Abstract
The use of molecular techniques to inform diagnosis, prognosis, and treatment design will play an important role in the future of medicine. Each new test, however, represents a new cost to the health care system, and significant effort is required to move new techniques to the clinical setting in the most cost-effective and efficient manner. Consequently, there is a compelling need for technological improvements that will facilitate clinical application of novel assays, by reducing cost and complexity in implementation. This is particularly important in cancer pathology. Here, we present a novel applicator technology that enables staining of individual biopsies in a tissue microarray (TMA) to provide low-cost, multiplexed biomarker testing at the level of intact tissue. The applicator is designed to deliver tens of nanoliters of aqueous reagent to arrayed tissue biopsies kept under a layer of oil-based Liquid Coverslip without contacting the biopsies. A pin consisting of concentric stainless steel electrodes separated by a hydrophobic insulator provides a balance between hydrophobicity and hydrophilicity to hold a reagent droplet on the tip of the pin, whereas a small electrical current passed through the droplet spanning the electrodes is used for drop sensing. This design is more amenable to repeatable manufacturing than a previous prototype, which in initial testing demonstrated successful immunohistochemical and in situ hybridization staining of individual biopsies in a TMA, but was difficult to produce. This new design was tested to investigate the factors affecting its operation, in terms of the volume of reagent picked up and its ability to successfully deliver reagent to the biopsies.
Introduction
The rise of molecular medicine has yet to fulfill initial expectations, leaving the brunt of diagnostic evaluation in the hands of conventional morphological analysis of intact tissues, with immuno-histochemical and in situ hybridization (ISH) assessment of biomarkers as useful ancillaries. 1 , 2 This dependency on histological analysis is likely to remain in place for at least the foreseeable future, even as more and more molecular determinants and indicators of disease are revealed. To ensure that these discoveries translate to improved patient care, a number of difficult challenges must be addressed, the most obvious of which is cost. A method is needed for multiplexed screening of biomarkers at the level of intact tissues in a low-cost, high-throughput microarray format. This will provide the ability to assess tissues for multiple biomarkers, while preserving the histological information necessary for conventional diagnosis.
Tissue microarrays (TMAs), in which up to 1000 tissue biopsies of 0.6-mm diameter are arrayed onto a single slide, provide the perfect platform (Fig. 1). 3 Originally designed to test for biomarkers in large banks of characterized tumor tissue, TMAs provided enormous savings in both biomarker reagent and tissue usage. 3 Hundreds of biopsies can be assayed with the same volume of reagent required for a single conventional biopsy. Representation of tissue heterogeneity, once a major concern with TMAs, is satisfied with triplicate or quadruplicate samples taken from each tumor. 3 5 The advantages of this platform are attractive to clinical diagnostics. However, under current staining methods, in which a single reagent is applied to the entire slide, the clinical utility of the TMA platform is limited. To achieve the potential per-biopsy cost savings offered by TMAs, a pathologist would have to wait for a full array of biopsies requiring the same test, an unlikely prospect even in the busiest hospitals. For an array of 500 biopsies, this could easily correspond to more than 100 patients.

A tissue microarray with 0.6-mm diameter core biopsies.
Background
To address this problem, we have developed an instrument (the Tissue Microarray Antibody Spotter or TMAS) (Fig. 2) capable of delivering reagent to individual biopsies within a TMA. 6 This offers two principal advantages. By delivering reagents to individual biopsies, it is possible to perform multiplexed tests on a single slide, and a TMA formed from cores of a patient's tumor biopsy, or a cell suspension array of fine needle aspirations, could be tested for a variety of biomarkers to obtain a complete cancer profile of the patient, all on one slide. This moves analysis from the conventional “one slide, one test” to a new “one slide, one patient, multiple tests” regime. Furthermore, when reagent is applied in nanoliter volumes directly to the biopsies, none is wasted to cover the empty glass substrate in between, and cost savings are realized beyond that already offered by the use of TMAs. A typical autostainer that applies 400 μL of primary antibody to a TMA slide with 400 biopsies gives an effective reagent usage of 1 μL per biopsy. 7 Delivering tens of nanoliters to each biopsy offers an order of magnitude of reagent savings. Because reagent use scales directly with the number of biopsies being assayed, there is no need to wait for a full TMA to achieve maximum cost savings of reagent, thus maximizing flexibility.
