Abstract

A PC based process was adopted by the medical center to support decentralized testing in the Departments of Pediatrics and Emergency Medicine. This process automates the creation of test orders and accession numbers, enables the central laboratory to remotely review testing performed at the point-of-service, and captures all data associated with the test and process. The additional goals and objectives of this performance enhancement included:
Introduce
Improve patient care and reduce the number of arterial and or venipunctures by combining the measurement of pH and arterial blood gases with the HCFA basic metabolic panel (Chem 7), Ca++, and lactate.
Eliminate the respiratory lab and process these samples (200 per day) directly at point-of-service locations.
Increase the number of critical care test available at the Point-of-Service
Reduction in cost of testing for both arterial blood gases (ABG's), electrolytes, and related critical care metabolites
The systems chosen for this performance enhancement were the STAT Ultra M critical care analyzer (

STAT Ultra M Critical Care Analyzer (Nova Biomedical, Waltham, MA)
REMOTE CONNECTIVITY OF POINT-OF-SERVICE TESTING
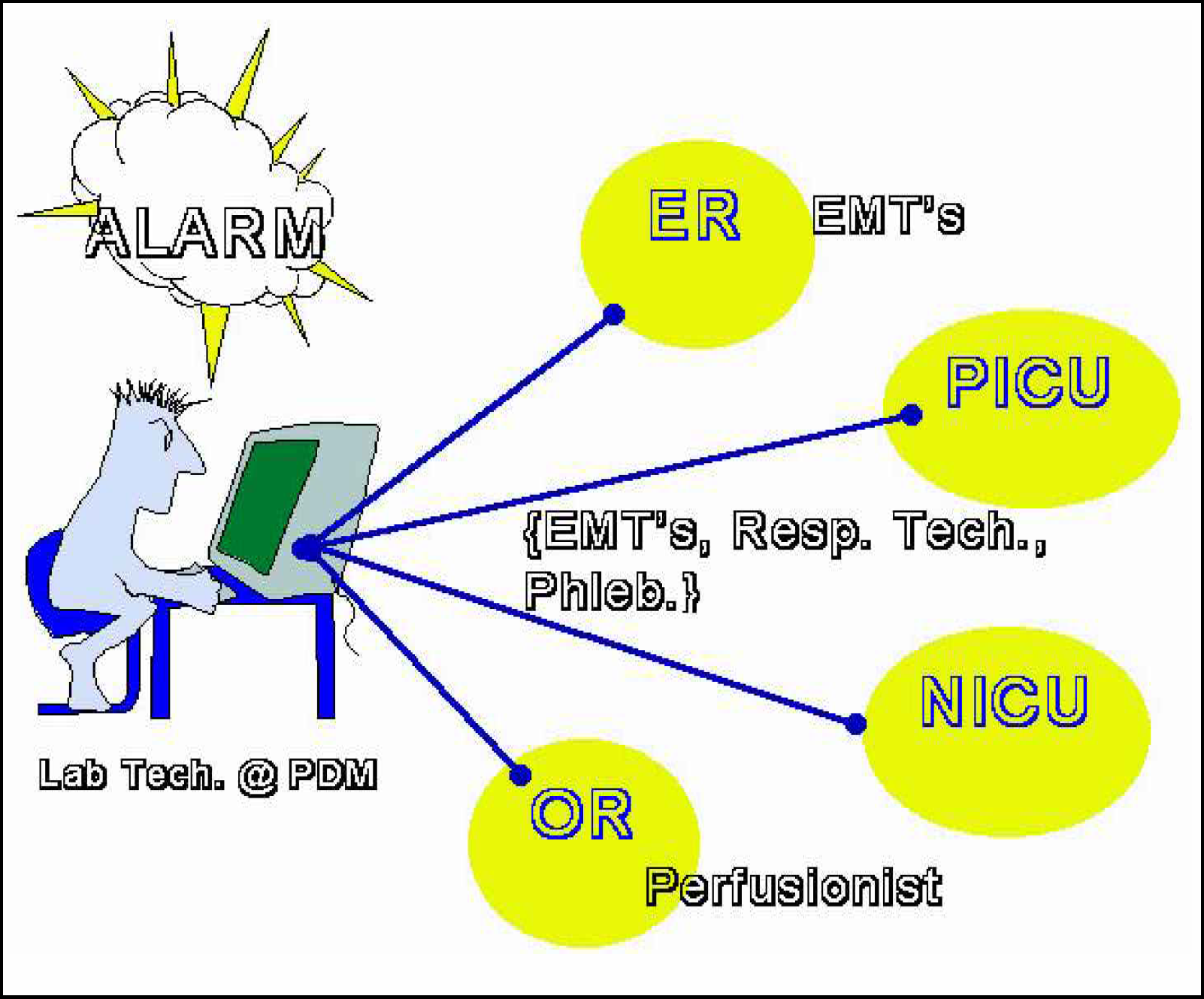
What follows is a description of the workflow between the remote sites and the Central Laboratory. STAT Ultra M analyzers were placed in the pediatric and neonatal intensive care units, the pediatric operating rooms, and in the emergency department. Another