Abstract
Automated parallel synthesis with multivariate analysis was employed in the optimization of a Pd-catalyzed three-component cascade reaction of 7-buta-2,3-dienyl-1,3-dimethyl-3,7-dihydropurine-2,6-dione with iodobenzene and morpholine (see Scheme 1). Initially a range of reaction factors were analyzed using experimental design to optimize for selectivity, conversion, and impurity levels. Solvents and bases were screened separately as the substantial number of discrete variables involved is unsuited to the fractional factorial design used. Instead, a solvent principal component analysis (PCA) model was used in selection of solvents for screening, so gaining maximum variation in solvent properties. Similarly, tertiary amines were chosen by pKa for screening as bases alongside inorganic salts. This investigation yielded twofold results, not only in enhancement of the reaction selectivity, but since significantly dierent findings were obtained on changing the solvent, the importance of the sequence of experimentation is also emphasized.
Introduction
This type of one-pot cascade process provides an atom efficient synthesis of tri-substituted double bonds. The mechanism proceeds via intermediate π-allyl palladium (II) complexes that can interconvert through rearrangement to the σ-allyl complexes. 1 Nucleophilic attack by morpholine occurs at the least substituted carbon of the syn- and anti-palladium π-allyl complexes furnishing the product as a mixture of E/Z stereoisomers (see Scheme 2). It is possible to obtain a single isomer from this type of cascade process, although this is not the case with every reaction; the chemistry presented here being one where the stereoselectivity has not responded to routine one-factor-at-a-time optimization. Previous experimentation had managed only an increase from a 1:1 to 4:1 Z/E (60% d.e.) mixture of stereoisomers, without consideration of yield or impurity profile, and only inorganic bases had been thoroughly screened. The goal of this investigation was to gain an understanding of the underlying reaction variables controlling stereoselectivity in order to increase the E/Z ratio as well as improving the impurity profile.
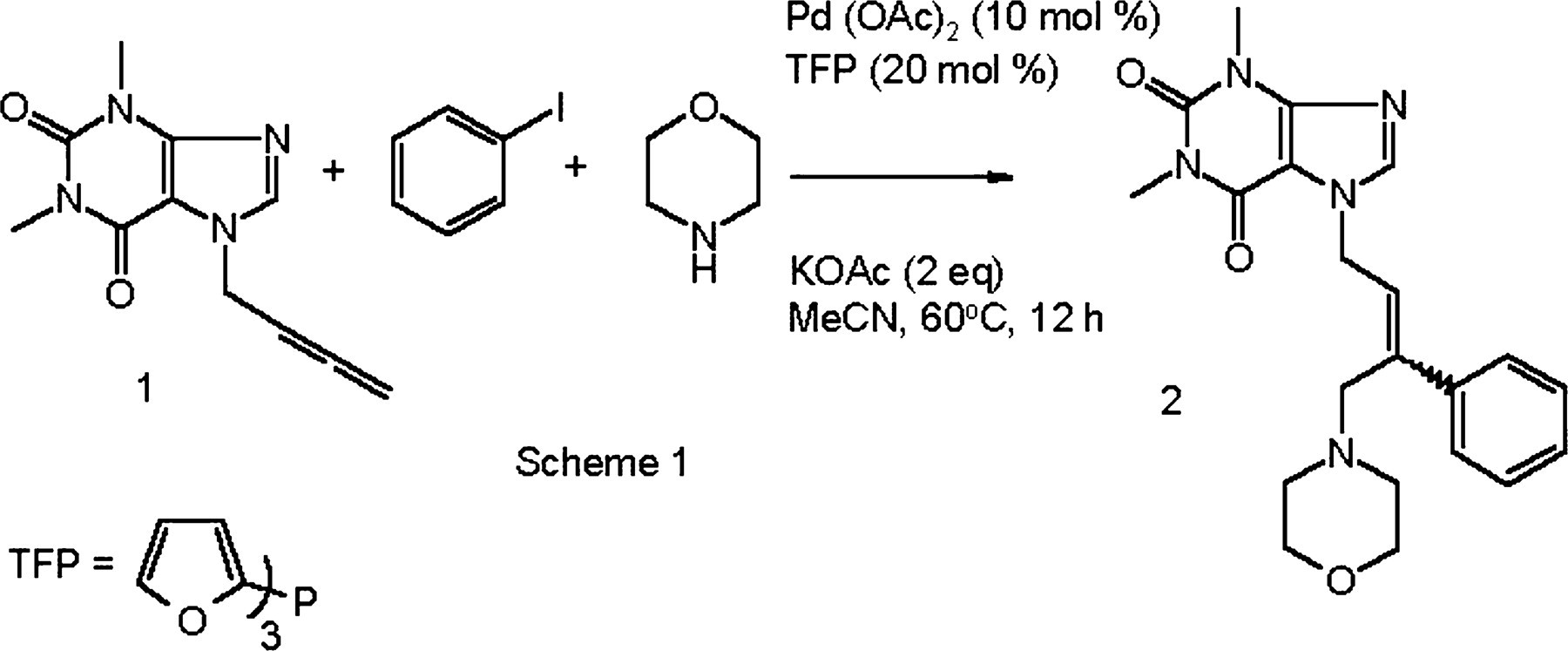
A palladium-catalyzed three-component cascade reaction.
