Abstract
The BD ProbeTec CT Qx Amplified DNA Assay and the BD ProbeTec GC Qx Amplified DNA Assay (both BD Diagnostics, Sparks, MD) represent two new assays developed for use with the BD Viper System with XTR Technology (BD Diagnostics). These assays were built on the foundation of the former BD ProbeTec ET assays (BD Diagnostics) and its accompanying instrumentation. The study described below compared the new assay format, Extracted Mode, to the former assay format, Nonextracted Mode, with the primary objective of examining and measuring overall time expenditures and efficiency of operation. An 80— 142-min reduction in “hands-on” total processing time was observed for the Extracted Mode whether testing urines or swabs. The second objective was to assess the accuracy of performance at low simulated analytical loads for the pathogens Chlamydia trachomatis and Neisseria gonorrhoeae. Performance accuracies for simulated positive and negative urine or swab specimens were calculated to be 99.97% for Extracted Mode and 99.76% for Nonextracted Mode.
Introduction
Chlamydia trachomatis (CT) and Neisseria gonorrhoeae (GC), the two most common urogenital disease pathogens, were reported with increasing frequency in the decade between 1999 and 2008. 1 During that time, there was a gradual acceptance throughout the clinical laboratory industry for the implementation of molecular methods and automation that could detect these sexually transmitted infections (STIs) in clinical practice settings. It is apparent that molecular diagnostic kits offer concomitant increases in clinical sensitivity and specificity compared with traditional culture methods, along with decreased turnaround time.2,3 It has generally been discussed and accepted that the heightened detection and reporting of these STIs has been attributed to the use of molecular methodologies. 4 Increased reporting of positive results has led to increased treatment of both symptomatic and asymptomatic patients. Toward the latter half of the decade, in many locales, there was a steady-rate of gonorrhea detected and a continual increase in the overall annual detection of chlamydia. Thus, it has been generally recognized that the commercial molecular diagnostic methods for STIs serve a useful purpose for the benefit of laboratorians, physicians, their patients, and the public health.
The BD Viper System with XTR Technology (Viper XTR; BD Diagnostics, Sparks, MD) represents a technological improvement of the preexisting BD ProbeTec ET (BD Diagnostics) method.
5
Viper XTR is capable of performing testing functions in two modes:
Nonextracted Mode, which automates amplification setup and real-time detection associated with the BD ProbeTec ET. Extracted mode, which uses ferric-oxide (FOX) extraction of nucleic acid and real-time strand displacement amplification (SDA)/detection of CT and GC DNA.
FOX extraction uses paramagnetic particles to isolate nucleic acid (nonspecifically) from potential inhibitors of real-time detection. Cell lysis is achieved via chemical means, followed by an extraction process that composes of binding of nucleic acid to the paramagnetic particles at low pH, washing to remove unbound material and elution at high pH. Eluate is then neutralized to buffer conditions optimal for real-time amplification, achieved by SDA. 5
There were two objectives in this study design: to measure the time-related efficiencies of the Viper XTR in Extracted Mode to that in Nonextracted Mode in terms of overall “hands-on” time and total processing time (TPT), and to correlate the accuracy of performance of Viper XTR in Extracted Mode to Nonextracted Mode at spike levels of CT and GC organisms indicative of low analytical loads of these pathogens.
Materials and Methods
Two sites were chosen for the implementation of this study: the John Stroger Hospital of Cook County in Chicago, IL and the Mecklenburg County Public Health Department, Charlotte, NC. Both facilities and their personnel were BD Viper System users having broad familiarity with the operation of the Viper in the Nonextracted Mode.
A total of 3821 simulated urine and swab samples were prepared at the BD Research and Development laboratories in Sparks, MD. Specimens were divided into two groups and distributed as unknowns to both institutions. A mixture of positive and negative urine and swab samples were provided such that the test specimen batches were generally representative of the prevalence rates of CT and GC known to both institutions. All samples were processed and tested according to the manufacturer's recommendations. The procedural steps differ between the Nonextracted Mode and Extracted Mode. The Nonextracted Mode procedure consisted of 25 operational steps (of which 11 were of pre-set duration) and 14 time-measurable steps. The Extracted Mode procedure consisted of 14 operational steps (of which four were of pre-set duration) and 10 time-measurable steps. Eliminated from the Nonextracted Mode procedure were six pre-set duration operational steps including cooling of prewarmed specimens, centrifugation, vortexing, and heat-lysing, along with a manual operation related to decanting supernatant after centrifugation and pellet suspension in diluent. Two technologists at each site were involved in testing and timing duties. A minimum of five complete test-timing runs were performed and analyzed at each institution.
