Abstract
Biochemical markers of bone turnover suffer from large analytical and natural fluctuations (20-30%), making small differences in bone resorption impossible to resolve. This limits the clinical utility of such markers for individuals with the skeletal complications associated with many disease states (e.g., metastatic cancer, renal failure, osteoporosis). We are developing the capability to measure small changes (5-10%) in bone turnover rate in vivo by tagging the living skeleton with 41Ca. Among the stable and radioactive calcium isotopes, only 41Ca is useful for direct quantitation of bone turnover because it is extremely rare in nature and radiologically benign (105 years half-life, pure electron capture decay). The ratio of this tracer to total calcium remains quantifiable in body fluids and excreta via accelerator mass spectrometry (AMS) for many years following a single physiological-sized oral or intravenous dose. The highly automated AMS instrumentation and streamlined sample preparation allows a single operator to prepare or run more than 100 samples per day—significantly more than other 41Ca programs worldwide. We intend to exploit these measurements for earlier diagnosis of pathological processes and interactive intervention with therapeutic agents, allowing modulation of these agents to obtain the best individual result for a patient.
High-throughput accelerator mass spectometry
The Center for AMS at Lawrence Livermore National Laboratory was set up for high-throughput, routine analyses of multiple nuclides (3H, 10Be, 14C, 26Al, 36Cl, 41Ca, 129I, 236U, and 239Pu). Notwithstanding instrumentation complexity, a highly automated AMS control system allows a single operator to set up and measure any of these nuclides daily. High throughput is enabled through a bright negative ion source (up to 3 μA CaF3 −), with highly efficient ion extraction and beam transport to the gas ionization detector, in addition to excellent vacuum maintenance.