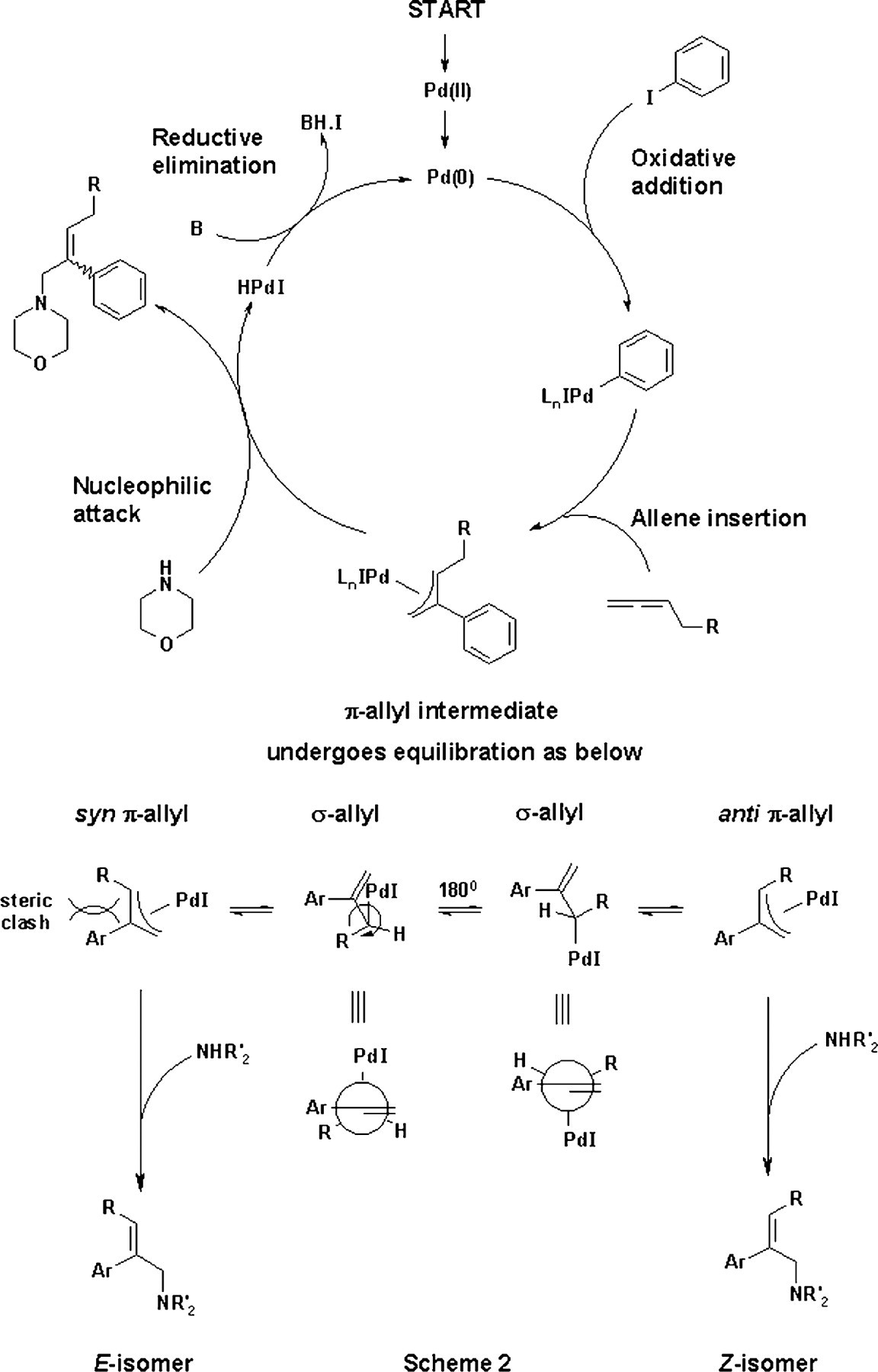
Mechanism of the three-component cascade reaction.
