Abstract
Multiple myelomas are uncommon neoplasms of the bone marrow of cats [Weber NA, Tebeau CS (1998) An unusual presentation of multiple myeloma in two cats. Journal of the American Animal Hospital Association
Multiple myelomas are plasma cell neoplasms of the bone marrow (Osborne et al 1968). Reports of multiple myeloma in cats are infrequent; there are 34 reported cases with no series describing more than six cats (MacEwen and Hurvitz 1977). Multiple myeloma accounts for less than 1% of feline haematopoietic neoplasm and plasmacytomas are rarer than multiple myelomas (Eastman 1996, Jacobs 1994).
Plasma cell tumours originating in the bone marrow and involving other organs are multiple myelomas, while solitary plasma cell tumours in organs other than bone marrow are plasmacytomas. They usually occur in aged cats, with no breed or sex predisposition (Jacobs 1994, Eastman 1996, Vail 2000). Neoplastic plasma cells secrete large amounts of paraproteins that appear as a monoclonal spike in serum protein electrophoresis commonly in the gamma globulin region (Weber and Tebeau 1998). There appears to be no relationship with feline leukaemia virus (FeLV) or feline immunodeficiency virus (FIV) (Jacobs 1994, Weber and Tebeau 1998, Vail 2000).
Presenting signs tend to be insidious in onset and non-specific, including depression, anorexia and weight loss. There have also been isolated cases with hindlimb paresis, ataxia or lameness (Mills et al 1982, Jacobs 1994, Weber and Tebeau 1998, Bienzle et al 2000).
There is little information on the responsiveness of multiple myeloma to chemotherapy; these patients are severely immunocompromised (Vail 2000). This, combined with administration of immunosuppressive drugs, indicates the need for concurrent administration of antibiotics at the beginning of therapy to prevent opportunistic bacterial infection (Eastman 1996, Weber and Tebeau 1998). The disease is usually progressive and fatal (Eastman 1996).
Complications of multiple myelomas seen in this cat study were similar to those seen by Hammer and Couto (1994) in the dog. They included renal failure, haemostatic abnormalities, infection and spinal cord compression.
Diagnosis of multiple myelomas requires meeting at least two of the following criteria:
Paraproteinaemia or monoclonal gammopathy.
Radiographic evidence of osteolytic bone lesions.
Bence Jones proteinuria.
Greater than 5% neoplastic plasma cells in the bone marrow.
These criteria were originally developed for use in the dog by MacEwen and Hurvitz (1977), although they have also been applied to the cat.
The purpose of this study was to describe nine cases of multiple myelomas in the cat, to show that multiple myelomas can co-exist with other conditions, and with polyclonal gammopathy, and to classify multiple myelomas to establish prognostic criteria.
Material and methods
A retrospective medical records search based on the diagnosis of myelomas was performed for nine cats between 1986 and 2002; all nine cats were referred and treated by the author.
Parameters abstracted from each cat's medical record: age, breed, sex, complete blood haematological and biochemical analysis results, feline leukaemia, feline immune deficiency and coronavirus (FIP) antibody titre test results, relative serum viscosity readings, serum protein electrophoresis and agar gel immunoelectrophoresis results, radiological findings, bone marrow aspiration cytologies or biopsy findings, routine urinalysis, urine electrophoresis and urine heat precipitation test results.
The owners were questioned about the medical history of the animal. Each cat underwent a full clinical, orthopaedic and neurological examination.
Haemoglobin and total white cell count were measured on a Sysmex 520 impedance counter (Vet Lab), packed cell volume (PCV) was measured by microhaematocrit, and MCV and MCHC derived arithmetically. Differential white cell counting was performed by microscopy.
All biochemistry analyses were performed by standard methods on a I lab 600 clinical chemistry system (Instrumentation Laboratories, Vet Lab). This included creatinine, and blood urea nitrogen calcium values (including ionised calcium). Serum electrophoresis was performed in all cats using the technique described by Kerr (1991): protein fractions were separated on a polyacrylamide gel at pH 8.4. The dried and stained gel was then scanned by a densitometer to produce the typical electrophoretic trace. This was compared to the normal patterns for the species.
Immunoelectrophoresis to determine the class of paraprotein using the technique by Schultz and Adams (1978) was performed in three cats.
Following electrophoretic separation of serum proteins, troughs were cut into the agarose gel parallel to the separated serum proteins. Antisera specific for each of the major Ig classes were loaded into the troughs and the gel was incubated for 24 h. The antiserum diffused from the trough and formed an arc of precipitation where it encountered specific antigen.
Urine was analysed in all cats using Nephur-6 test chemical strips (Roche-Vet Lab), specific gravity was measured by refractometry. Bacteriology was carried out by standard bacteriological culture methods.