STAT Ultra M analyzer is maintained on a mobile stat cart as a backup for any of the point-of-service locations. Samples are introduced at the point-of-service locations by the clinical staff (nurses, EMT's, respiratory therapists, etc.). Total monthly sample volume for the Stat Profile analyzers is approximately ∼7,000. Each STAT Ultra M analyzer is interfaced to the PDM located in the central laboratory. The PDM is directly interfaced to the LIS. The LIS then exchanges data with the hospital information system (HIS).
POST WORKFLOW USING THE PDM
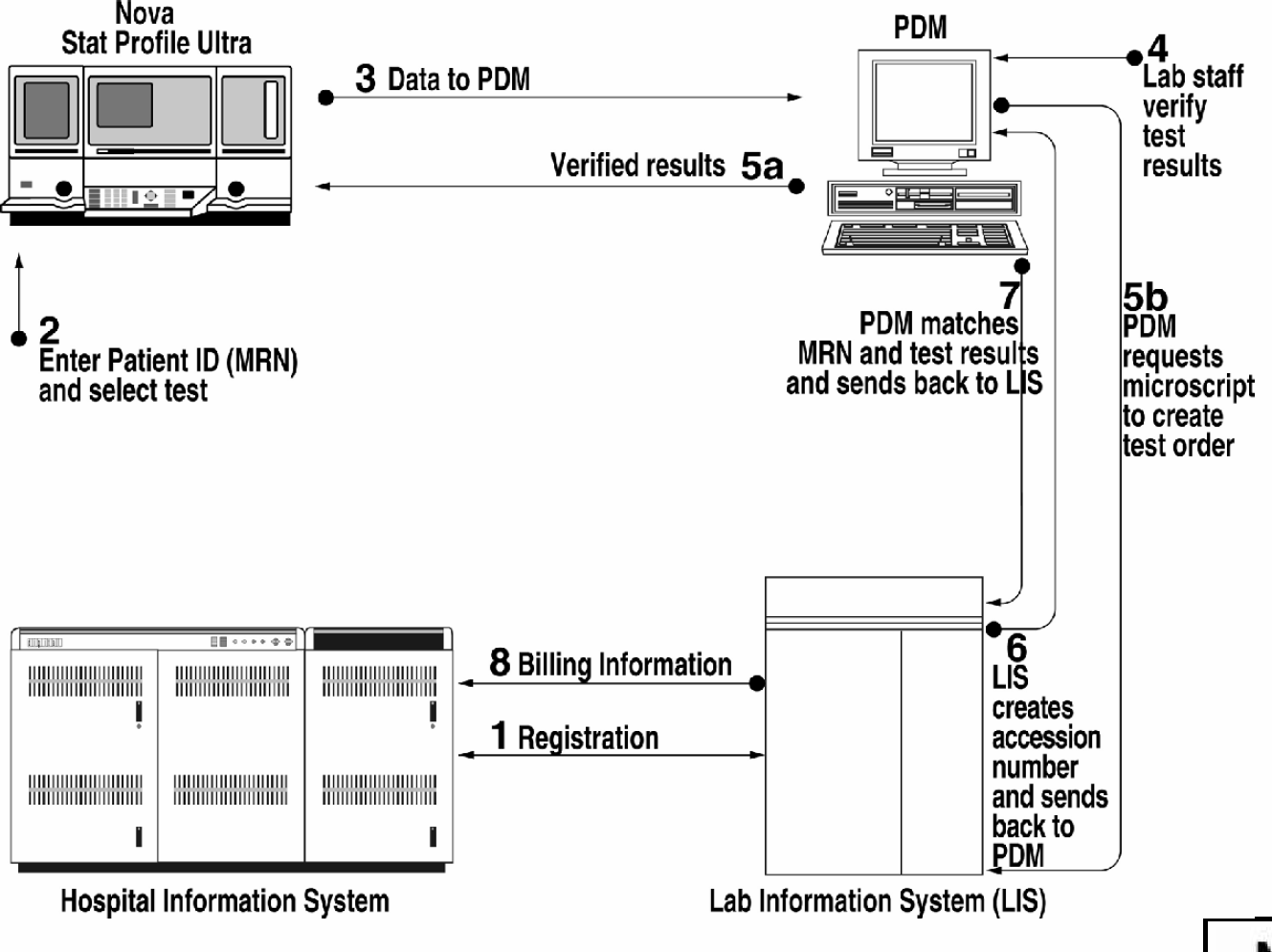
Sample introduced at point-of-service analyzer by clinical staff. Patient identification (medical record number — MRN) and test code entered. “Analyze” button pressed.
Test results, MRN, and test code transmitted to PDM system in central laboratory. PDM provides audible signal to central laboratory staff that test results have been received.
PDM initiates a test order and requests an accession number from the LIS. The LIS provides an accession number for the test order created by the PDM. This process eliminates the need for a person in the laboratory who would manually obtain this information from the LIS and create a lab-initiated test order. The accession number is returned to the PDM system, which is holding the test results; the accession number is matched with the test results.
