Abstract
An 11-year-old cat was presented with an approximately 2-month history of dysphagia, intermittent regurgitation and weight loss. An oesophageal foreign body was identified on plain radiographs, and an oesophagotomy was performed to remove a large V-shaped bone from the caudal cervical oesophagus. A gastrostomy feeding tube was placed to allow nutritional support postoperatively. Medical treatment for oesophagitis was initiated after surgery. No complications were encountered and the cat was discharged 4 days after surgery.
An 11-year-old female neutered domestic shorthair cat was referred with a 2-month history of dysphagia, weight loss and intermittent regurgitation. Initial presenting signs to the referring veterinary surgeon were gagging and choking, associated with feeding. Apart from a significant reduction in weight, clinical findings were unremarkable. A change in diet was recommended and the cat was started on soft food. The majority of clinical signs persisted along with exaggerated swallowing efforts, regurgitation and a visible swelling in the cervical region. Radiographs were taken which showed a radiopaque foreign body at the thoracic inlet. No further treatment was provided and the case was referred.
On clinical examination at the Royal (Dick) School of Veterinary Studies, Hospital for Small Animals, the cat was bright, alert and responsive. Body condition was fair (score 3/9), with a weight of 2.5 kg. Moderate hypersalivation was present. A soft tissue swelling was found at the thoracic inlet, which appeared sensitive on palpation. On cardiac auscultation the cat had an arrhythmia, with runs of tachycardia (>300 beats per min). Femoral pulses were strong and symmetrical. There were no abnormalities on thoracic auscultation or percussion.
Blood samples were taken for routine laboratory evaluation. Haematology revealed a mild mature neutrophilic leukocytosis (WBC 22×109/l, RR 7–20; segmented neutrophils 17.6×109/l, RR 2.5–12.8) and mild monocytosis (1.32×109, RR 0.07–0.85). On serum biochemistry there was a slightly decreased albumin (26.1 g/l, RR 28–39) and increased potassium (5.8 mmol/l, RR 4–5); other parameters were unremarkable.
An electrocardiogram and echocardiography were performed. No abnormalities occurred during the recording and neither structural nor functional cardiac abnormalities were observed on echocardiography.
The cat was premedicated with acepromazine (ACP: Novartis) (0.05 mg/kg) and morphine sulphate (Morphine; Evans) (0.3 mg/kg), administered intramuscularly. General anaesthesia was induced with propofol (Rapinovet; Schering-Plough) (4 mg/kg) intravenously, and was maintained with isoflurane (Isoflo; Abbott) in a mixture of oxygen and nitrous oxide using a non-rebreathing system (Ayres T-piece). Prophylactic antibiotics (cephazolin (Kefzol; Lilly) 20 mg/kg IV) were given at induction and repeated every 90 min throughout the surgery.
Cervical and thoracic radiographs were repeated under general anaesthesia. On the lateral view, a large V-shaped radiopaque foreign body was identified, at the thoracic inlet, surrounded by increased soft tissue opacity (Fig 1). No attempt at endoscopic removal was made owing to the positioning and size of the foreign body, the chronicity of the problem, and the high risk of perforation.