Urine was also evaluated for the presence of light chains of myeloma proteins (Bence Jones) by heat precipitation methods described by Terry et al (1973).
Urine was heated to 40–60°C. If Bence Jones proteins are present a protein precipitation is observed which redissolves on boiling at 100°C and reprecipitates on cooling again from 85 to 60°C. Light chains appear electrophoretically as homogeneous band. Relative serum viscosity was determined by the RBC pipette method as described by Wright and Jenkins (1970).
The red cell pipette was filled by suction of 1 ml of plasma from a standard tube. The time flow between the lines immediately above and below the bulb reservoir was recorded, the flow time of the plasma in seconds was divided by the flow time for water in seconds, this gave the relative viscosity of the plasma carried at room temperature. The normal flow of water is 90 s, 2–3 min for normal plasma.
Thoracic and abdominal radiography was performed in all cats and survey bone radiography was performed in all cats to evaluate the presence of osteolytic bone lesions. Contrast myelography was performed in two cats.
Ante-mortem surgical bone marrow aspirates were collected in eight cats (cases 1, 3, 4, 5, 6, 7, 8 and 9) with a Jamshidi bone marrow biopsy needle. The anatomical sites that were aspirated differed in different cats were: case 1 (L4/L5), case 3 (L6/L7), case 6 (C2/C3/C4), case 8 (left proximal tibia), case 9 (L5 and left and right stifles). The anatomical site of aspiration was based on the suspected sites of multiple myelomas noted on the radiographs (as in the five cases described above) or the most accessible site for bone aspiration as in cases 4, 5 and 7 where the proximal humerus was aspirated.
A post-mortem section of the affected bone was sampled for histopathology in cases 1, 2, 4, 8 and 9. They were fixed in 10% neutral buffered formalin, decalcified in 20% formic acid, embedded in paraffin wax and sections were stained with haematoxylin and eosin.
Initial treatment included intravenous administration of sodium chloride 0.9% or lactated Ringer's solution, to correct dehydration if present and improve renal perfusion. Supportive treatments including antibiotics, diuretics and vasodilators were prescribed in individual cases as follows. Nitroglycerine ointment (1 cm topically every 6 h) to help heart conditions if present. Methylprednisolone sodium succinate (30 mg/kg body weight, IV) initially, followed by two treatments of 15 mg/kg body weight, IV every 6 h. Therapy was used to relieve spinal cord compression if present. Therapy for hypercalcaemia, if present, consisted of rehydration with intravenous saline. Cage immobilisation to induce rest was employed if pathological fractures were present.
Anti-neoplastic therapy
Therapy included melphalan (Alkeran; Wellcome) at a dose of (0.1 mg/kg) 0.5 mg (¼ tablet) melphalan 2 mg, once daily for 14 days, then every other day until clinical improvement or leukopenia developed. This was followed by a continuous maintenance therapy of (0.1 mg/kg) 0.5 mg (¼ tablet) melphalan 2 mg once every 7 days.
Melphalan therapy was combined with prednisolone (Prednicare; Animalcare) given at a dose of 0.5 mg/kg of body weight orally once daily. To cats which presented with leukopenia in addition to anaemia, melphalan was given at a dose of 0.1 mg/kg of body weight orally (¼ tablet) melphalan 2 mg, every 3 days.
If severe leukopenia developed melphalan was discontinued, but prednisolone continued. When WBC counts returned to normal the cats recommenced melphalan at a maintenance dose ¼ of a tablet weekly.
Patients were seen initially at weekly intervals for 4 weeks, and monthly thereafter, when no more improvement could be achieved the frequency of assessment was usually reduced to every 2–3 months.
Owners were questioned at each consultation on the degree and duration of the clinical signs after starting the treatment (eg, anorexia, lameness, weakness, lethargy).
An initial complete blood cell count and serum protein were repeated at weekly intervals for 4 weeks, then monthly for 3 months and 6 monthly thereafter. Liver enzymes were measured in order to evaluate hepatotoxicity before starting the treatment, then at the same time as testing for cell count and serum protein.
A good response to therapy was based on:
Clinical reduction in the presenting signs (ie, lameness, weakness, anorexia, dyspnoea, melena, haematuria and bone pain).
Laboratory evidence of response to therapy (ie, reduction in serum total protein).
Radiographic evidence of response to therapy (ie, reduction or complete absence of lucent lesions).
Cats were classified into those with more aggressive disease and those with less aggressive disease on the basis of the system proposed by Matus et al 1986. These criteria were originally developed for use in the dog.
Prognostic criteria were based on:
Clinical stage of multiple myeloma (ie, PCV value, serum calcium level, extent of radiographic evidence of boneless, light chains of myeloma protein (Bence Jones) in the urine, changes in the serum protein level and renal abnormalities).