Sample 41Ca to total calcium (41Ca/Ca) isotope ratios are measured to <3% precision for unknowns and standards with known isotope ratios. Software monitoring of all instrumental parameters assures accurate results: data generation is paused when any beamline component strays from online control charts. Accuracy is further assured with frequent standard measurements interposed between unknown samples and blanks. As outlined in Figure 1, AMS 41Ca measurements are accomplished via: (1) mass separation of CaF3 − molecules, (2) acceleration of these molecules through 9 million volts, (3) removal of nine electrons through charge exchange in a thin carbon foil at the high-voltage terminal (thus destroying all molecular interferences), (4) a second stage of acceleration to ground potential, (5) fragment mass analysis, (6) measurement of 40Ca8+ in an offset Faraday cup, and (7) ion identification and single particle counting of 41Ca8+ in a multianode gas ionization detector.
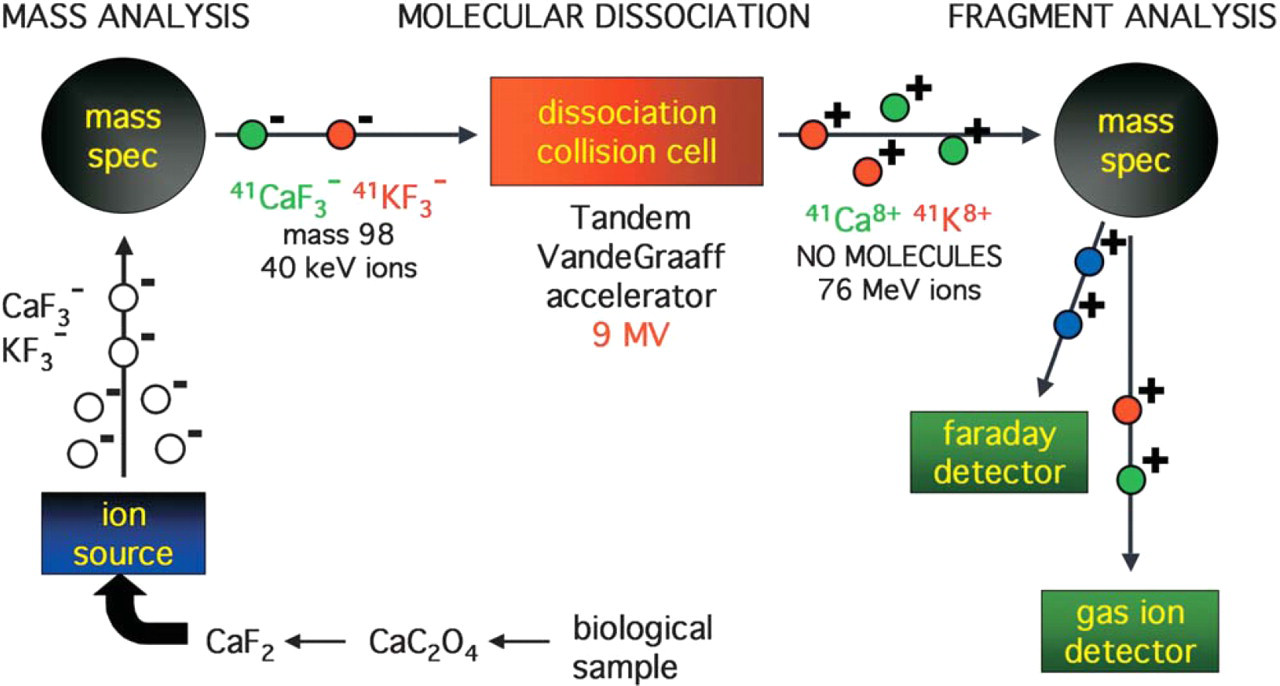
Schematic of AMS instrumentation.
Our analytical 41Ca/Ca background level is 2 × 10−14. Our limit of quantitation is 41Ca/Ca < 1 × 10−13, which equates to about 0.01fmol 41Ca per 25 mL urine or 2.5 mL plasma. Measurement precision is monitored using secondary standards similar in isotope ratio to unknowns, and is currently <3%, assuming sufficient sample 41Ca content to identify at least 1,000 41Ca ions.
Sample processing involves selective precipitation (as calcium oxalate) and ion exchange purification of Ca, followed by calcium fluoride production using HF. 1 We are also exploring the use of less dangerous sources of fluoride ions, with promising results from most fluoride salts examined.
Model delvelopment
We used AMS measurements of urinary 41Ca/Ca from a human subject (Figure 2) to develop a linear kinetic model for human calcium homeostasis (Figure 3), using the Berkeley Madonna software package (Berkeley, California). We used this model to predict 41Ca/Ca change with bone turnover rate increases of 10–100% over baseline (Figure 4). These small changes in turnover rate cannot otherwise be measured, and we anticipate that our method will enable early diagnosis of metastasis as well as close monitoring of therapeutic effects on bone turnover.