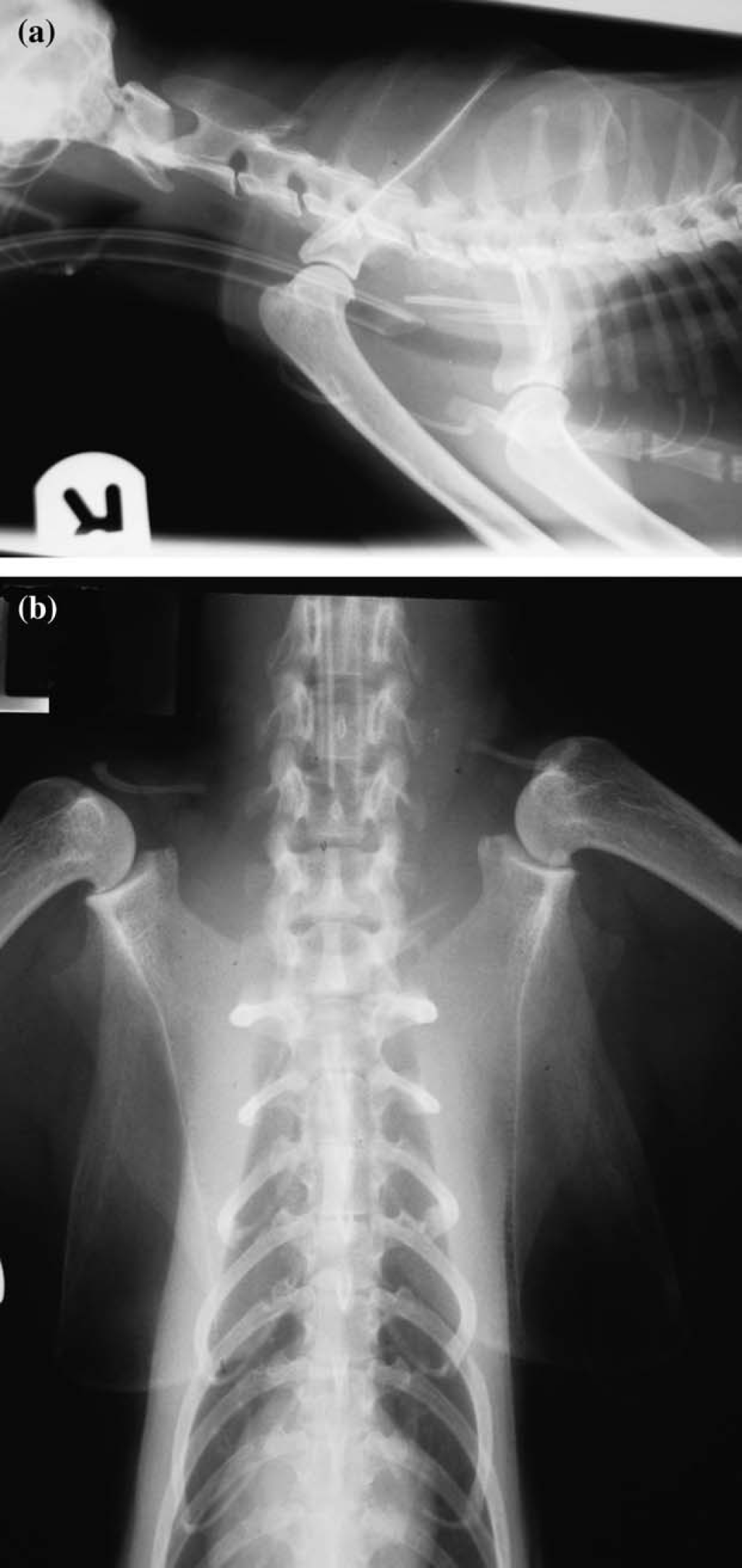
Lateral and ventro-dorsal radiographic views showing a large V-shaped radiopaque foreign body at the thoracic inlet, surrounded by increased soft tissue opacity.
A distal cervical oesophagotomy was carried out, using a ventral midline approach. The oesophageal wall was incised longitudinally (Fig 2). A hard bone presumably a clavicle from a wild bird was removed (Fig 3). The lumen of the oesophagus was inspected. There was no visible perforation, although significant damage to the oesophageal mucosa and mural thickening were identified, consistent with chronicity. The oesophagotomy incision was closed with 2 metric poliglecaprone (Monocryl; Ethicon) in a single layer appositional pattern. A gastrostomy feeding tube (12 French silicone percutaneous feeding tube; Cooks) was placed to allow nutritional support postoperatively and to bypass the surgical site to facilitate healing.