Response to therapy (ie, marked improvement, temporary or little improvement and no improvement).
Our criteria for complete remission are transient or absence of clinical signs, insignificant or complete absence of lucency on the radiographs of the effected bone and return of serum protein level to normal when re-measured 8 weeks after treatment and partial when the above mentioned criteria were not achieved.
Results
In the nine cats, clinical signs included lethargy and anorexia (7/9, 78%); polyuria and polydypsia (4/9, 44%); lameness (1/9, 11%); paresis/paralysis and/or pain (4/9, 44%). CNS deficits (dementia with concurrent deficiency in mid brain or brainstem) were documented in three out of nine cats (33%) (Table 1).
Summary of clinical findings radiographic findings and ante mortem needle aspirate findings

M = male, F = female, Mo = months, Yrs = years.
Common metabolic abnormalities included hyperglobulinaemia (globulin>50 g/l) in all cats, renal azotaemia (creatinine>110 μmol/l) in four out of nine (44%) cats, hypercalcaemia (calcium>3.00 mmol/l) in three out of nine (33%) cats. Ionised calcium (>5.7 mg/dl) in three out of nine (33%) cats and hypoalbuminaemia (albumin<25 g/l) in six out of nine (67%) cats (Table 2).
Summary of laboratory findings of multiple myelomas in nine cats

Initial pretreatment leukocyte counts were variable ranging from 2.4 to 19.4 (×10/l) (mean, 8×10/l; range 5–18×10/l). Leukopenia was noted in cases 1, 4, 8 and 9. The leukocyte count was within normal range in cases 3, 5 and 7. Leukocytosis was noted in cases 2 and 6 (Table 2).
Non-regenerative anaemia was found in cases 1, 2, 4, 8 and 9. Routine urinalysis revealed proteinuria (protein>30 mg/dl) in (7/9, 78%) cases 2 and 9 recorded 21 and 19 mg/dl protein in the urine, respectively (reference range, 0). Abnormalities in urinalysis included changes in specific gravity and deposition of urine sediment (see Table 2).
Electrophoresis of blood samples revealed monoclonal gammopathy in seven out of nine cats (Fig 1) and polyclonal gammopathy in two out of nine cats (cases 2 and 3). Electrophoresis of urine samples revealed polyclonal gammopathy in case 4, while blood electrophoresis revealed monoclonal gammopathy.

Case 8, serum protein electrophoresis. Note the narrow monoclonal spike in the gamma region.
Light-chain protein (Bence Jones protein) was found in five out of nine (56%) cats. Serum immunoelectrophoresis to determine the immunoglobulin class was performed in three cats and found to be associated with IgG (cases 7 and 8) and IgA (case 4). Attempts to identify this protein were not made in other cases (Table 2).
Plain radiography of the thorax and abdomen performed in all cats revealed no soft tissue organ abnormalities in eight cats, but in case 2, thoracic radiography showed blurring of the pulmonary vasculature, linear and nodular densities in the lungs and general loss of contrast, typical of interstitial pulmonary pattern and cardiomegally. Abdominal radiograph showed a distended bladder. Routine thoracic and abdominal radiograph were not sufficiently diagnostic of the lytic bone lesions, therefore, close-up bone surveys are essential to detect the bone lesions.
Radiographically, skeletal lesions characterised by well-circumscribed osteolytic bone lesions were seen in the cervical spine, (case 6), the lumbar spine, (cases 1, 2, 3 and 9), the left stifle, (case 8) (Fig 2) and the left and right stifles (case 9) (Fig 3).

Lateral radiograph of the left stifle, showing multiple focal lucencies at the left stifle and oblique fracture through the diseased bone in the proximal tibia.