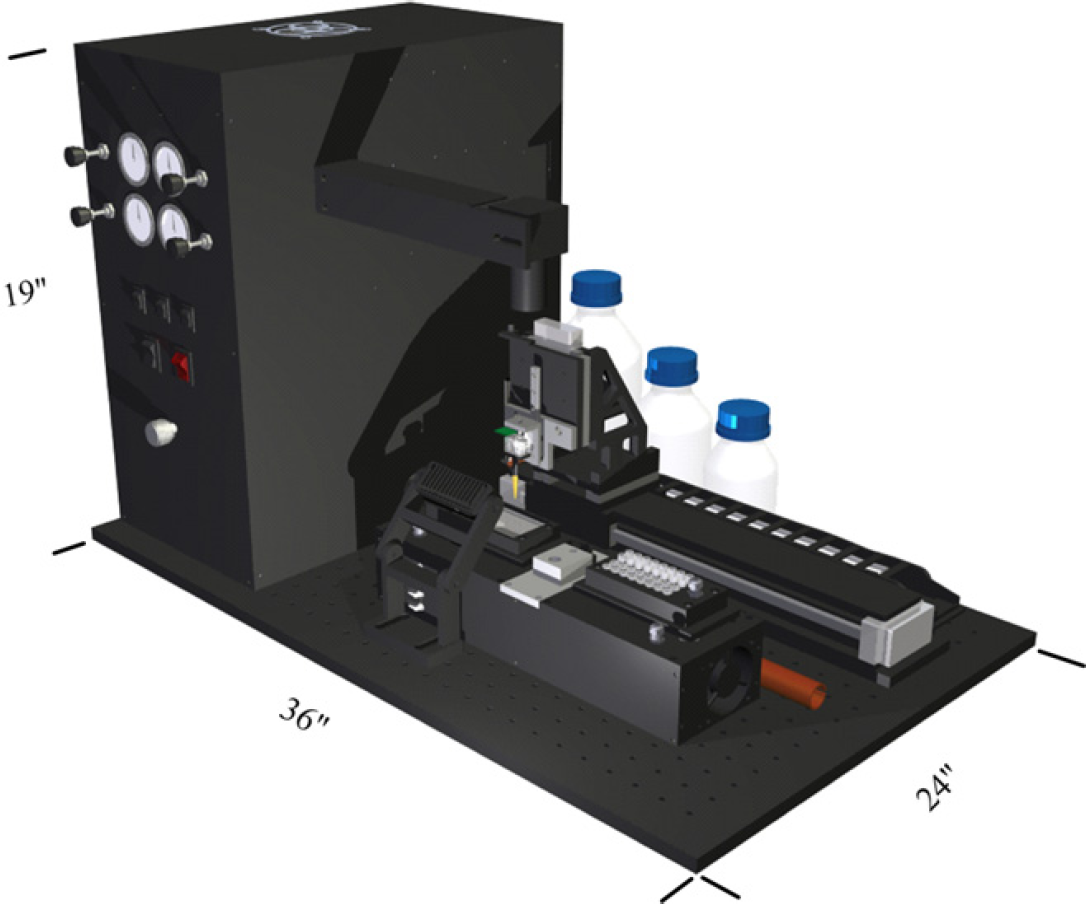
The Tissue Microarray Antibody Spotter instrument.
Although precision drop dispensing is achievable with a variety of technologies, 8 the specific requirements of staining individual biopsies within a TMA required the development of a novel method of reagent delivery. Because the biopsies must remain hydrated after deparaffinization, and they are not simply coated with a layer of reagent as in conventional staining, they must be covered with a layer of Liquid Coverslip–a mineral oil-based compound originally developed for use in Ventana autostainers. This layer of oil, meant to prevent dehydration of the biopsies, prevents the use of conventional drop dispensing technologies. Manual delivery of reagent to individual biopsies in a TMA is not feasible due to the layer of Coverslip present and the compact geometry of the array, where the empty space between each core is often less than 1 mm and the large number of biopsies present in each array would make it exceedingly difficult to track reagent application. Furthermore, any physical contact with the biopsy would destroy the morphological information necessary for diagnosis.
The core technology of the TMAS is a novel-pin design that uses the hydrophobic properties of the pin material and integrated droplet-sensing electrodes to achieve contact-free delivery of aqueous reagent to tissue biopsies. The original design of the pin is shown in Figure 3, and the current, improved version is shown in Figure 4. Both designs consist of two stainless steel electrodes separated by a hydrophobic, nonconducting substrate and use the same principle of operation. When the pin tip is brought into contact with reagent during pickup, the reagent is attracted to the hydrophilic stainless steel components, but repelled by the hydrophobic insulating material. As the pin is withdrawn from the reagent, a droplet will remain on the tip of the pin, attracted by the hydrophilic components. This droplet will remain on the pin even when carried though the oil interface. Presence of the reagent on the pin tip can be repeatedly confirmed by applying a voltage across the electrodes and measuring the resulting current. To deliver the reagent to the biopsy, the applicator is lowered and raised above the selected biopsy in successively larger and larger steps, as indicated in Figure 5. When the meniscus of the reagent droplet comes in contact with the hydrated tissue sample a bridge is formed between the pin tip and the biopsy, and as the pin is retracted from the biopsy the reagent is wicked off of the applicator pin tip and onto the biopsy. When the pin is returned to its starting height the absence of current flow through the electrodes indicates a successful, contact-free delivery.