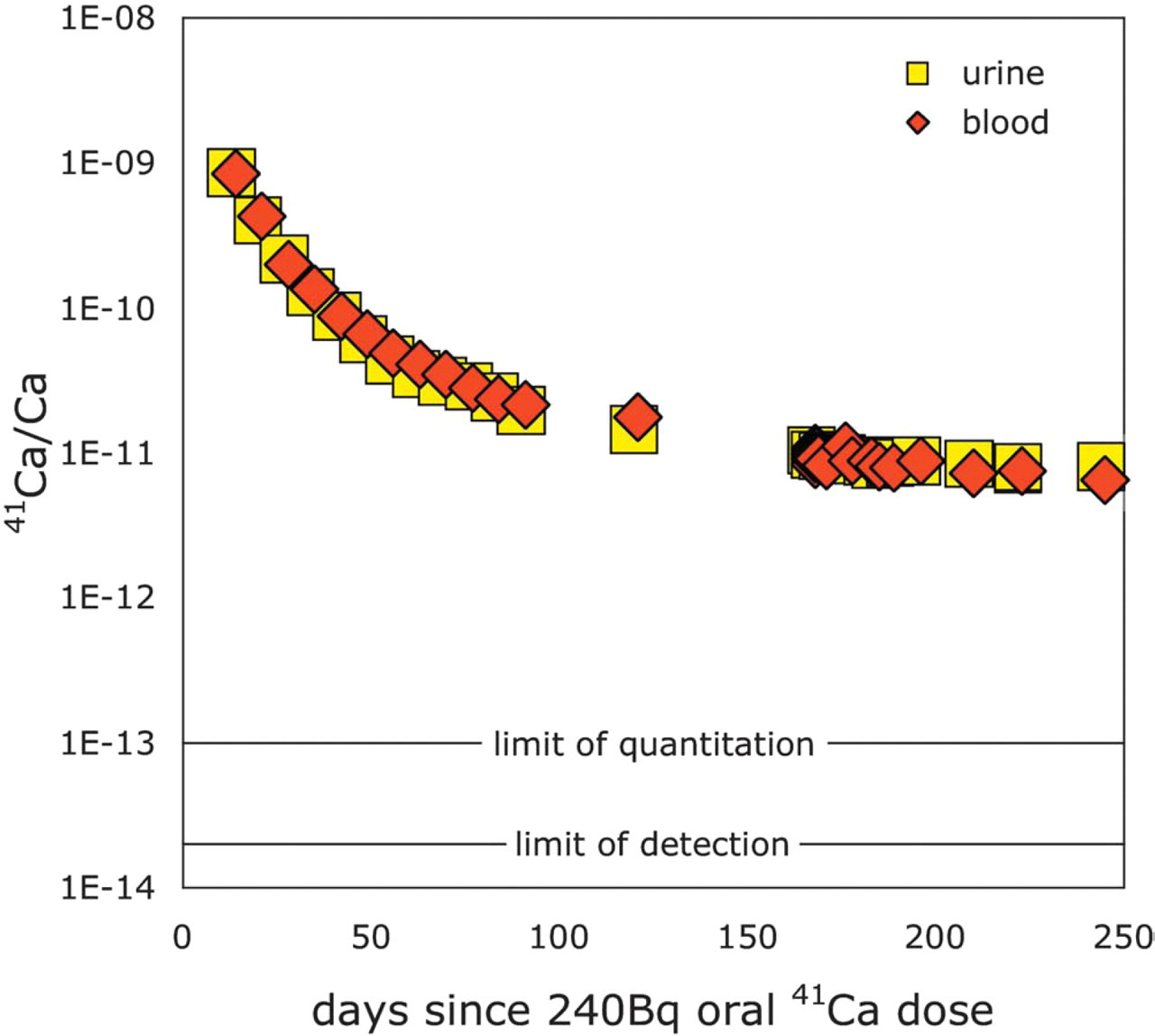
Urinary and serum 4lCa/Ca following a single oral 4lCa dose in a healthy postmenopausal woman.