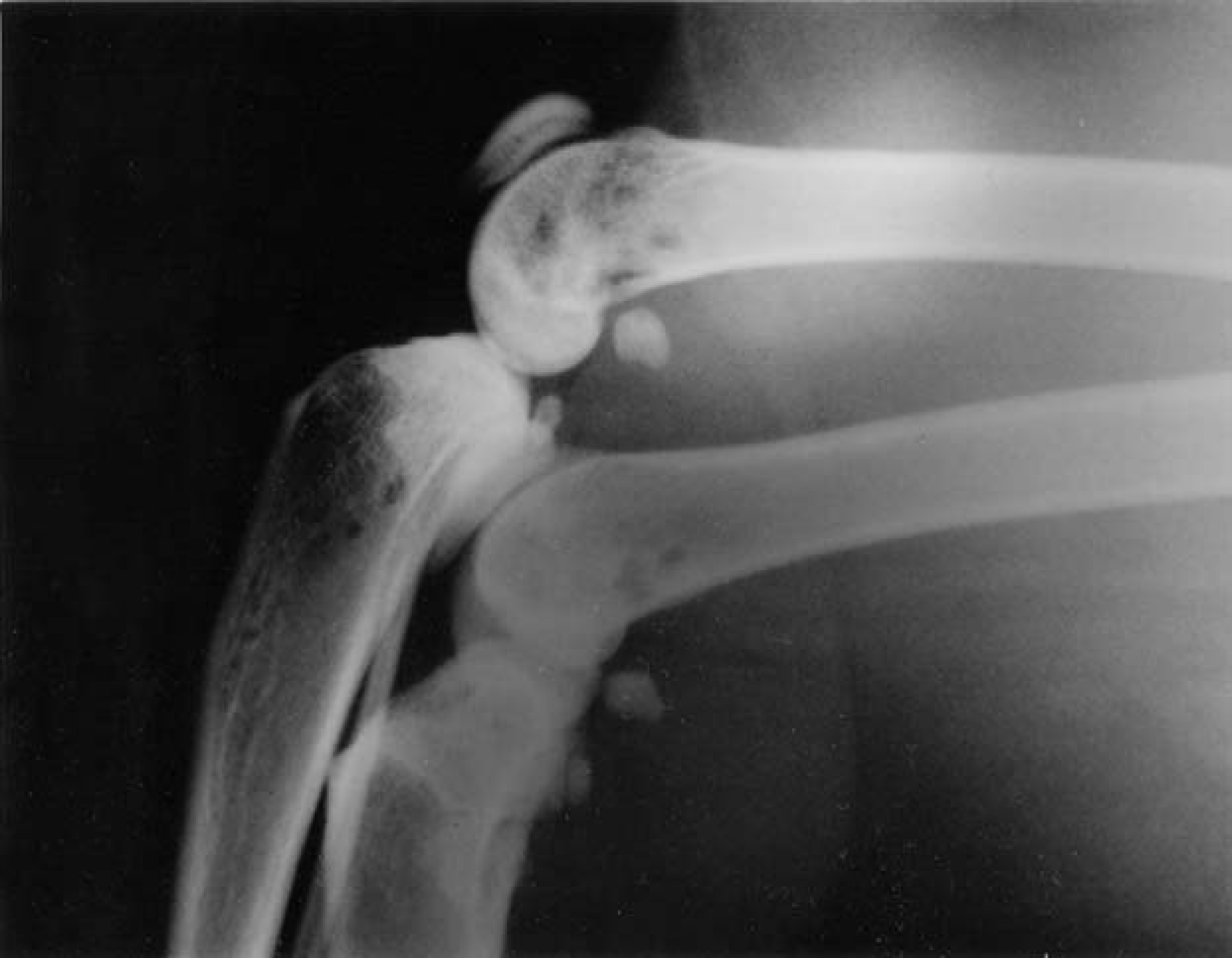
Lateral radiograph of the left and right stifles showing multiple focal lucencies at the left and right stifles.
Contrast myelography was performed in two of the cats and revealed extradural compressive lesion caused by tissue expansion from the bone marrow in cases 6 and 3 (Fig 4).

(A and B) Lateral and ventrodorsal contrast myelogram of the lumbosacral spine showing interruption of dye columns over L6/L7 vertebrae.
All eight cats that had ante-mortem surgical bone biopsies exhibited plasma cell infiltration (>20% plasma cell population in bone marrow aspirate) (Table 1).
Post-mortem microscopic examination of sections of neoplasms exhibited abundant (>20%) plasma cells in the bone marrow. These cells were larger, pleomorphic and contain two or more nuclei (Fig 5).