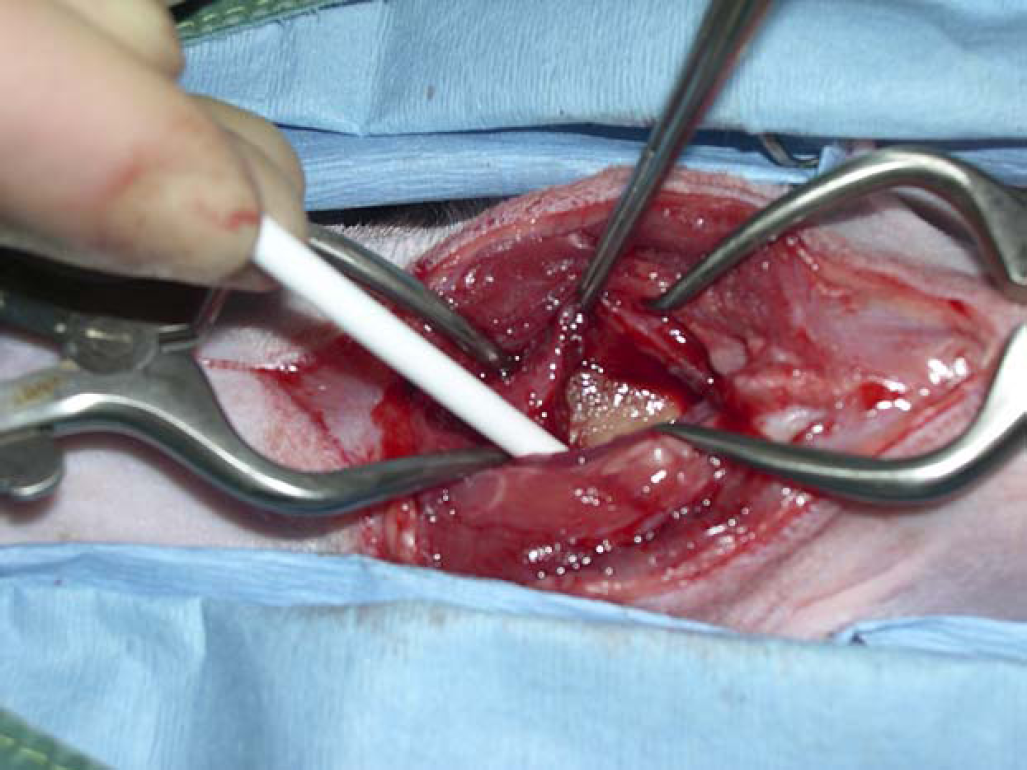
The oesophageal wall was incised longitudinally to remove the foreign body in a distal cervical oesophagotomy using a ventral midline approach.

A hard bone, presumably a clavicle from a wild bird removed from the oesophagus.
The cat remained in the intensive care unit for 2 days after surgery. Treatment with sucralfate (Antepsin suspension; Chugai) (250 mg orally every 8 h) and ranitidine (Zantac suspension; Glaxo) (3.5 mg/kg via the feeding tube q12h) was initiated. Antibiotic therapy with cephazolin (Kefzol; Lilly) (20 mg/kg intravenously q12h) and intravenous fluid therapy with an isotonic crystalloid solution (Isolec; Ivex Pharmaceuticals) (2 ml/kg/h) were administered. Analgesia was provided with buprenorphine (Vetergesic; Alstoe) (0.01 mg/kg intravenously every 8 h). A complete nutritional supplement (Fortol; Arnolds) was given by feeding tube commencing 24 h after surgery. On the second day, intravenous fluid therapy was stopped, antibiotics continued as an oral preparation (cephalexin (Ceporex; Schering-Plough) 20 mg/kg via feeding tube q12h) and small amounts of food were gradually introduced.
The cat progressed well, was eating voluntarily, and was discharged 2 days later. Treatment with sucralfate and ranitidine was carried on a further 2 weeks and the owner advised initially to feed blended or kitten food. The feeding tube was removed 1 week later, by the referring veterinary surgeon.
Interestingly, the cardiac arrhythmia was not noticed following surgical removal of the foreign body. The authors suspected that was likely to be a sinus tachycardia, which is a common stress related disorder in cats.
Oesophageal foreign bodies (FB) are a sporadic problem in cats, excluding linear FB extending from entrapment at the base of the tongue. Exposure usually occurs because of their hunting or playing behaviour (Johnson 1994). Ingestion of V-shaped avian bones (wishbones) has been described as a reason of obstruction of the pharynx and proximal oesophagus (Rendano et al 1988).
FB may lodge in the oesophagus after direct ingestion or may become lodged when vomited from the stomach (eg, with hairballs) (Sellon and Willard 2003). The majority are found at the thoracic inlet, heart base or epiphrenic area as extra-oesophageal structures limit dilation at these sites. The FB may be too large to pass these points or may have sharp edges that become embedded in the oesophageal mucosa (eg, wishbones, which easily perforate the mucosa). The presence of a FB within the oesophagus stimulates peristaltic activity (Hedlund 1997). If it remains at one location for several days, repeated peristaltic waves over the FB can produce pressure necrosis of the mucosa, submucosa and external layers of the oesophageal wall at the contact points. The extent of secondary oesophageal damage depends on the type of object, its size and shape, and the duration of time in contact with the mucosa (Johnson and Sherding 2000, Gualitiere 2001). Oesophagitis typically results and interferes with oesophageal motility and the gastro-oesophageal reflux barrier. Food which cannot pass the obstruction accumulates and may eventually be regurgitated or cause proximal oesophageal distention. Distention disrupts normal neuromuscular function and decreases peristalsis (Hedlund 1997).