Urine specimens were prepared and divided into neat urine and urine transported in urine preservative tubes (UPT urine). Swabs were separated into male and female swab specimen types. All specimen tubes were uniquely identified with an individual coded accession number but without indication as to being positive or negative. All four specimen types were tested using the Nonextracted and Extracted Modes on the Viper XTR. The 3821 specimens were prepared using Qx Swab Diluent, CT/GC Sample Diluent (all BD Diagnostics), and synthetic urine. Synthetic urine is a proprietary combination of sodium chloride, urea, potassium chloride, potassium phosphate, proclin, and water. There were no known inhibitors added to the microorganism-diluent suspension to test the efficiency of the extraction. The simulated specimens were prepared as follows: negatives (unspiked media), positive for CT only, positive for GC only, and co-spiked CT/GC. Positives samples consisted of a known concentration of CT serovar H and/or GC strain ATCC 19424. Initial CT and GC stock concentrations for urine specimens were prepared and measured to contain approximately 275 elementary bodies/mL (EBs/mL) for CT and 446 colony forming units/mL (CFU/mL) for GC. Initial CT and GC stock concentrations for swab specimens were prepared and measured to contain approximately 413 EBs/mL for CT and 669 CFU/mL for GC. It is estimated that approximately 10–20% of the initial sample is delivered to the amplification reaction in both Extracted Mode and Nonex-tracted Mode. CT and GC concentrations differed as the simulated urine and swab specimens were prepared from different initial stock solutions. At each institution, microtiter plates were filled entirely (96 wells) with 46 specimens tested for CT and GC plus positive and negative CT and GC quality controls.
Technologists at both sites were provided with complete training on the Viper XTR in the Extracted Mode. Technologists at one of the facilities were also trained to process urine specimens using the Nonextracted Mode, because at this institution swab specimens were the only standard accepted specimens of choice for testing on the BD Viper System. This institution did not routinely test urine specimens for STIs because of the additional hands-on time needed in preanalytical processing.
Positive specimens were randomly interspersed among the negative specimens during the preliminary setup and all were tested as unknowns. At the completion of all testing, the results were compiled, evaluated, and collated for presentation of the data findings. A total of 7642 results were obtained from the 3821 specimens because of the fact that both CT and GC were tested per specimen. Of the 3821 specimens, 2012 specimens were processed by Institution A and 1809 specimens were processed by Institution B.
Results
Table 1 shows the distribution of specimens tendered for testing via Nonextracted and Extracted Modes. There was no distinction made for gender in simulated urine specimens. Because of the differences in swab sizes used for male urethral and female endocervical specimens, a delineation between the specimens was made as indicated in the table. Overall, there were 1960 simulated urine specimens and 1861 simulated swab specimens.
Distribution of specimens
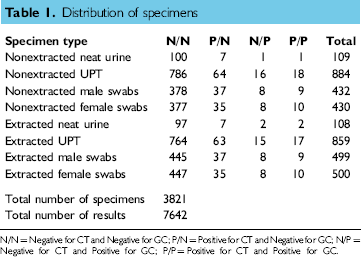
N/N = Negative for CT and Negative for GC; P/N = Positive for CT and Negative for GC; N/P = Negative for CT and Positive for GC; P/P = Positive for CT and Positive for GC.
Table 2 shows the distribution of the results for the tested specimens generated by the two institutions and correlation of the results achieved to the known identities of the specimens. The overall percentage agreement between the reported results and the actual results for the combined CT/GC results was 99.92%, as there were only 6 specimens that disagreed from the expected result outcome (was 7636 of 7642). Five of the six discrepant specimens were observed in Nonextracted Mode (performance accuracy = 99.76%, 3705 of 3710) and the remaining single discrepant result was obtained in Extracted Mode (performance accuracy =99.97%, 3931 of 3932). CT and GC test levels were selected to reflect low organism loads.6–8
Distribution of results over two institutions
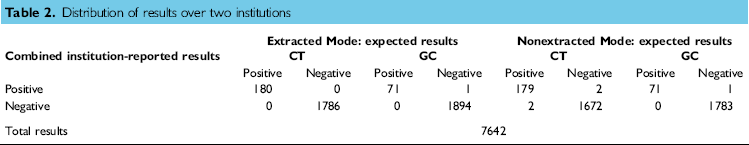
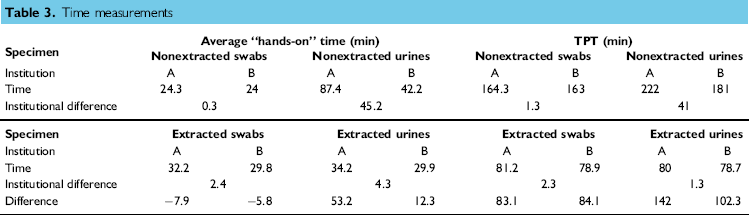
The average “hands-on” time (min) and TPT to achieve completed runs in Nonextracted and Extracted Modes, shown in Table 3, was determined by subtracting the measured time unit for Extracted Mode from NonExtracted Mode. Institutional differences were also calculated. Although there was a slight increase in “hands-on” time at both institutions of between 5.8 and 7.9 min observed for swabs processed in Extracted Mode compared with “hands-on” time in Nonextracted Mode, the TPT decreased at both institutions by between 83.1 and 84.1 min. The time savings for urine specimens in the Extracted Mode was even greater. “Hands-on” time at both institutions for urines in Extracted Mode decreased by between 12.3 and 53.2 min and the TPT decreased between 102.3 and 142 min. The majority of the time spent in “hands-on” processing devoted to Nonextracted Mode was decreased through elimination of manual steps involving centrifugation, decantation, rehydration, and vortexing of specimens.