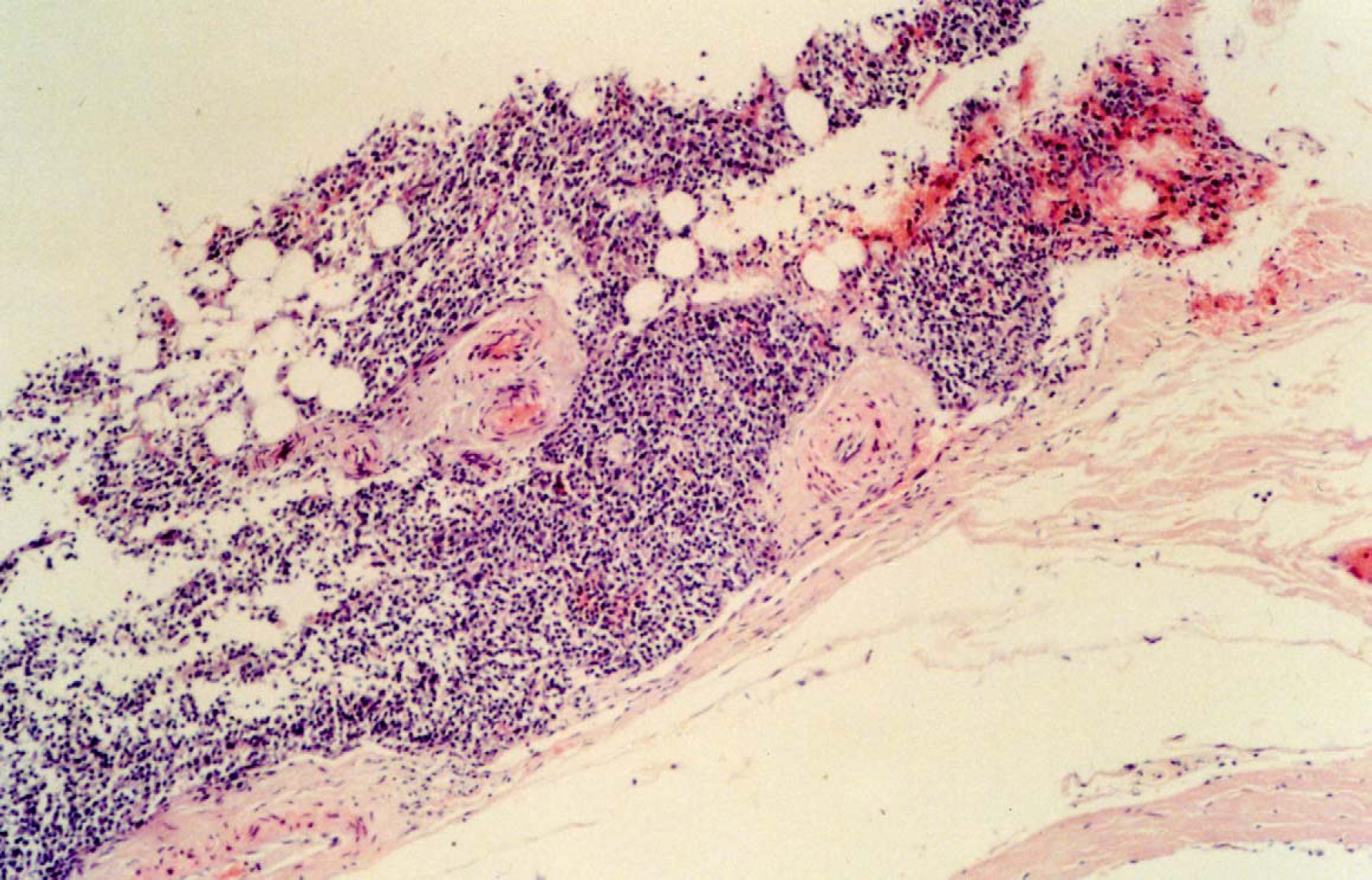
Case 2, histological section of bone marrow showing extradural infiltration of cells in the ventral aspect of the cord, these cells were medium sized oval cells with eccentric oval nuclei having the appearance of plasma cell neoplasia.
In case 2, post-mortem histopathology of the bone biopsy exhibited abundant (>40%) plasma cells in the bone marrow, with many features of malignancy (multinucleated cells and immature nuclear features). No neoplasia was found within organs of the abdomen and/or the thorax, instead granulomatous lesions, which appeared as white foci on cut surfaces, were found in the liver, kidney, abdominal lymph nodes and spleen (Table 3). The cat also had pulmonary haemorrhage/oedema, myocardial necrosis and blood filled bladder.
Summary of response to treatment, follow-up and necroscopy findings in nine cats with multiple myeloma

Bleeding or haemorrhagic disease was observed in four cats in this study. In case 1 retinal vessels were dilated and tortuous, and intraocular haemorrhage was detected on ophthalmoscopic examination. In case 2, on clinical examination there was melena and haematuria, and on post-mortem examination there was approximately 25 ml of serosanguinous fluid, mixed with clots of blood in the thoracic cavity, pulmonary haemorrhage and bladder haemorrhage. In case 4, focal areas of perivascular haemorrhage were detected in the retinas of both eyes on ophthalmoscopic examination. In case 8, melena was detected on clinical examination.
Relative serum viscosity which was determined by the RBC pipette method was in case 1, 4.51; case 2, 3.65; case 4, 5.46; and case 8, 6.18. Relative serum viscosity in clinically normal cats is reported to be <1.40.
An estimate of overall improvement was made based on all the data collected from the owners, related to clinical improvement or deterioration (eg, degree and duration of morning stiffness and stiffness related to activity, increased appetite, resolution of melena and haematuria) and our own assessment related to disease severity (eg, the degree of clinical signs (neurological, cardiac or ocular abnormalities), the degree of lucency seen on the radiographs, haematological parameters (serum protein level, liver enzymes measurement) and functional activity, eg, ability of the animal to move).