Design 1
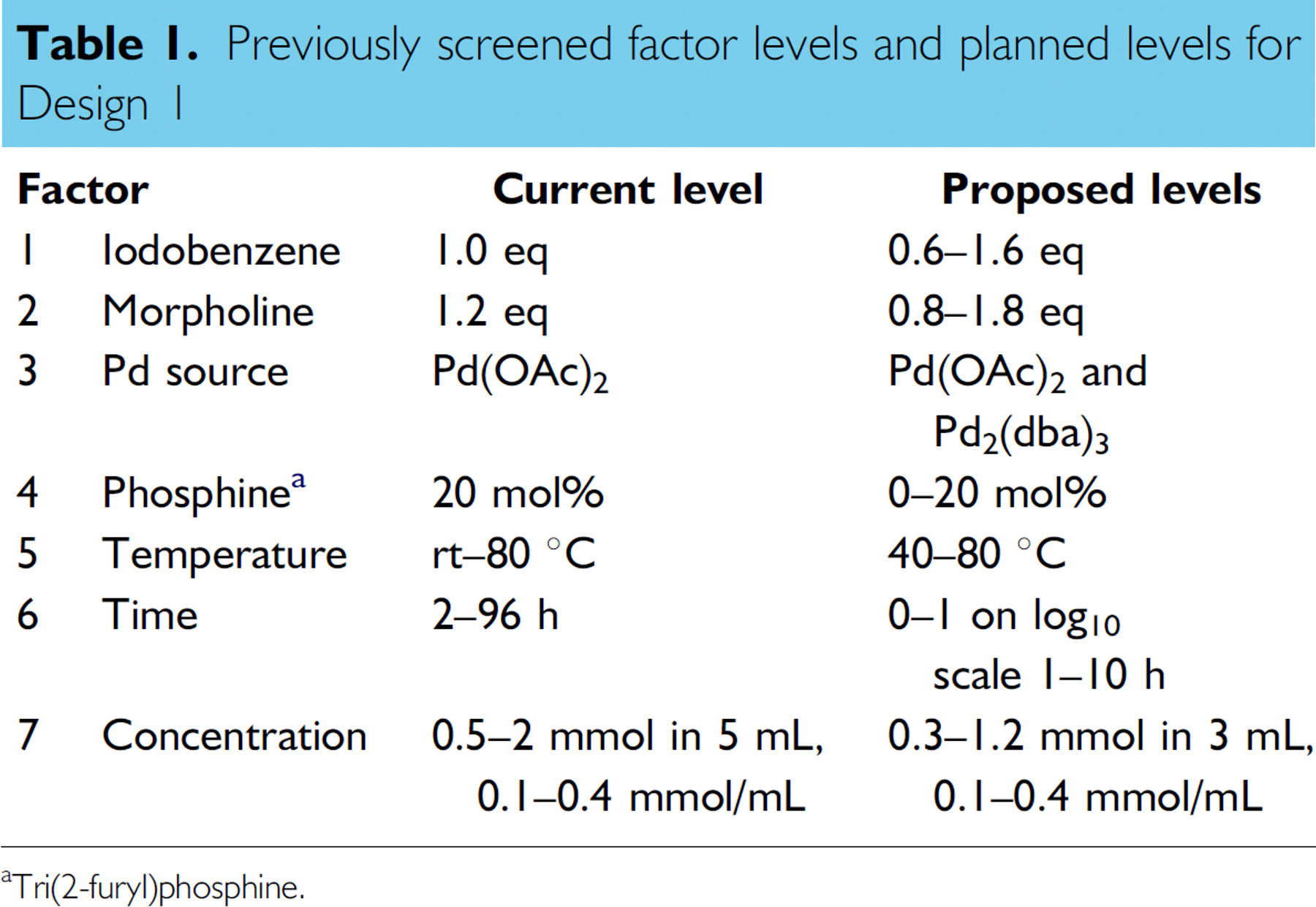
In order to screen as large a number of reaction variables as possible in the initial design, solvent and base were screened independently as the inclusion of a large number of categorical variables in a factorial design would result in a combinatorial explosion! Thus, solvent and base conditions selected for the pre-screen design, acetonitrile and potassium acetate, were those providing best selectivity in previous studies. Design 1 examined the effect of six numerical factors and catalyst type on the responses (Table 1), which were Z-isomer peak area (PA), E-isomer PA, and selectivity (Z/E).
Previously screened factor levels and planned levels for Design 1
Tri(2-furyl)phosphine.
All reactions employed the Anachem SK233 Workstation with online HPLC monitoring; chromatography data were collected and integrated in Unipoint before analysis in Design Expert 6 (Stat-Ease Inc., 2021 East Hennepin Ave., Suite 480, Minneapolis, MN 55413) and with custom Excel macros.