Time measurements
Discussion
The results suggest that the advanced automation and processing provided by the Viper XTR in Extracted Mode allows for significant “hands-on” time savings for urine samples compared with the Nonextracted Mode. Additionally, the TPT was decreased significantly using the Viper XTR in Extracted Mode for both urine and swab samples.
The personnel involved in these studies performed very well with the Extracted and Nonextracted Modes of the Viper XTR. However, the most dramatic “TPT” savings was observed when testing urine specimens in Extracted Mode (142 and 102.3 min at institution A and B, respectively). Personnel from Institution A were experienced with handling and testing swab specimens collected from male or female subjects. As a result, urine specimens were new to the personnel at this institution and there was a significant learning curve with regard to correct processing of these specimens. Initially, it took 45.2 min longer on average, for personnel from Institution A to perform the same “hands-on” steps as personnel from Institution B for processing urines in Nonextracted Mode. However, it was equally extraordinary that the time difference in “hands-on time” was greatly reduced when both institutions tested urines in the Extracted Mode as only 4.3 min, on average, separated their respective performances. Most of the time spent in “hands-on” processing devoted to Nonextracted Mode was eliminated through automation of those steps in Extracted Mode operation. Overall, an examination of the data accumulated over the course of the testing events showed that the total amount of “hands-on” time necessary for the performance of Nonextracted Mode decreased rapidly over five test runs, indicating an increasing level of familiarity, confidence, and assurance and that the “learning curve” was of short duration and rapidly implemented as a routine. With the exception of urines tested in Nonextracted Mode, the amount of “hands-on” time ranged from a low of 24 min at Institution B for swabs processed using the Nonextracted Mode to a high of 34.2 min at Institution A or urines processed using the Extracted Mode. This higher time measurement is consistent with the measurement determined by Felder et al. 5 “Hands-on” timing measurements in the Extracted Mode were relatively consistent between both institutions and ranged between 29.8 and 34.2 min.
There was a 5.8—7.9-min increase in the total amount of “hands-on” time necessary for processing swabs in the Extracted Mode compared with Nonextracted Mode at both testing facilities. This can be attributed to the increased amount of time necessary to recap the pierced specimen tubes before saving or discarding.
Overall, there were dramatic decreases in the TPT savings observed for the Extracted Mode compared with the Nonextracted Mode on the Viper XTR at both institutions. The average TPT for swabs in Nonextracted Mode at both institutions (Institutions A and B listed in order, in all cases) was 164.3 and 163 min. The average TPT for swabs in Extracted Mode at both institutions was 81.2 and 78.9 min. This represents a time savings of between 83.1 and 84.1 min through the use of the Extracted Mode. Similarly, the average TPT for urines in Nonextracted Mode was 222 and 181 min. The average TPT for urines in Extracted Mode was 80 and 78.7 min. This represents an overall time savings of between 102.3 and 142 min through the use of the Extracted Mode.
Finally, the fact that overall TPT measured for Extracted Mode at both institutions was essentially consistent for swab and urine specimens (range from 78.7 to 81.2 min) is additional evidence that manual, time-consuming, operations are eliminated with the Viper XTR.
There was broad agreement achieved by both institutions when performance results were compared across the 3821 specimens and two assays resulting in 7642 results. Of the six discrepant results, five were observed in specimens tested in Nonextracted Mode and one was in Extracted Mode. One of the advantages of the Viper XTR in Extracted Mode is that urine specimens do not require the additional “hands-on” time for the processes that have now been automated. It is clearly evident that both the Nonextracted and Extracted Modes on the BD Viper System provide comparable and accurate detection of CT and GC while operating near the stated limits of detection.6–8 Microorganism concentration were prepared to mimic an initially low sampling presence of the pathogens at <103 CFU/mL. This analytical investigation simulated what could occur within a clinical investigation. Detection of these infections is possible in patients with low levels of microorganisms, such as patients with residual molecular traces of microorganisms following therapeutic treatments, patients from whom a collected specimen may be of poor quality, and patients who are asymptomatic for disease presence. Undoubtedly, both Non-extracted and Extracted Modes of testing allow for exquisite testing detection, but Extracted Mode enhances that capability and does it in a shorter TPT.
Conclusion
Viper XTR in Nonextracted and Extracted Modes provide accurate results for the detection of CT and GC at low analytical loads of these pathogens. There was a 99.84% overall percent agreement achieved, while the Extracted Mode showed the greater accuracy in this investigation. The enhanced automation of the Viper XTR significantly reduces hands-on time associated with testing urine specimens and will allow institutions with limited resources and manpower to add these assays and methodology to their testing menu for STIs.
Footnotes
Competing Interests Statement: The authors disclose that Elliot Rank and Michelle Althouse are employees of B.D. Diagnostic Systems.
Acknowledgment
This study was supported by BD Diagnostic Systems, Sparks, MD.