Little or no improvement was noted in cases 1, 2, 8 and 9. Temporary improvement was noted in case 4 and marked improvement in cases 3, 5, 6, and 7. Cases 3, 5, 6 and 7 were judged to be in complete remission.
In those cats with complete remission, transient or absence of the clinical signs were seen in cases 3, 5, 6 and 7, insignificant or complete absence of lucency on radiographs of the affected bone in cases 3 and 6, and return of serum protein level to normal in cases 3, 5, 6 and 7 when serum proteins were re-measured 8 weeks after treatment.
At the onset of therapy ALT, ALP and bile acids were found to be within normal limits. A mild elevation of ALT (average 90 IU/l) and ALP (average 115 IU/l) occurred in five cats (cases 3, 4, 5, 6 and 7) 4 weeks after treatment, but returned to normal when liver enzymes were re-measured 4 weeks later (cases 3, 4, 6 and 7) and 8 weeks later (case 5). During that time cats were fed a balanced diet with moderate to high levels of high biological value protein and vitamin supplementation.
Lethargy after starting treatment increased in all cats, but returned to normal 4 weeks later.
All animals presented in this study with the exception of case 2 have definite classic ante-mortem multiple myelomas by following the criteria described originally by MacEwen and Hurvitz (1977) in dogs. Case 2 also satisfied these criteria but only after a post-mortem examination was performed.
Haematological and radiological signs that were repeatedly noted in cases which had less favourable outcome were listed in the aggressive group, and other haematological and radiological signs which had favourable outcomes were listed in the less aggressive group (Table 4). The survival time of the four cats in the aggressive group (cases 1, 2, 8 and 9) was 2, 5, 14 and 7 days, with a median survival time of 5 days. The survival time of the five cats in the less aggressive group (cases 3, 4, 5, 6 and 7) was 450, 120, 135, 510 and 720 days, with a median survival time of 387 days. There was significant difference between survival time and mean survival time in the aggressive and less aggressive groups. This may be helpful to assess the tumour burden and outcome of treatment in the future.
Classification of multiple myelomas

Discussion
Cats in this study manifested clinical signs associated with the proliferation of plasma cells in the bone marrow and other soft tissues; these findings agree with other investigators' findings (Osborne et al 1968, Martinson et al 1973, Matus et al 1983), and the production of immunoglobulin (Ig) A or IgG. Immunoglobulins have been identified as IgA, IgD, IgM, or IgE by Binta et al (1995) and IgA or IgG by Bienzle et al (2000).
Myeloma light-chain protein (Bence Jones proteins) in this study developed in five cats (56%) and was associated with the development of renal insufficiency in four cats (44%). The relationship between the development of renal disease and the presence of Bence Jones proteins may be related to the fact that Bence Jones proteins are nephrotoxic (Hurvitz et al 1971) or the production of light-chain protein metabolic degradation by renal tubular cells causing direct tubular damage and kidney dysfunction (Levi et al 1968, Fer et al 1981). Cases 1, 8 and 9 had increased total calcium and ionised calcium concentrations. Measurement of ionised concentration is essential as increased serum protein concentration may result in ‘falsely’ increased total serum calcium concentrations as more calcium is bound to the proteins in the blood. Two of these cats (cases 1 and 8) had evidence of renal disease. Possible causes of renal insufficiency that occurs with multiple myelomas are first, hypercalcaemia can cause renal mineralisation (Kruger and Osborne 1994); second, myeloma cells can infiltrate the kidney (Osserman et al 1987); third, paraproteins can infiltrate the kidney (Osserman et al 1987, Alexanian and Dimopouls 1994).
Hypercalcaemia may develop as a result of direct osteolysis by the infiltrative plasma cells (Sheofor et al 1996) or the production of parathyroid hormone (PTH) like substances, parathyroid hormone related peptide (PTHrP) (Rosol and Capen 1992). In cases 1, 8 and 9, increased total and ionised calcium concentration may be consistent with PTH-like effect from the tumour. Explanation of how hypercalcaemia develops in these cats would require quantification of PTH related protein.
Renal failure in multiple myelomas is rarely reversible and a poor prognostic sign. Management is directed primarily towards control of the myelomas with anti-neoplastic therapy (Fer et al 1981).
Immunophenotyping was performed in three cats in this study. Of these three cats only one cat with IgG is still in remission 24 months after treatment. This may highlight the potential prognostic value of immunophenotyping and support the findings of Bienzle et al (2000). The prognosis of cats with IgG myeloma may be better than that of cats with IgA myeloma.