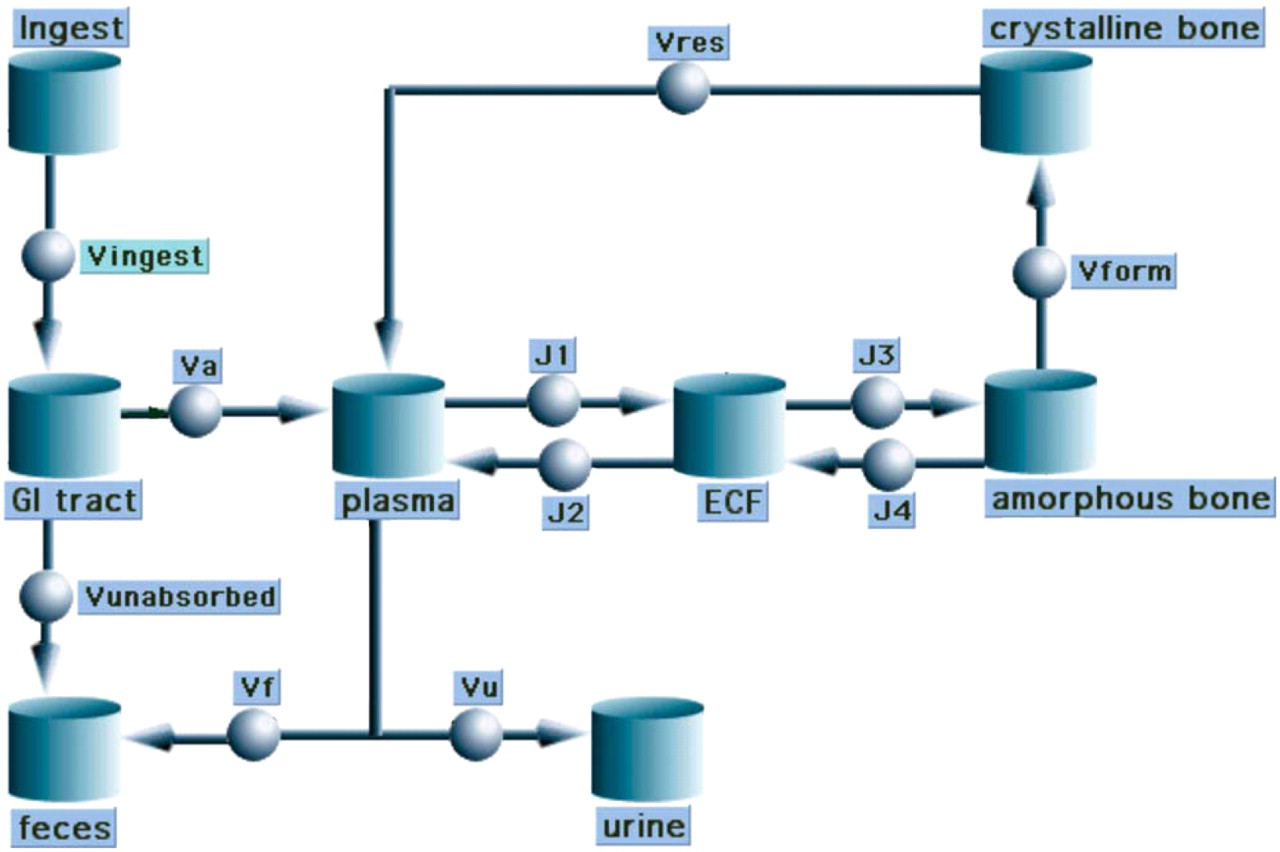
Compartmental model for calcium homeostasis.
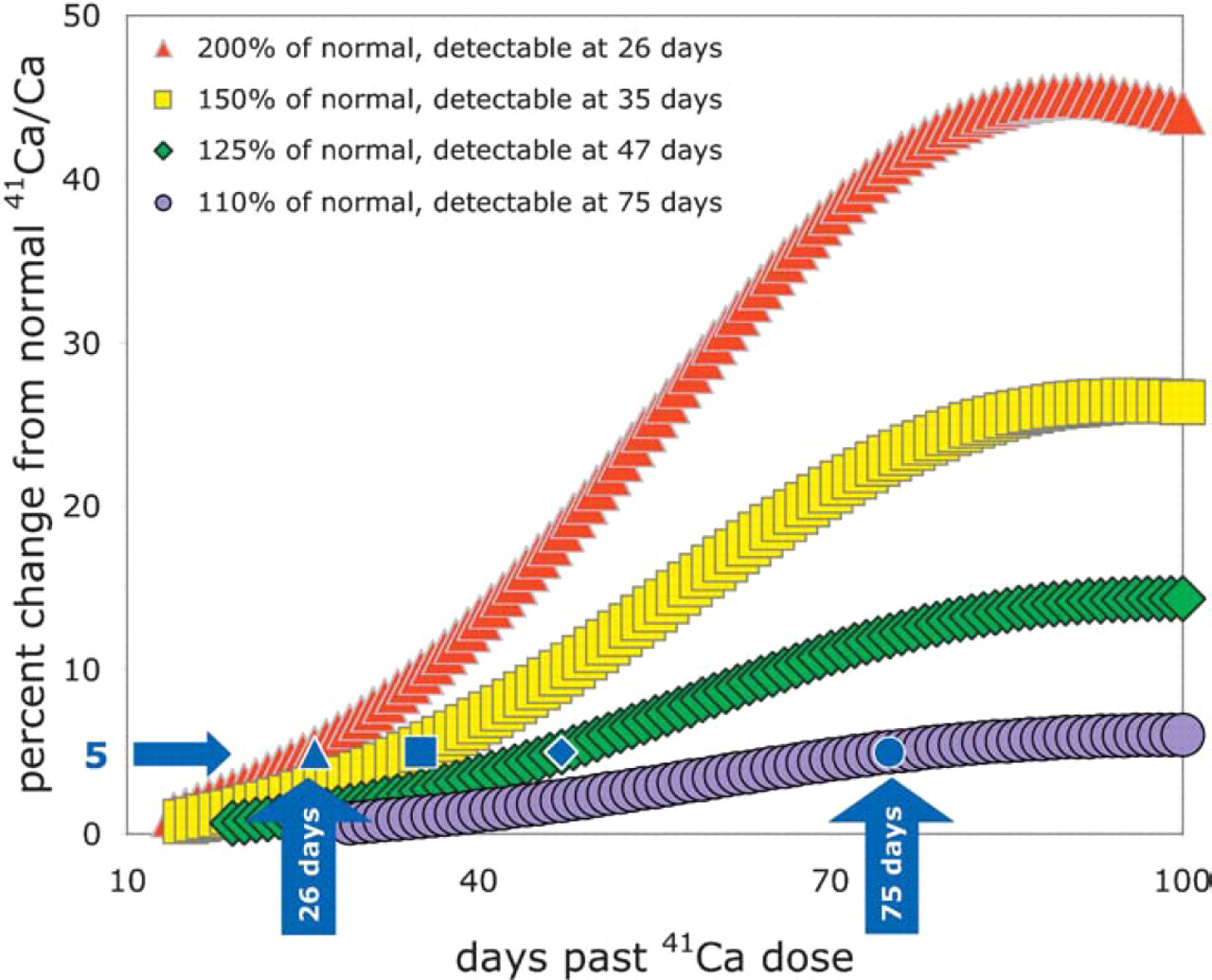
Anticipated deviations from normal 41Ca/Ca for individuals with increased bone resorption rate; time to reach 5% deviation from normal is relevant because typical AMS uncertainties are 2–3%.
Cancer research
When cancer spreads to the skeleton, there are three principle disturbances to the continuous bone remodeling sequence: (1) increased bone turnover rate, (2) imbalance at remodeling sites, and (3) uncoupling of bone formation and resorption. Figure 5 is a schematic of the transition from primary neoplasia through to uncoupled bone turnover and patholocial humoral fracture (in this case, from metastatic breast cancer). The process of localized skeletal metastasis may be approximately understood as a vicious cycle where normal bone remodeling cells are stimulated under the influence of invading cancer. 2 Increased bone resorption produces locally concentrated bone-derived growth factors, stimulating further bone destruction through formation and activation of bone resorbing cells (osteoclasts). Certain growth factors may also contribute to uncoupled bone formation through stimulation of normal bone forming cells (osteoblasts). Tumor invasion results in disruption of normal bone turnover, leading to additional cancer cell growth, leading to further disruption of the bone remodeling sequence.