Design 1 identified the major factors affecting selectivity and product yield as reaction temperature and the mol % of phosphine ligand employed. These factors also interact to influence the reaction outcome; in the presence of phosphine, selectivity is moderate at 40 °C and declines as temperature increases, showing that more of the reaction proceeds via the sterically more hindered syn π-allyl. Under phosphine-free conditions the lack of a donor ligand results in poor selectivity at low temperature as both isomers are formed in approximately equal proportions, as the temperature is increased, selectivity for the anti-allylpalladium complex increases.
A set of optimal conditions for use in screening of solvent and bases can be found by setting lower limit constraints for selectivity and product peak area data to create a contour overlay plot, Figure 1. The area highlighted in yellow defines the region of the reaction space in which the response data fits the constraints applied, in this case selectivity > 3.5:1 Z/E, E-isomer PA < 4.0 × 106 and Z-isomer PA > 1.0 × 107. Although the importance of the presence of phosphine for high yield was demonstrated, the low temperature range recommended from this initial study was not used in solvent and base screening due to issues with poor conversion.
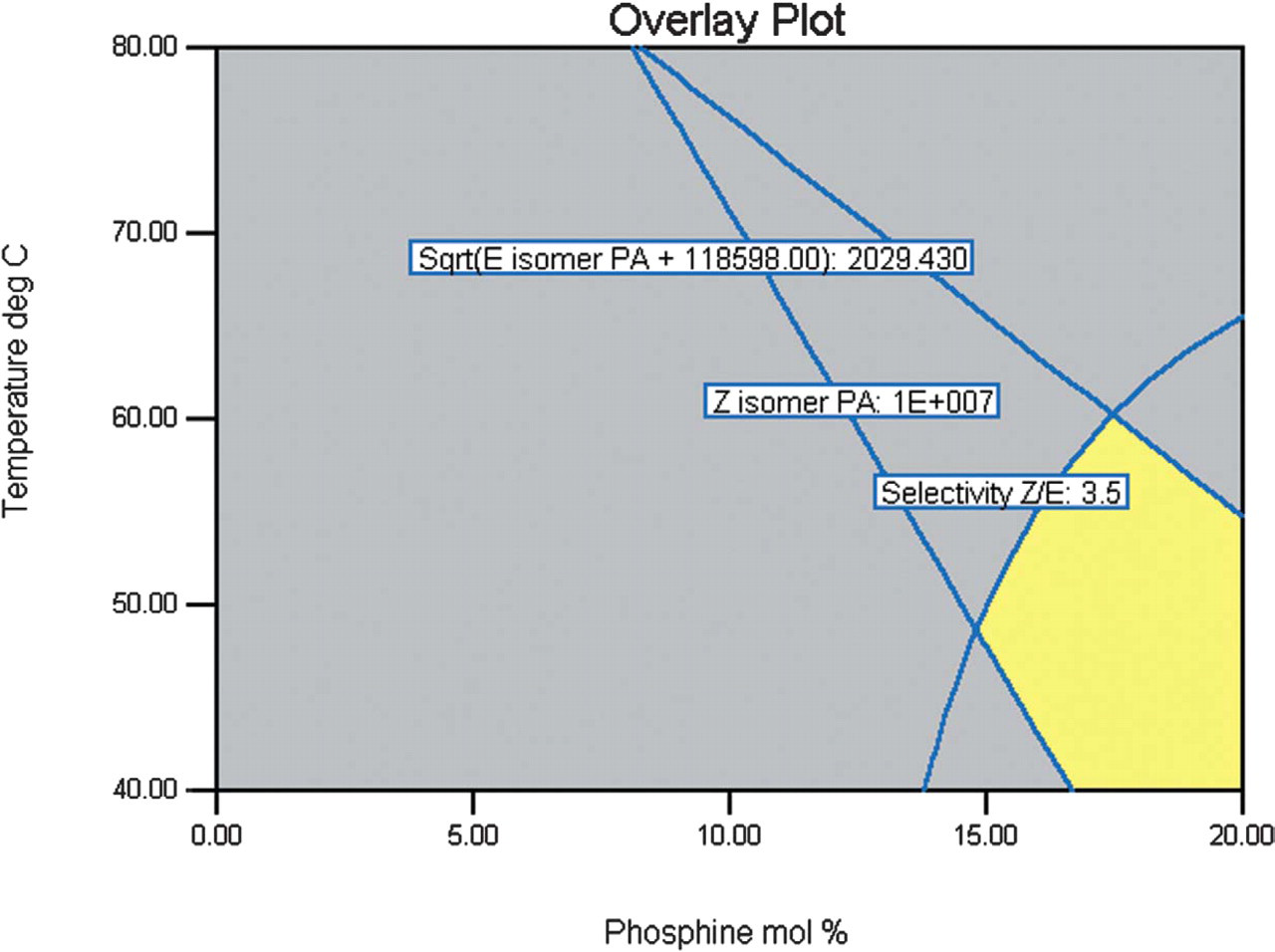
Design 1 contour overlay plot.
Solvent and Base Screen
PCA is a chemometric technique providing data compression and facilitating extraction of information. The variance in a data set is projected onto lower dimensional axes, which can represent underlying variables in the system. 2 For example, the corrected Carlson model 3,4 used reduces an eight-dimensional data set into a two-dimensional plot in which the PC1 axis represents solvent polarity and PC2 solvent polarizability.
A screening experiment involving ten bases and nine solvents, Table 2, was performed using the GSK μDART (a modified version of the Anachem SK233 which has the facility for 96 parallel reactions) with HPLC analysis. Solvents were selected from the PCA model such that all areas of the solvent space were covered, although not necessarily in a regular lattice, and chosen from the solvent classes as defined by Chastrette 5,6 to gain a wide distribution of solvent properties.