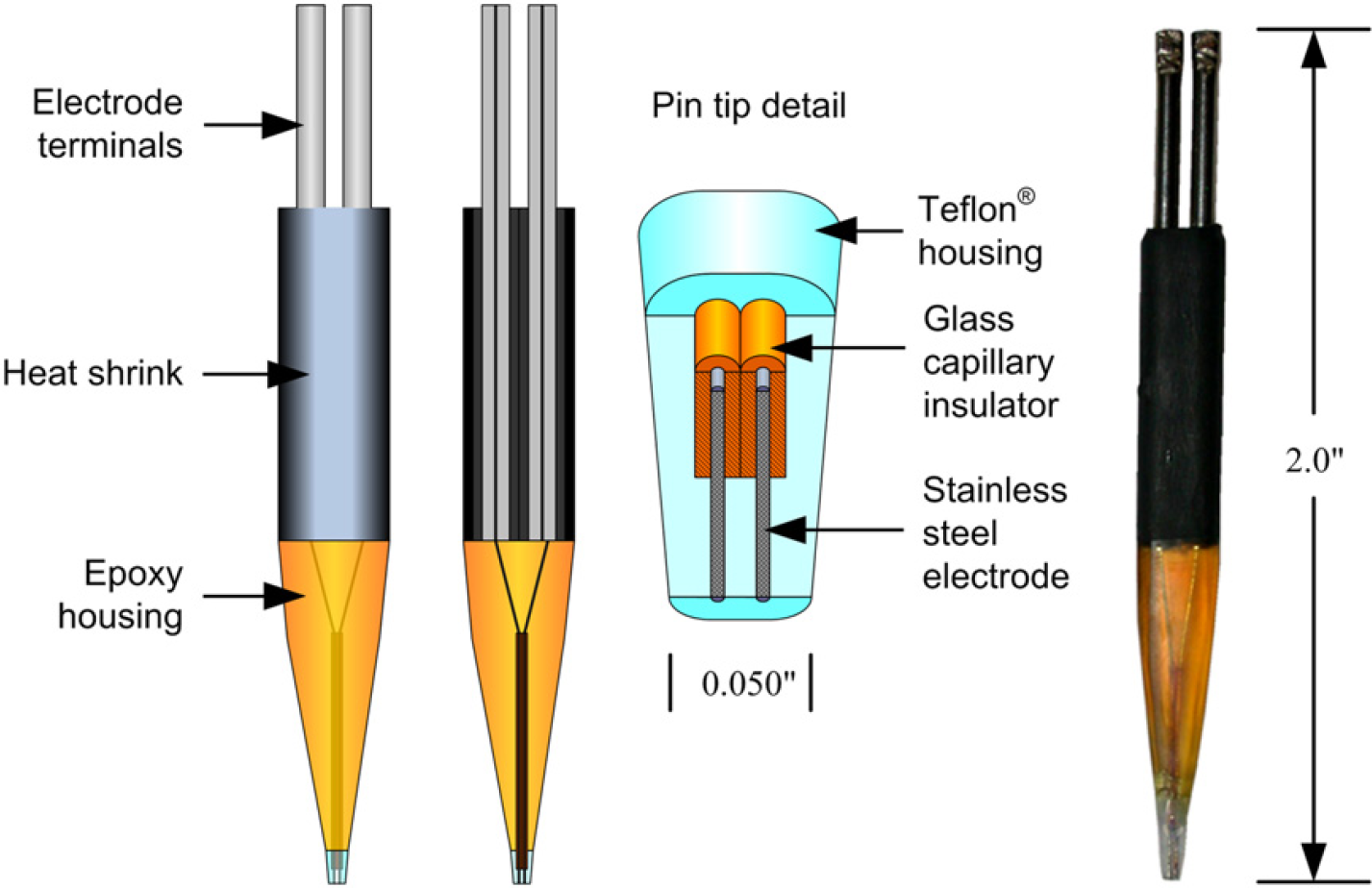
Previous pin design with two stainless steel electrodes side by side.
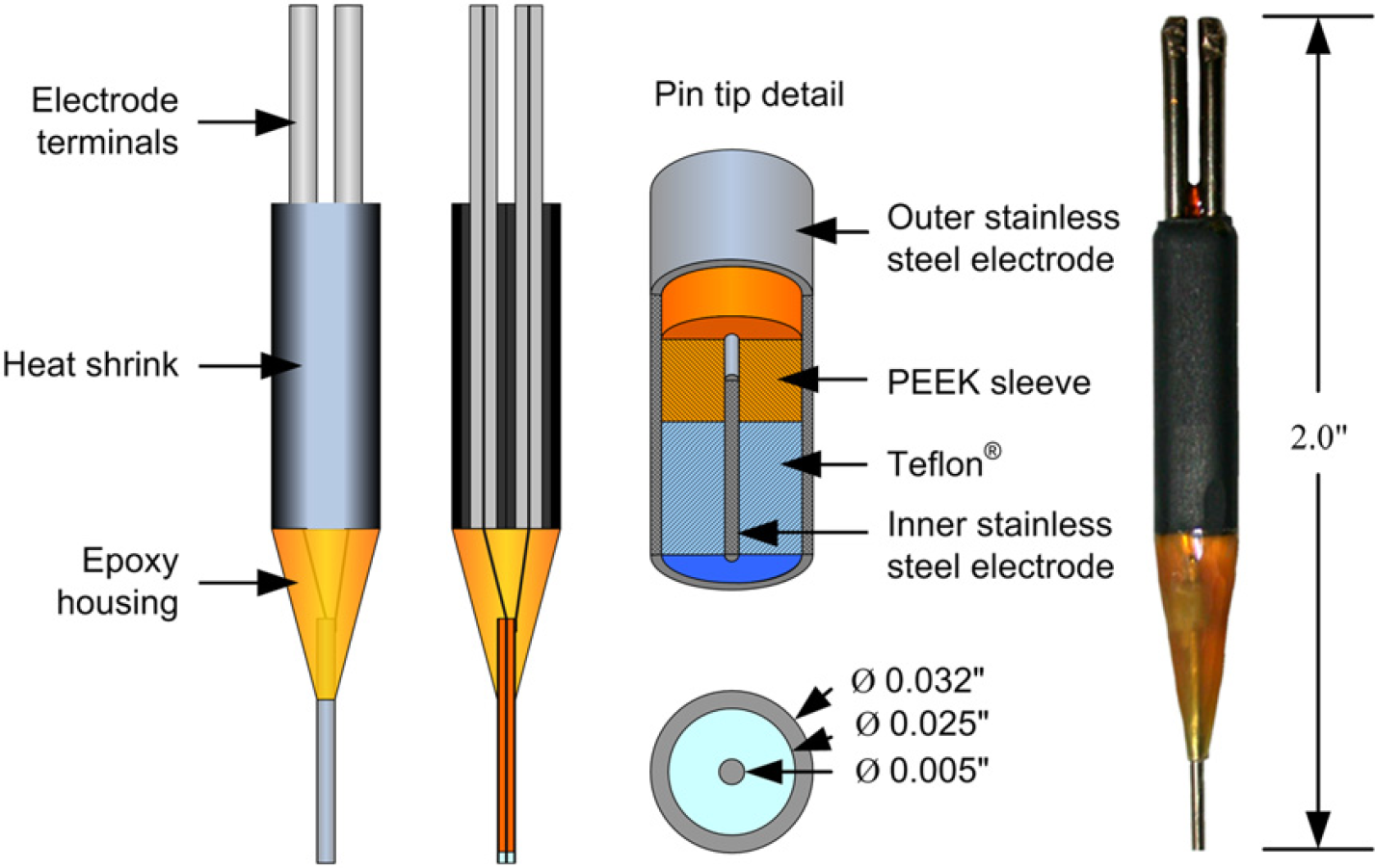
Current pin design with concentric stainless steel electrodes.
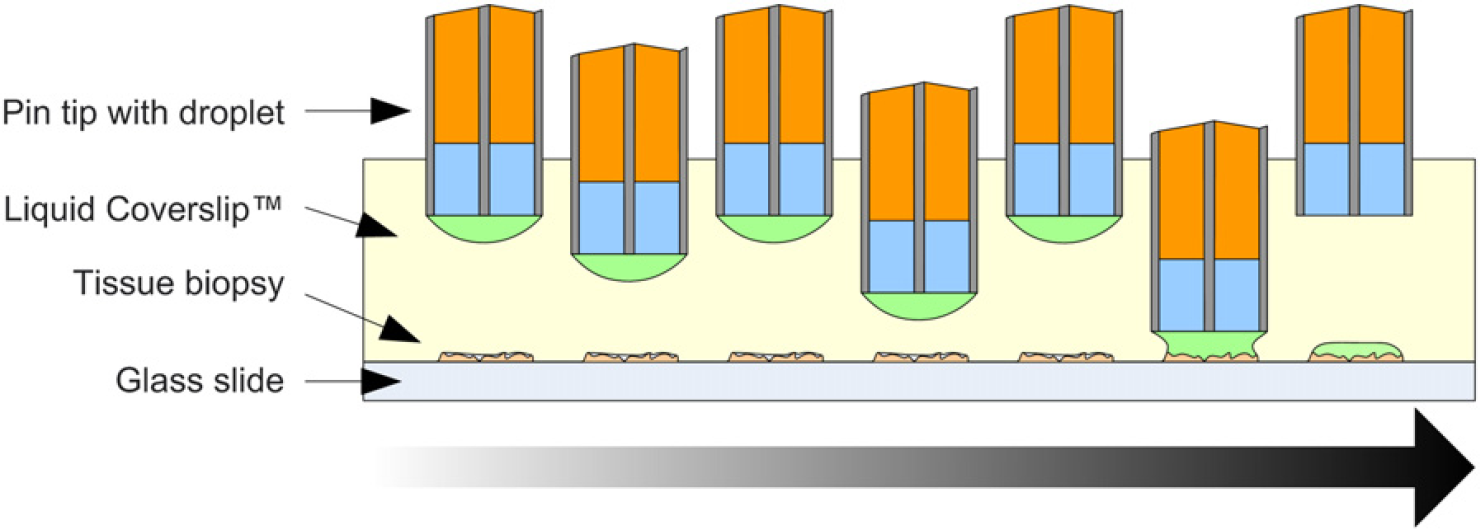
The reagent delivery procedure.
Pin Design and Construction
To achieve the above operation of the applicator pin, it is necessary that the pin tip have a flat, smooth face formed from a hydrophobic insulating material, with two exposed stainless steel surfaces to provide the hydrophilic attraction of the reagent and a conductive path for its detection. The required size of the pin tip is on the order of 0.050 in. diameter to produce nanoliter-sized droplets.
The tip of the first applicator pin (Fig. 3) was formed by inserting the stainless steel electrodes into a piece of double-barreled glass capillary tubing to both physically and electrically separate them. Approximately 2 mm of the electrodes were left exposed, and were then coated with Teflon to form the hydrophobic housing. The tip was polished and ground with fine grain sand paper to expose the electrode faces and to produce a smooth Teflon and stainless steel surface with an oval shape that is approximately 0.050 in. by 0.035 in. Although this design demonstrated the principle of operation, it was difficult to manufacture, and there was significant variation in the geometry of each tip as a result of the hand polishing and grinding with sand paper.
