Schwann cell proliferation (‘onion bulbs’) following repeated episodes of demyelination and remyelination. Instead, these cells most likely represent a local proliferation of fibrocytic-like cells.
Brachial plexus neuritis is a rare condition in domestic animals with very few reports in the dog (Cummings et al 1973, Alexander et al 1974) and cat (Bright et al 1978). It has been suggested in these reports that the suspected neuritis was the result of a hypersensitivity reaction following prophylactic inoculations such as tetanus antiserum similar to serum neuritis in man (Cummings et al 1973, Bright et al 1978). The etiopathogenesis of serum neuritis in people is unknown although a variety of antecedent events, such as immunizations, viral infections, vaccination with viral antigens, surgical procedures, trauma, collagen vascular disease or childbirth have been associated with its development (Neundorfer et al 1984, Cusimano et al 1988, Dumitru and Liles 1989, Maas et al 1996, Fabian et al 1997, Stumpo et al 1999, Auge and Velazquez 2000).
In people, hypertrophic neuritis is a rare tumor-like, chronic inflammatory, focal or multifocal, mainly demyelinating neuropathy, of unknown origin (Stumpo et al 1999). Corticosteroid treatment is usually ineffective in these cases and progression of the disease is particularly variable between individuals (Stumpo et al 1999). The pathological findings in human with hypertrophic brachial plexus neuritis consist of unilateral or bilateral fusiform segmental swelling of the trunks of the brachial plexus, mimicking a nerve sheath tumor macroscopically (Cusimano et al 1988, Stumpo et al 1999). Although histological findings on our cat bore some striking similarities with this human condition, ‘onion bulb’ formation, considered as the hallmark of hypertrophic neuritis (Stumpo et al 1999), was lacking. It is, therefore, not known if our case represents an unusual form of idiopathic hypertrophic neuritis or a different pathological entity.
Our case emphasizes the principle that enlargement of the brachial plexus is not necessarily due to a neoplastic process and other differentials such as chronic hypertrophic neuritis must also be considered.