Solvent and base screen
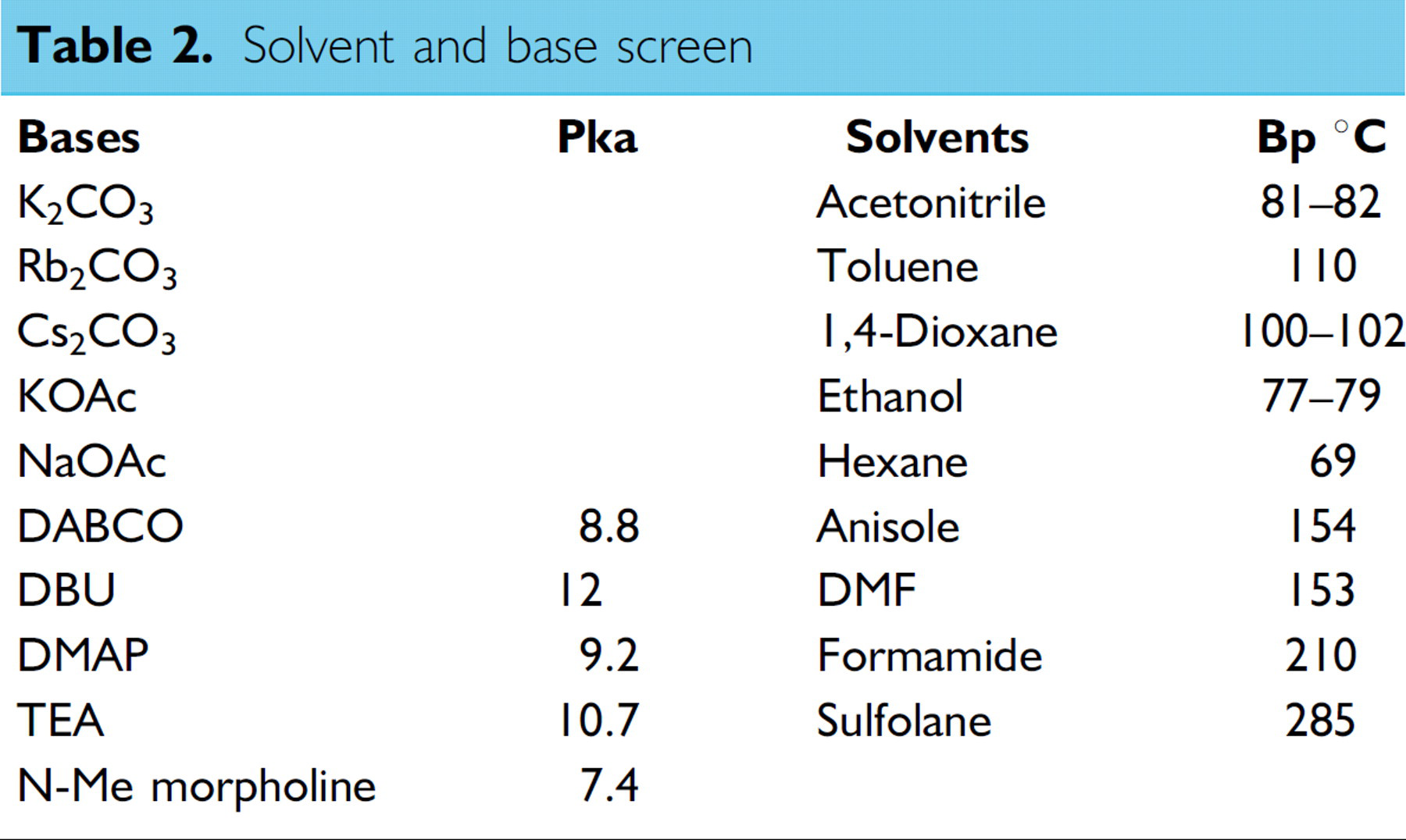
The results of the solvent and base screen were analyzed for “hits” displaying high selectivity and yield with minimum amounts of benzene by-product. Significant levels of benzene are observed under certain solvent and base conditions, the mechanism of formation of this byproduct, which formally arises from iodobenzene by replacement of iodide by hydride, is not clear. However the reversal of oxidative addition by attack of adventitious “hydride” on the arylpalladium halide, generating ArPdH, appears the most likely explanation. Interestingly, the combination of strong bases (DBU, Cs2CO3, and Rb2CO3) and protic ethanol as the solvent generate almost double the amount of benzene as the next nearest set of conditions.