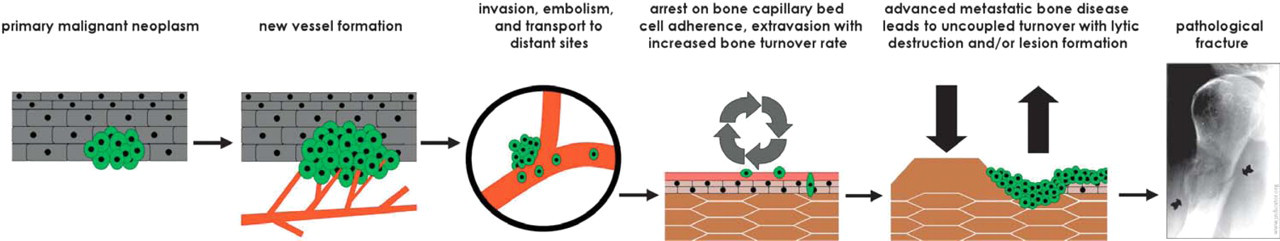
Cancer progression from primary site to bone.
Changes in bone turnover as the result of diseases such as cancer and osteoporosis have been widely studied using serum and urinary bone degradation biomarkers such as deoxypyridinoline and N-telopeptides, which are bone specific cross-linked fragments of Type I collagen. Such organic biomarkers are currently of limited utility in detecting skeletal metastases because of low measurement precision. 3 In addition, these markers cannot distinguish individuals with diagnosed metastatic bone disease from healthy individuals. However, when data from many individuals of similar pathology are averaged, the pathological increases in bone turnover become distinguishable. We hypothesize that de-mineralized skeletal calcium released during increased bone turnover will be quantifiable as a change in 41Ca tracer in body fluids and excreta.
Initial application of our method in cancer research will involve a control group of healthy men compared to individuals with diagnosed metastatic bone disease from prostate cancer. As illustrated in Figure 4, increases in bone turnover that accompany tumor growth and metastatic bone disease will be measured as a change in measured 41Ca/Ca from the control group. Normal bone is destroyed and replaced at a rate of about 500 mg/d, and the initial stages of metastatic progression to bone would cause a local increase in turnover rate 3 : if 2% of the active bone resorption sites show a five-fold increase in turnover rate, the resulting global resorption rate would be 550 mg/d. This 10% increase in resorption should change the measured 41Ca/Ca versus healthy individuals; according to our kinetic model, this difference should become measurable by 75 days past 41Ca dose administration (Figure 4). Larger tumor burdens should become differentiable from the normal range in time frames of several weeks. For patients who have been given the 41Ca tracer prior to the onset of metastatic bone disease, the increase in 41Ca/Ca will be more easily detected because the tracer will have time to clear from all body calcium pools except the skeleton—effectively decreasing the body-wide 41Ca/Ca and making a small change in bone turnover immediately detectable. Therefore, our method should be useful for both detecting and quantifying the extent of clinically apparent skeletal metastatic sites (thus allowing accurate disease staging) as well as detecting the early onset of metastatic bone disease in apparently healthy individuals.
Conclusion
We developed a linear kinetic model of human calcium balance and used it to predict that our method is capable of detecting a 10% change in bone turnover rate for individual human subjects. This capability should allow us to closely monitor individuals with changes in bone turnover associated with various diseases, including osteoporosis, cancer, kidney diseases, arthritis, and many others. We have initiated a research program to study long-term calcium balance in prostate cancer patients, towards better disease staging, clinical management and pre-symptomatic determination of disease progression.
Acknowledgments
The authors would like to acknowledge Marta VanLoan, Erik Gertz, Stewart Freeman, and Leonard Deftos for valuable discussions and advice. This work was performed under the auspices of the U.S. Department of Energy by the University of California Lawrence Livermore National Laboratory under Contract No. W-7405-Eng-Y8 and under the National Center for Research Resources Grant No. RR13461. This project is funded under the Laboratory Directed Research and Development, Exploratory Research Institute program.