Test results are reviewed on the PDM and verified by laboratory personnel. (Step 3 is completed within seconds, before laboratory personnel respond to the audible signal announcing receipt of test results from the point-of-service analyzer.)
Verified results are released to the analyzer at the point-of-service for use by clinical staff.
Verified results are released to the LIS.
Data package transmitted from LIS to HIS.
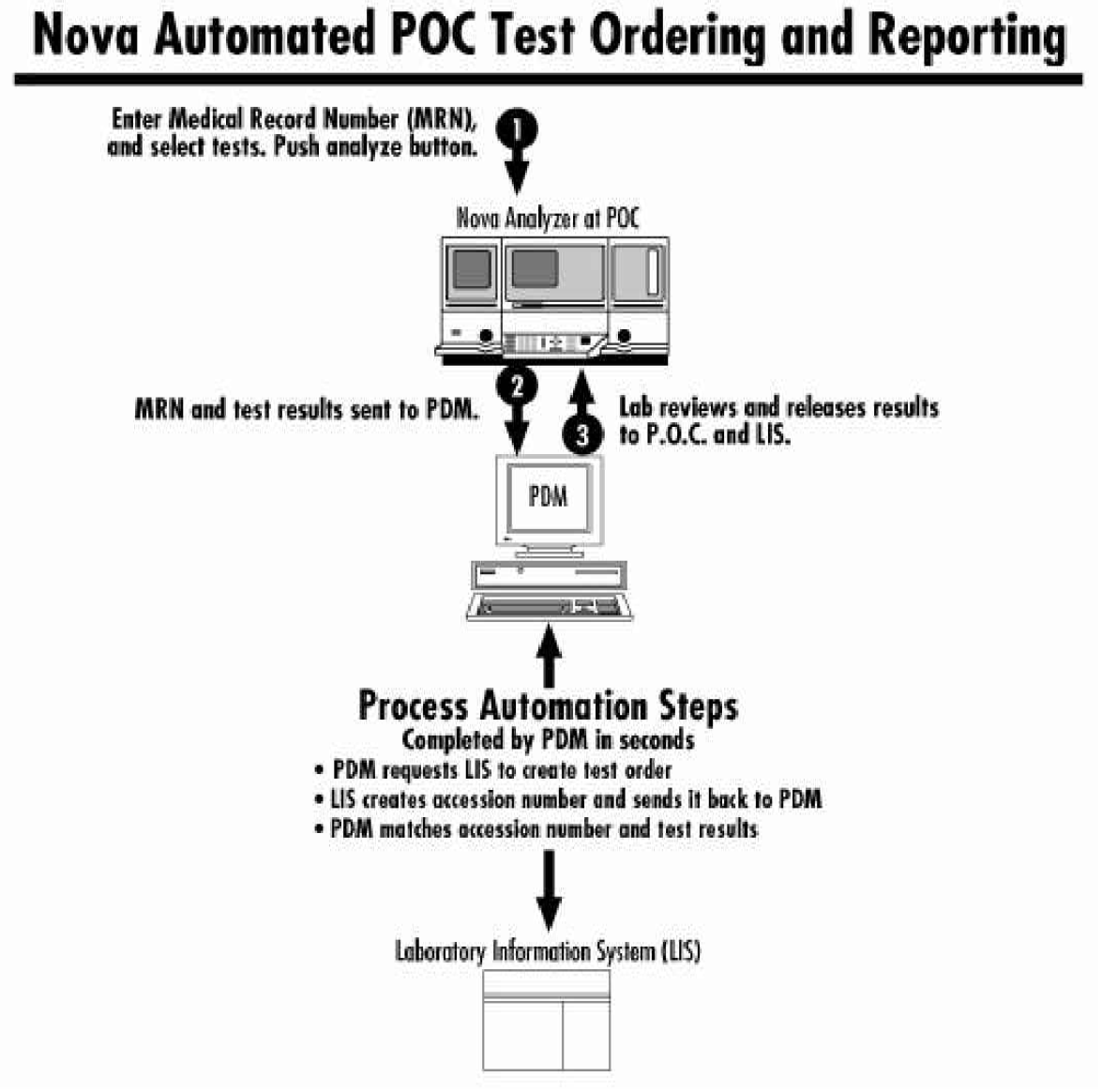
Automated process as seen by clinical and laboratory staff.
CLINICAL STAFF
Introduce sample, enter MRN and test code
Start the analysis cycle by pressing the analyze button
Receive verified test results in approximately 3 minutes
LABORATORY STAFF
Respond to audible signal from PDM system
Review test results
Press “Release” button to release results to point-of-service analyzer and LIS
OPERATIONAL RESULTS AND SUMMARY OF BENEFITS
The results of this initiative are still being evaluated. Prior to implementation of point-of-service testing, the turnaround time for STAT samples processed in the Central laboratory was ∼60 minutes. With point-of-service testing, the turnaround time has been reduced to 5–8 minutes, depending on the service, for the 7,000 samples processed per month at the four remote testing locations. Consequently, patient care has been enhanced through the reduced number of specimen collections and improved TAT's, in addition to the immediate availability of special critical care analytes, such as, Ca++, Mg++, and LACTATE. For example, immediacy of testing, and availability of the above mentioned special critical care analytes have supported pediatric thoracic surgeries while the patient is on a cardiac by-pass perfusion system. It is expected that mortality rates as a consequence should decrease. Medical necessity and clinical utilization will be evaluated in 1999, along with monitoring length of stay impact. Preliminary data indicate an improvement in both areas. Service perception is at an all time high for PICU, NICU, DEM, and Pediatric OR's. It appears that cost reductions should exceed $175,000 for the combined testing which requires less labor. Those samples previously processed by the respiratory department in a dedicated respiratory lab (∼200 samples/day) have been converted to point-of-service testing without the need for additional employees.
Additionally, workflow automation enabled by the PDM system eliminated pre-analytical delays and labor requirements resulting from manual creation of test orders and accession numbers. Automated transfer of verified test results from the PDM system to the LIS leads to further cost savings. This PC based process eliminated the need for manual entry of test results and obviates the need for a conventional, high cost LIS interface. With a simple user interface at the point-of-service locations and remote review capabilities, this system has eliminated the need for laboratory personnel at the remote testing locations without jeopardizing quality assessment and regulatory compliance.