These difficulties motivated the second design (Fig. 4), which consists of concentric stainless steel electrodes separated by a layer of Teflon at the tip. The concentricity of the electrodes is achieved with the use of a polyether ether ketone (PEEK) sleeve with outer and inner diameters matching the corresponding diameters of the inner and outer electrodes. Molten Teflon is drawn into the tip of the pin by a weak vacuum applied to the opposite end of the outer electrode. This design offers several advantages: stock electrode materials in the form of stainless steel capillaries, easier construction from commercially available components, and more symmetric geometry, allowing for easier analysis. Because the outer surface of the pin is formed from commercially available stainless steel capillary tubing that is manufactured to high precision, the variability in the relevant dimensions of the applicator is significantly reduced from pin to pin. The operation of this second pin design is described below.
Analysis of Pin Operation
To pickup a reagent for delivery to a biopsy, the pin is lowered into the reagent well (an uncapped microcentrifuge tube) until current is detected between the electrodes, indicating contact with the reagent surface. As the pin is retracted from the reagent surface, the hydrophilic stainless steel pulls reagent from the well, forming a fluid bridge between the pin tip and the fluid surface in the reagent well. The surface of the fluid bridge assumes a shape such that the total energy of the system is minimized—in this case the gravitational potential energy of the reagent and its surface energy. During this retraction, the contact angle of the liquid–air–steel interface with respect to the end surface of the pin decreases to favor the shape of fluid bridge that minimizes energy (Fig. 6). What is necessary to achieve consistent drop formation is that this contact angle does not decrease below the receding contact angle for the liquid–steel–air interface. If this were to occur, the edge of the droplet would recede inward across the outer electrode, come into contact with the Teflon, and wick off the pin altogether. To prevent this from happening, the shape of the fluid bridge must reach a point where system energy is minimized by separation of the droplet (via a necking mechanism) from the reagent well, forming a separate droplet that covers the entire surface of the pin tip.
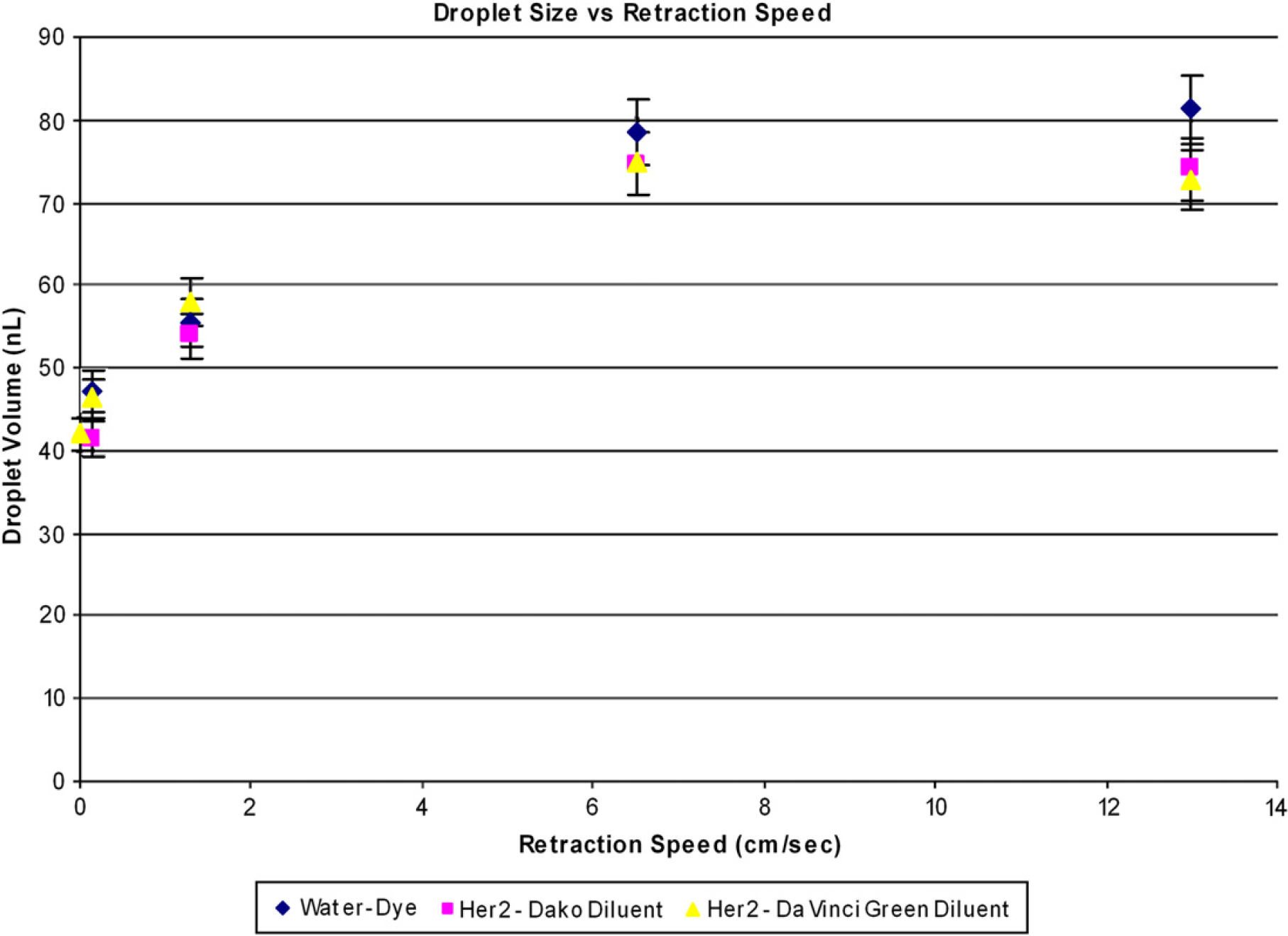
Reagent droplet formation detail.