In cats oesophageal obstruction is less common than other gastrointestinal obstructions (Bebchuk 2002). The common clinical signs of oesophageal FB are dysphagia and regurgitation, although the exact presentation depends on the level and degree of obstruction. If the obstruction is incomplete, allowing the movement of liquid or semi-solid food, it can be present for an extended period of time before diagnosis (Parker et al 1989) and ultimately signs of chronic wasting such as emaciation may be observed, as in this case (Bebchuk 2002). Other clinical signs may include gagging, excessive salivation, retching, inappetence, dehydration and restlessness (Hedlund 1997). Barrs et al (1999) reported acute collapse caused by oesophageal obstruction. Signs of depression, anorexia, fever, cough and dyspnoea suggest secondary aspiration pneumonia or oesophageal perforation with secondary mediastinitis/pyothorax (Sherding 2001). Diagnosis of oesophageal FB is usually based on physical examination, clinical signs and diagnostic imaging. In the present case, the foreign body could be identified on survey radiographs. Generally radiopaque FB can be detected on good quality radiographs. Radiolucent FB will occasionally cause accumulation of gas and fluid cranial to the obstruction that can be recognized on survey radiographs but confirmation of radiolucent FB may require a positive-contrast oesophagram (Guilford and Strombeck 1996, Aronson et al 2000).
Endoscopy allows visualization and location of the FB, and the majority can be successfully removed without recourse to surgery. Damage to the adjacent oesophageal mucosa can be assessed allowing formulation of an appropriate therapeutic plan (Spielman et al 1992). Traction with forceps has been successfully reported in one case by Court et al (1994).
In a retrospective study from the University of Bristol (Moore 2001), 51 of 65 dogs, primarily terrier breeds, had oesophageal foreign bodies successfully retrieved under fluoroscopic control; despite some short-term complications the long-term outcome with low rates of complications or recurrence supported the use of fluoroscopic-guided removal as an alternative to endoscopy in dogs. However, there is no information regarding this method in cats.
Surgical removal is recommended when the FB cannot be removed by other means, the risk of oesophageal perforation or laceration is high, or there is evidence of perforation (Michel et al 1995).
As all animals with oesophageal FB have some degree of oesophagitis, medical treatment is imperative for at least 7–10 days after removal (Spielman et al 1992). Following the retrieval of FB, oral food and water are withheld for 24–48 h and parenteral fluids are administered (Bright 2000). It is essential to maintain adequate nutrition after surgery, if necessary by means of an indwelling gastrostomy feeding tube. The ulcerated oesophagus is subject to secondary infection; therefore, broad-spectrum antibiotics are indicated in animals with severe ulceration and/or perforation (Washabau 2000).
Decreasing gastro-oesophageal reflux and promoting gastric emptying can be achieved with either cisapride (Prepulsid; Janssen-Cilag) or metoclopramide (Maxolon; Shire). Gastric proton pump inhibitors, such as omeprazole (Losec; Astra-Zeneca) and lansoprazole (Zoton; Wyeth) or histamine H2-receptor antagonists, such as ranitidine (Zantac; Glaxo) or famotidine (Pepcid; MSD) should be administered to decrease gastric acidity, and oral sucralfate suspension (Antepsin; Chugai) should provide mucosal cytoprotection (Spielman et al 1992, Sherding 2001, Willard and Weyrauch 2000).
The two major postoperative complications of oesophageal surgery are wound dehiscence, infection and stricture formation (Parker and Caywood 1987). Glucocorticoid therapy may be used to minimize the likelihood of stricture formation secondary to severe oesophagitis (Johnson 1994, Washabau 2000). An anti-inflammatory dose of prednisolone (0.5–1.0 mg/kg/day) is used to prevent fibroplastic proliferation and contraction. However, their benefit in animals has not been proven (Sellon and Willard 2003) and is not routinely recommended by all authors.