Survey radiographs showed osteolytic bone lesions in six cats and myeloma light-chain protein was found in five cats. The frequent findings of bony lesions and Bence Jones proteins in this study have not been observed by other investigators, this may be because other studies have not performed the required investigations or because the diagnosis requires high quality radiographs.
Haemorrhagic disease and ocular disease
Bleeding or haemorrhagic disease was observed in four cats in this study (cases 1, 2, 4 and 8).
Although various mechanisms that might cause prolonged bleeding in patients with multiple myelomas have been proposed, the exact mechanism is unknown. In our study cases 1, 2, 4 and 8 all had adequate platelet number. Bleeding could have resulted from decreased platelet adhesiveness after being coated by the myeloma proteins, overloading of the blood vessels with thickened blood causing it to rupture or precipitation of plasma cells in the blood vessel wall causing its weakening and rupture (Hendrix et al 1998).
Although serum HVS is diagnosed infrequently in cats, it should be considered in cats with monoclonal gammopathy, especially if the cat has CNS neurological deficits, cardiac and ocular abnormalities (eg, perivascular haemorrhage, vessels tortuosity, retinal detachment or intraocular haemorrhage).
Although plasmapheresis has not been used in this study, its use may have been beneficial in reducing plasma protein concentration and serum viscosity and render neoplastic cells more susceptible to chemotherapy.
Multiple myeloma associated with polyclonal gammopathy
Not all cases of multiple myelomas revealed monoclonal gammopathy on serum electrophoresis. In case 2, abnormalities in the blood analysis included elevated serum total proteins, globulin and urea. The cat had hypochromic non-regenerative anaemia (PCV 00.19 or 19%, Hb, 74 g/100 ml, RBC 4.63/0 l). The cat was leukothrophic (total WBC 19.4/0 l) with neutrophilia (neutrophils 16.8/0 l).
The cat tested negative for feline leukaemia and feline immune deficiency but coronavirus (FIP) antibody titre was 1/1280. The cat had a polyclonal gammopathy with elevated gamma globulin that is consistent with chronic inflammation.
Urine protein electrophoresis and Bence Jones protein test could not be run as an inadequate amount of protein was present in the urine (21 mg/dl), the laboratory needed at least 25 mg/dl.
Although osteolytic lesions in the lumbar spine and pathological fracture at the fourth lumbar (L4) vertebra were seen on the radiographs, no permission was given for needle aspiration of the osteolytic lesions. Blood samples were suggestive of feline infectious peritonitis and the only evidence that multiple myeloma might be present was the presence of osteolytic bone lesions on radiographs of the lumbar spine and relative serum viscosity was 3.65 which may suggest hyperviscosity syndrome.
In this cat final confirmation of the diagnosis was only made on post-mortem histopathology of the bone biopsy where abundant (>40%) plasma cells were present in the bone marrow, most with features of malignancy (ie, multinucleated cells with eccentric nuclei and immature nuclear features), compatible with the diagnosis of multiple myelomas. No neoplasia was found within organs of the abdomen and/or the thorax. Instead granulomatous lesions, which appeared as white foci on cut surfaces were found in the liver, kidneys, abdominal lymph nodes and spleen, confirming feline infectious peritonitis. The cat also had pulmonary haemorrhage, oedema, myocardial necrosis and blood filled bladder indicating that this cat is likely to have had hyperviscosity syndrome secondary to its elevated globulins, which probably led to cardiac hypoxia, pulmonary haemorrhage, oedema and bladder haemorrhage. The cat did not have classic monoclonal gammopathy; rather, it had a polyclonal gammopathy. A possible explanation for the absence of monoclonal gammopathy is the presence of feline infectious peritonitis. The virus could have been present subclinically before the development of the plasma cell neoplasia, and feline infectious peritonitis became clinically apparent when normal immunoglobulin levels became severely depressed.
Decreased immunoglobulin synthesis may be caused by proliferating B-cells that release an inhibitory factor which may block proliferation of normal B-cells. This cat was initially treated for spinal cord compression because of the history, clinical signs, results of the neurological examination and results of the radiological examination. However, the intravenous steroids were not sufficient to reduce the neurological signs. Effort to alleviate respiratory distress using frusemide (Lasilaction: Lasix 2 mg/kg IV every 4–6 h) while maintaining hydration status and electrolyte balance with lactated Ringer solution was unsuccessful.
The cat was given anabolic steroid Nandrolone (Laurabolin; Intervet at a dose of 5 mg/kg SC every 21 days) to stimulate appetite and prophylactic amoxicillin clavulanic acid (Synulux; Pfizer 20 mg/kg body weight, PO every 12 h) but the haematuria did not resolve within the 5 days. This may be because the cause of haematuria was hyperviscosity syndrome rather than infection. In our experience one of the main differential diagnoses of multiple myelomas in cats is feline infectious peritonitis. Both may be presented with a similar clinical and haematological picture. The main differences were the identification of high levels of coronavirus titre; absence of Bence Jones protein in the urine and electrophoresis consistently produced polyclonal gammopathy in both blood and urine in feline infectious peritonitis.