It is interesting to note at this point that the hydrophobicity of the insulating layer does not come into play during reagent pickup, provided the contact angle at the air–reagent–pin interface does not fall below the receding angle for this interface before necking.
The delivery of reagent to the TMA biopsy is essentially the reverse of this process. When the pin is brought down over the biopsy and the reagent droplet comes into contact with the hydrated biopsy, a fluid bridge forms between the pin tip and the biopsy. However, this exchange takes place under a layer of oil coverslip, instead of in open air, as in pickup. When the pin is retracted from the biopsy, the contact angle of the reagent–oil–steel interface and the end of the pin decreases below the receding angle of the interface, and causes the edge of the fluid bridge to pull across the outer electrode so that it comes in contact with the Teflon. Because the receding angle for the Teflon interface is greater than that for the steel interface, the interface will continue to recede across the Teflon toward the inner electrode before the fluid bridge can break.
After delivery, the pin is put through a cleaning process that includes spray washing with isopropanol, deionized water, and then drying with a burst of hot air. Testing with the first pin design has shown the cleaning method to be sufficient for preventing crosstalk between runs. 6
Although the staining results shown (Fig. 9) were achieved using the first pin design (Fig. 3), all of the tests described below were performed using the newer pin design (Fig. 4). Full testing of the new pin design, including the evaluation of antibody and ISH staining quality and consistency, will be completed as part of the validation studies for the entire TMAS instrument.
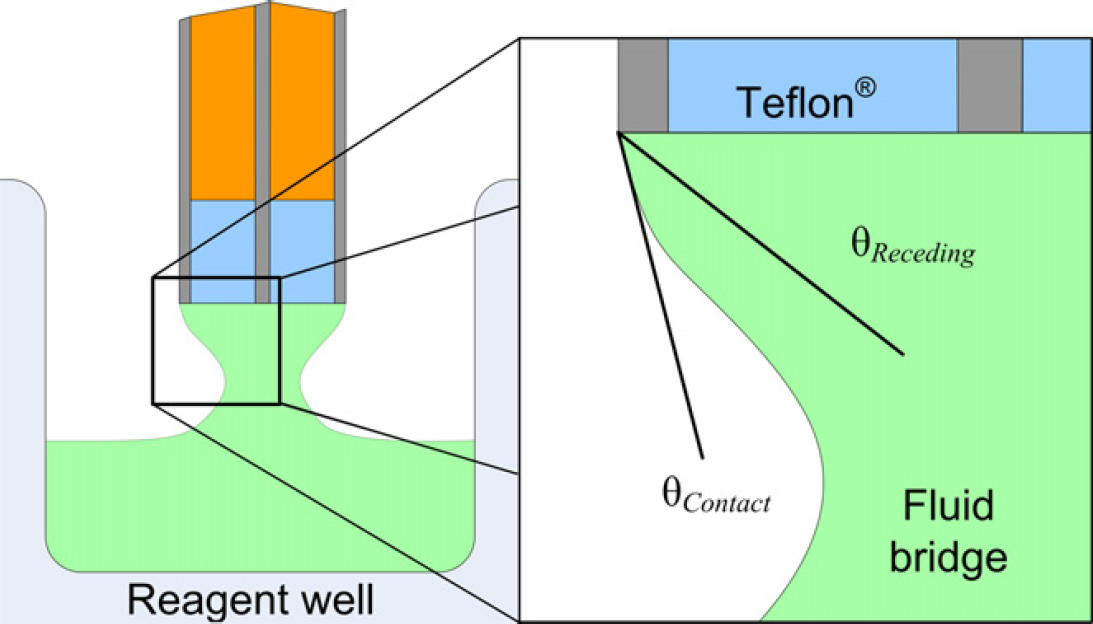
Droplet size versus retraction speed from reagent well.