The response values for each of the eight hits identified were weighted and scored, revealing two solvents providing maximum selectivity and yields with effective elimination of benzene formation (Table 3) these were prioritized by selectivity and analyzed by experimental design. Notably, the trend in the amount of benzene produced under these conditions correlates with the ability of triethylamine (Table 3, entry 2) and 1,4-dioxane (Table 3, entry 3) to produce HPdX species by dehydrogenation.
Prioritized solvent and base conditions
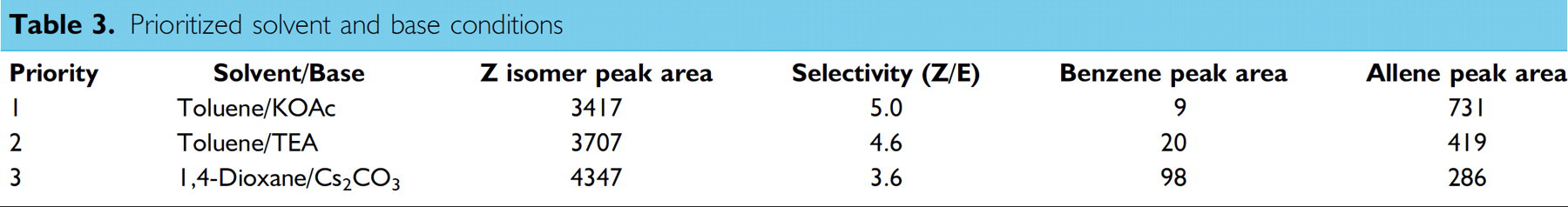
Toluene and 1,4-dioxane possess very low dipole moments (μ∼1–1.5 × 1030 Cm) and dielectric constants (ε∼2.2–2.4) suggesting low solvent polarity is important for high Z-isomer selectivity. The effect of changing solvent polarity on rotational/conformational equilibria is well documented 7,8 and it is often found that one conformer or rotamer predominates in a certain solvent, and that this is reversed on changing solvent polarity. Where conformers have different polarities, it is a general rule that the conformer of higher dipole moment is more favored in solvents of high relative permittivity. This trend was observed in our screening results using highly dipolar formamide, where a reversal in stereoselectivity was seen in six out of nine reactions, to a maximum 4:1 E/Z. This result suggests that there is a significant difference in dipole moments between the syn- and anti-allylpalladium intermediates and between the transition states leading to them. The syn π-allyl complex possesses more dipolar character which provides stabilization of this more sterically hindered species in high polarity solvents.
Design 2
A second optimization was then performed using toluene and potassium acetate, including only the significant reaction variables defined in Design 1, Table 4, and responses as before.
Design 2 factors and ranges

Temperature and time were identified as the major factors affecting selectivity and yield. Phosphine and base stoichiometry have no effect. Figure 2 was constructed using the constraints; selectivity > 6:1 Z/E, E-isomer PA < 4.0 × 106 and Z-isomer PA > 2.0 × 107 and shows that optimum conditions are found at 54 °C and 9 h. Manual and automated experiments were then performed to verify this result; analysis of the crude reaction mixture by 1H NMR and HPLC established selectivity to be 8.5:1 Z/E (∼80% d.e.). Crystallization of mixed isomers was possible after column chromatography due to higher quality product and increased conversion, and resulted in an enhanced stereo-selectivity of 21:1 Z/E. An authentic sample of the major isomer was then obtained by prep-HPLC to gain an X-ray crystal structure and nOe spectrum, which confirmed the Z-configuration of the major product.