To the author's knowledge this is the first reported case where multiple myeloma existed at the same time as feline infectious peritonitis, and illustrates the need for caution when interpreting the results of serological tests for FIP and multiple myelomas and the danger of relying on serology alone for making a diagnosis.
Polyclonal gammopathy, which is rare in multiple myeloma, was also found in case 3 in the present study. Possible explanations for the absence of monoclonal gammopathy in case 3 are the presence of a secondary infection the tumour was non-secretory, the paraprotein had broken before electrophoresis was performed, or multiple plasma cell types involved in the neoplasm that secreted different paraproteins (Kato et al 1995).
This finding shows that other pathological changes can be present at the same time as multiple myeloma and great care should be taken when reading the radiographs and interpreting haematological and urinalysis; the presence of feline infectious peritonitis in case 2 had a great effect on treatment and prognosis. This can confirm that multiple myelomas can be associated with polyclonal gammopathy.
In case 4 monoclonal gammopathy was present in the blood, but polyclonal gammopathy was present in the urine. This may confirm that monoclonal gammopathy can be present in the blood and polyclonal gammopathy in the urine or vice versa in the same cat.
In this study neutropenia developed after starting the treatment in cases 3, 4, 5, 6 and 7 at 3, 2, 3, 4 and 1 week after treatment, respectively. In these cats melphalan treatment was discontinued but prednisolone treatment continued. WBC counts were repeated weekly until returned to normal. Once WBC counts returned to normal, melphalan was recommenced (2 mg) at a maintenance dose of X of a tablet weekly.
Initial melphalan therapy can cause profound neutropenia, this may be related to:
Overdosing, because the pill size (2 mg) is inconvenient to divide in cats.
Individual sensitivity in different cats.
Profound neutropenia was present initially.
Infections are a common cause of death in animals with multiple myeloma and must be treated promptly. Antibiotic therapy is highly recommended, even if there is no obvious sign of infection, especially during the diagnostic work up (MacEwen and Hurvitz 1977).
One reason for an increased infection rate may be that normal immunoglobulin levels are usually severely depressed. Proposed mechanisms that might cause infection are: decreased immunoglobulin synthesis seems to be caused by proliferating B-cells which release an inhibitory factor which may block the proliferation of normal B-cells and another reason is that severe bone marrow infiltration and replacement by neoplastic cells results in granulocytopenia (MacEwen and Hurvitz 1977).
Although radio responsiveness of multiple myeloma has not been evaluated in the nine cats in this study, one cat had an exceptional response to a combination of surgery, radiation and alkylating therapy and survived nearly 2 years (Bienzle et al 2000). Therefore, radiotherapy may have been beneficial in the treatment of the nine cats in the study. Radiotherapy can be used in the treatment of focal myeloma lesions (Bienzle et al 2000).
Genetic predisposition may have been a possible aetiological factor in cases 8 and 9 as they were siblings. Genetic predisposition was an important factor in the development of plasmacytomas in mice as these neoplasms occur only in a few mice strains (Dunn 1957, Potter and Robertson 1960). Further studies are requested to support this hypothesis.
Relapse of the clinical signs and therefore the tumour in case 4, could be due to the presence of multiple cell types in the tumour and the response of one type but not the other or due to the development of resistance of the neoplasm to the chemotherapy drugs, or a combination of the two (Weber and Tebeau 1998).
Finally, although multiple myelomas may progress to fatal termination it has been classified into aggressive and less aggressive to help decide the prognosis. Characteristic clinical, radiological and laboratory findings were associated with the four cats in the aggressive group; they may be considered a poor prognostic indicator because they were associated by short survival and mean survival time.
Conclusion
Multiple myeloma should be suspected when the total protein and globulins are shown to be elevated, early diagnosis and treatment is paramount for a rapid early response, all coming together in a strategy of total therapy for the disease.
What has become apparent is that patients can now be put into complete remission, a state where using ordinary laboratory methods and radiography, the disease can be detected and the patient treated early. It is in this state that the patients are able to come back to a normal quality of life.
Be vigilant for any additional conditions that may co-exist with multiple myeloma because this will have a direct effect on treatment and prognosis, and in multiple myelomas can be associated with polyclonal gammopathy.
Footnotes
Acknowledgements
The author is grateful to Mr Hamish Denny for reading the manuscript and to those veterinary surgeons who referred cases, which have been included in this study.