Materials and Methods
The outer cylindrical electrode is type 304 stainless steel hypodermic tubing, with an outer diameter of 0.032 in. and an inner diameter of 0.025 in. (HTX-21X, Small-Parts Inc., Miami Lakes, FL). The inner electrode is type 304 stainless steel straight, drawn wire, with a diameter of 0.005 in. (GWX-0050, Small-Parts Inc.). The insulating material is Teflon, extracted from dual-type Teflon heat-shrink tubing (SMDT-036, Small-Parts Inc.) and the PEEK sleeve used to align the electrodes has an outer diameter of 0.025 in. and an inner diameter of 0.005 in. (F-182, Upchurch Scientific, Oak Harbor, WA). This pin is mounted into a custom-built electrical socket attached to the tri-axis linear motion system of the TMAS instrument. 6
Three different reagents were used in the tests described:
Water–dye solution: 200 μL green food coloring, 400 μL tap water;
Antibody solution 1: 12 μL Her2 TAB250, 600 μL Dako-Cytomation Antibody Diluent (Dako, Carpinteria, CA); and
Antibody solution 2: 12 μL Her2 Dako A485, 600 μL Da Vinci Green Antibody Diluent (Dako).

Detail of droplet on pin tip.
The antibody solutions were mixed at higher than normal concentrations to exaggerate any possible effects the antibody might have on the pin performance.
Droplet sizes on the pin tip after reagent pickup were measured from a photograph of the pin tip obtained from a digital camera attached to a microscope viewing the pin under 80× magnification (Fig. 6). The volume of the droplet was determined from the droplet's geometry, assuming that it formed a semispherical cap over the tip, and using the size of the outer electrode as a reference dimension (calculations not shown). The error for this method was determined to be ±10%.
Results
Factors Affecting Reagent Pickup
The above discussion of reagent pickup assumed a simple quasi-static scenario in which momentum and kinetic energy do not play a role. In reality, however, the speed of pin retraction has an important effect on the volume of the reagent droplet formed on the pin tip. 9 Several tests were performed using the three different reagent solutions and the results are plotted in Figure 8. An upper bound on droplet volume exists above approximately 6.5 cm/s, whereas a lower bound exists below 0.1 cm/s, for the given pin geometry. There was no significant variation in droplet volume across the different reagents.

Staining of biopsies with food coloring achieved using the first pin design. The variation in the shape is due to the initial shape of the biopsy. (A penny in the background indicates the size of the biopsies.)
The shape of the meniscus inside the fluid well was found to be an important parameter, as would be expected from the aforementioned fluid bridge model. A concave meniscus was found to produce 45 ± 3 nL droplets, whereas a convex meniscus resulted in 75 ± 4 nL droplets when the retraction speed was 0.13 cm/s. To minimize the effects of meniscus shape on droplet volume, it is important to ensure that the level of reagent within the reagent well is within the bounds required to produce a consistent meniscus–that is not too full (convex) and not too empty (affected by the shape of the reagent well bottom). It is also important to ensure that the reagent is retrieved from the center of the circular well, so that the fluid bridge geometry has radial symmetry to ensure consistency of droplet size well to well.
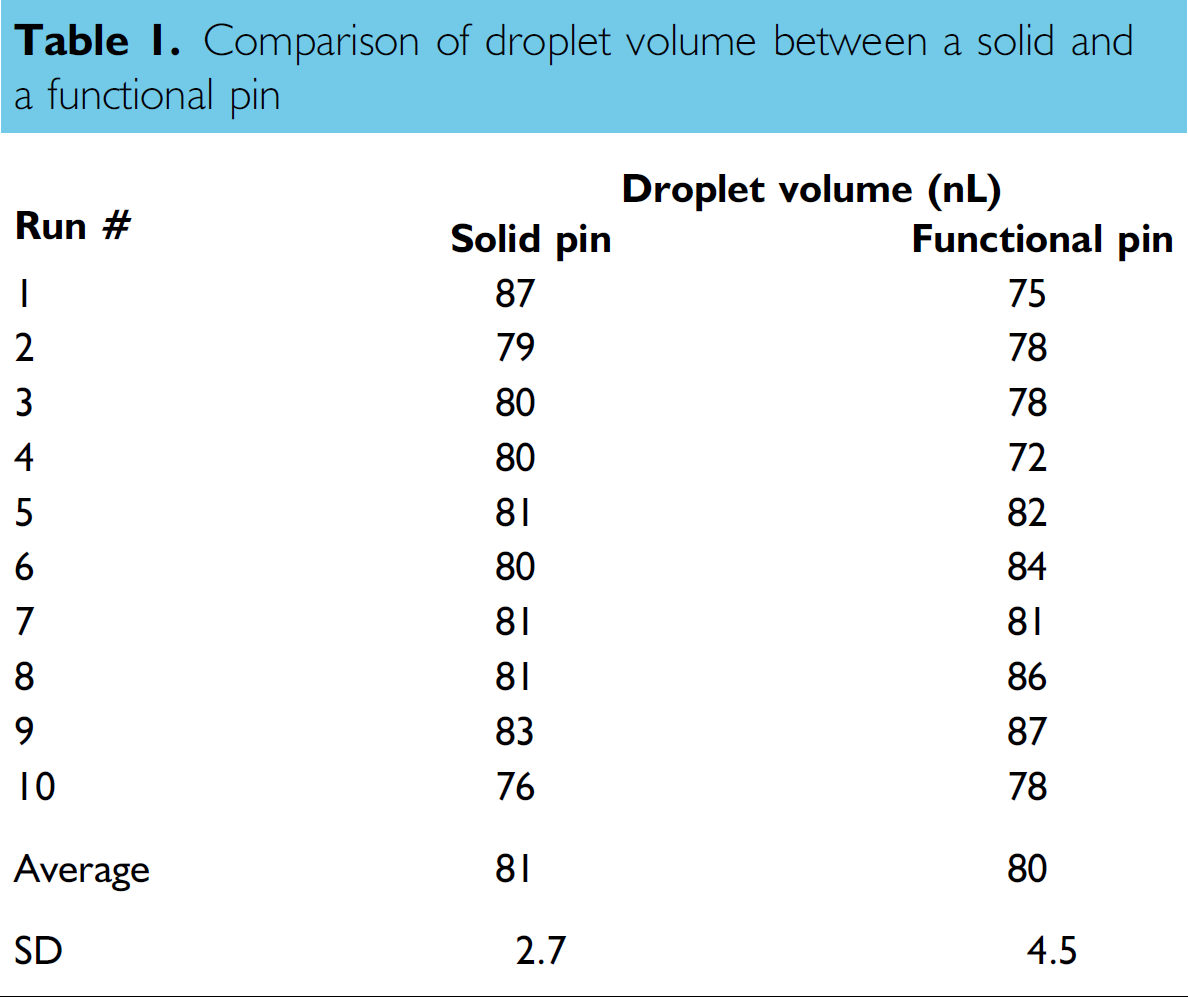
To test the validity of the claim that the hydrophobic insulator layer does not affect the pickup of the reagent as long as the contact angle is greater than the receding angle, the average droplet size produced by a solid stainless steel pin was compared to that of a pin with the described construction, both of which had the same outer diameter. The averages of the drop sizes produced by the two pins are nearly identical, as shown in table 1.
Comparison of droplet volume between a solid and a functional pin
Factors Affecting Reagent Delivery