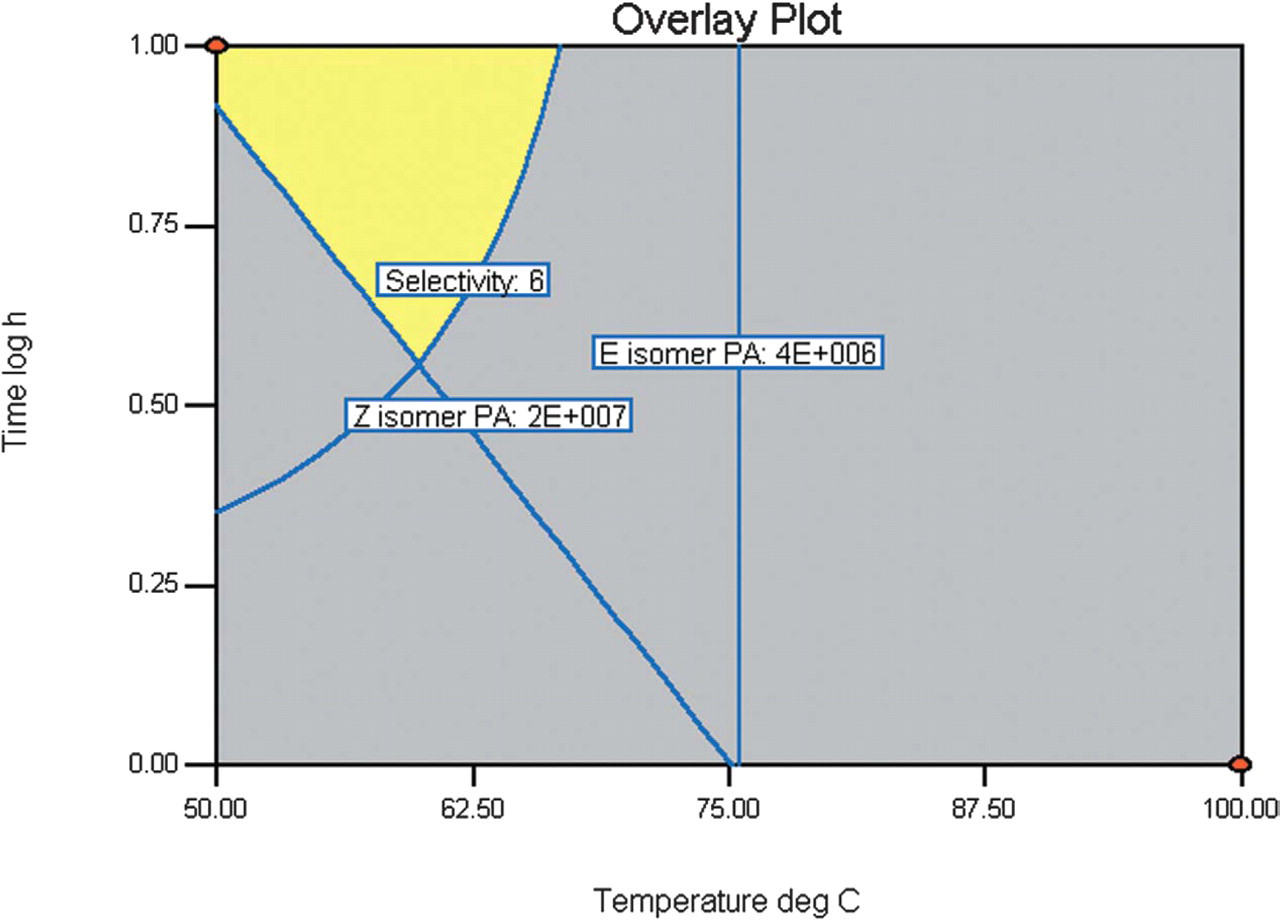
Design 2 contour overlay plot.
CONCLUSIONS
In polar solvents such as formamide, sulfolane, and acetonitrile, the syn-allylpalladium (II) species would appear to be stabilized over and above that of the anti-allylpalladium (II) species, suggesting that the syn-configuration has more dipolar character than the anti-species providing dielectric stabilization of the sterically more hindered species. It may also be possible that increased solvation of the polar theophylline substituent in dipolar solvents attenuates the steric clash in the syn-allylpalladium configuration.
The greater stability of the anti-allylpalladium complex in toluene and other less polar solvents permits the use of higher temperature, 54 °C rather than 40 °C in acetonitrile, giving increased conversion without affecting stereoselectivity. The divergence in results obtained from optimization in the two solvents also indicates the importance of solvent screening prior to designed experiments.
Acknowledgments
The authors would like to thank Dr. Sridharan and all in MIDAS for assistance, Marion Chatfield and the Strategic Technologies Lab Automation group at GSK Stevenage, and the Mary and Alice Smith Scholarship Fund for support.