A successful delivery in this application can be considered as the deposition of the majority (90%) of the reagent to the biopsy in such a manner that no reagent remains spanning the two electrodes, so that the controller is able to identify that delivery has occurred. Other than the hydrophobicity of the insulating layer discussed above, the most important factor affecting droplet delivery to the tissue biopsies is the initial volume of the reagent droplet after pickup. Because the tissue biopsies are small (0.6-mm diameter, 5-μm thickness), the amount of water they hold after hydration, and before they are placed under the Liquid Coverslip, is somewhere in the order of tens of nanoliters. This number represents, to a certain extent, the nominal volume of water that each biopsy can hold. When reagent is applied to the biopsy, the amount of liquid the biopsy is asked to hold is effectively doubled. To test delivery of a variety of droplet sizes, the evaporation of reagent from the pin tip was carefully monitored and used as a means of providing a range of droplet sizes. After a number of tests, it was determined that the biopsies have difficulty accepting droplets above 25 nL. At this volume, the delivery success rate was greater than 95% (107/112), whereas it was less than 10% for 50-nL droplets. During a failed delivery, enough reagent remains on the tip of the pin to span the electrodes, and the controller is unable to recognize that liquid delivery has occurred. As a result, the controller continues to lower the pin toward the biopsy in the iterative procedure described above until the pin tip makes mechanical contact with the slide and the biopsy and a limit switch trips to stop the delivery.
As previously discussed, the hydrophobicities of the inner materials do not have an impact on the volume of the reagent picked up, though they play an important role in delivering reagent to the biopsy. Once the contact angle for the reagent–oil–electrode interface decreases below the receding angle for this interface, the reagent boundary will pull back to the insulating layer. If the hydrophobicity of the insulating layer is greater than that of the stainless steel electrode, the angle condition at this interface will already be broken and the liquid will pull back toward the inner electrode. Because the inner electrode is stainless steel, and therefore hydrophilic, it is important that its diameter be as small as possible so that any reagent that is retained on the inner electrode has the smallest possible volume. Furthermore, by reducing the size of the inner electrode, the amount of hydrophobic material that a remaining droplet must span to keep the circuit closed and cause a failed delivery is increased.
PEEK capillary tubing shown in Figure 4 was also tested as the exposed insulating layer at the pin tip. Despite the ease of manufacturing offered by commercially available PEEK components, it proved to be incapable of delivering reagent to the biopsies. Its lower surface energy and greater affinity to aqueous fluids result in a receding angle that is much smaller than that for a Teflon interface.
Conclusion
To answer the specific need of staining individual biopsies in a TMA, we have developed a novel applicator technology that is capable of noncontact delivery of small volumes of aqueous reagents to hydrated tissue biopsies through a layer of Liquid Coverslip. Working from a proven general concept, we have developed a new, more easily fabricated and uniform applicator pin that will be amenable to manufacturing in high quantities. The demonstrated pin geometry can be adapted to dispense smaller volumes to prevent biopsy overloading. Although tests on the previous pin design are promising, testing continues to determine the useable lifetime of the new pin design.
Acknowledgments
This work is supported by the Genome British Columbia Technology Development Platform, an initiative to provide engineering support to help expedite the research process, leading to faster, better, and more efficient solutions. The authors would like to thank the members of the UBC Applied Biophysics Laboratory, specifically Jason Thompson and Ivan Chan for their help with various aspects of this project, as well as the UBC Engineering Physics Program, specifically Drs. Harold Davis and Jonathan Nakane, for supporting the project upon which this manuscript is based.
