Abstract

Introduction
The greatest potential of proteomics is discovery of disease-related proteins
For resolution, polyacrylamide gels remain the tool of choice
The rapid rise in MALDI-MS throughput demands faster protein excision than can be achieved with current spot-picking instruments
To meet this demand, Genetix, in conjunction with the Max Planck Institute in Berlin (Nordhoff et al., 2001), have developed GelPix [Figure 1]
Based on Genetix' high-performance genomics robotic platforms, GelPix is the fastest and most robust protein excision system
GelPix now excises from both 1D and 2D gels, and dispenses into a wide range of microplates.

The Genetix GelPix.
Specifications
Dramatically faster excision due to proprietary multi-channel excision head [Figure 2]
Rapid imaging with cooled-CCD camera
Precise 2D spot and 1D band excision
Consistently high spot retrieval rate
Minimal set-up time
Optimised for Coomassie, silver and Sypro Ruby™
Unique non-UV fluorescence imaging
Accepts large and small unbacked and backed gels
Ultra-clean pin wash procedures
Durable, long-life excision pins (1.4mm and 2mm)
Easy-load gel holder
Collection to wide range of microplates or filter plates
Easy-to-use software with sample tracking
Imports and matches previously analysed gel images
Fully enclosed HEPA-filtered environment
Precise humidity control
Wash and waste level sensors

The unique 8-channel excision head.
The GelPix Protein Excision Process
LOADING GELS
Removable gel holders support unbacked and plastic-backed gels, and slot easily and firmly onto the bed of the GelPix. These allow gels to be imaged, analysed and excised without moving the gel.
IMAGING
Imaging is via the on-board high dynamic range CCD camera. Coomassie- and silver-stained gels are visualized with white light [Figure 3]. Sypro Ruby™ gel images are exposed using a unique non-UV light source to give cleaner background than when captured using UV light or laser scanners [Figure 4].
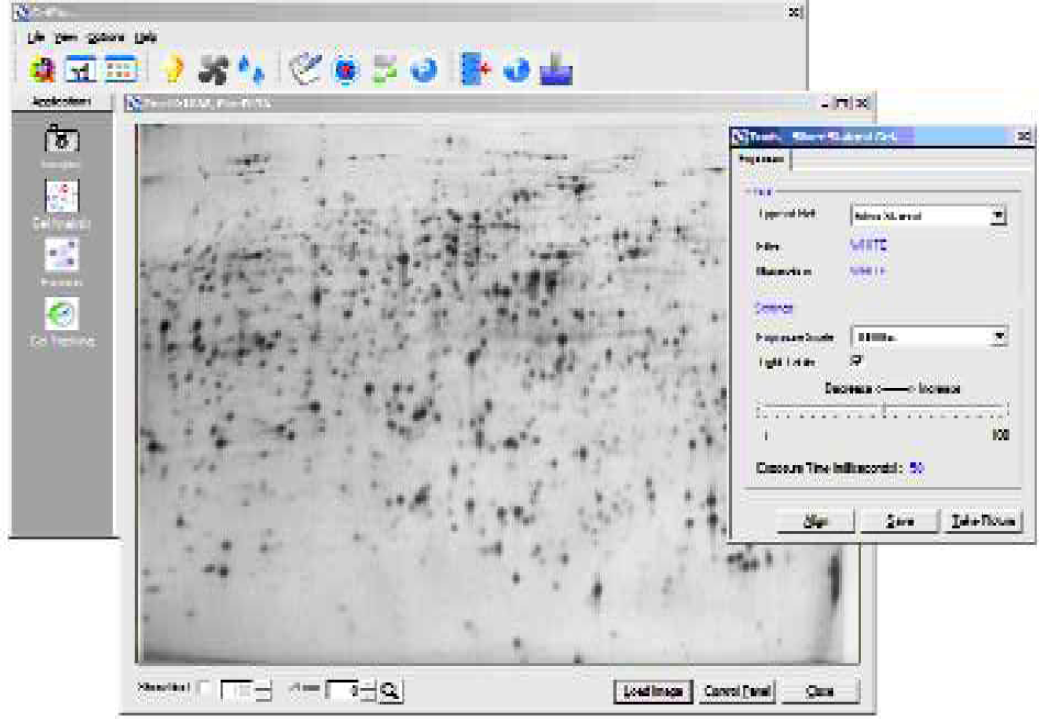
Gel imaging is quickly and conveniently achieved with the on-board cooled CCD camera.
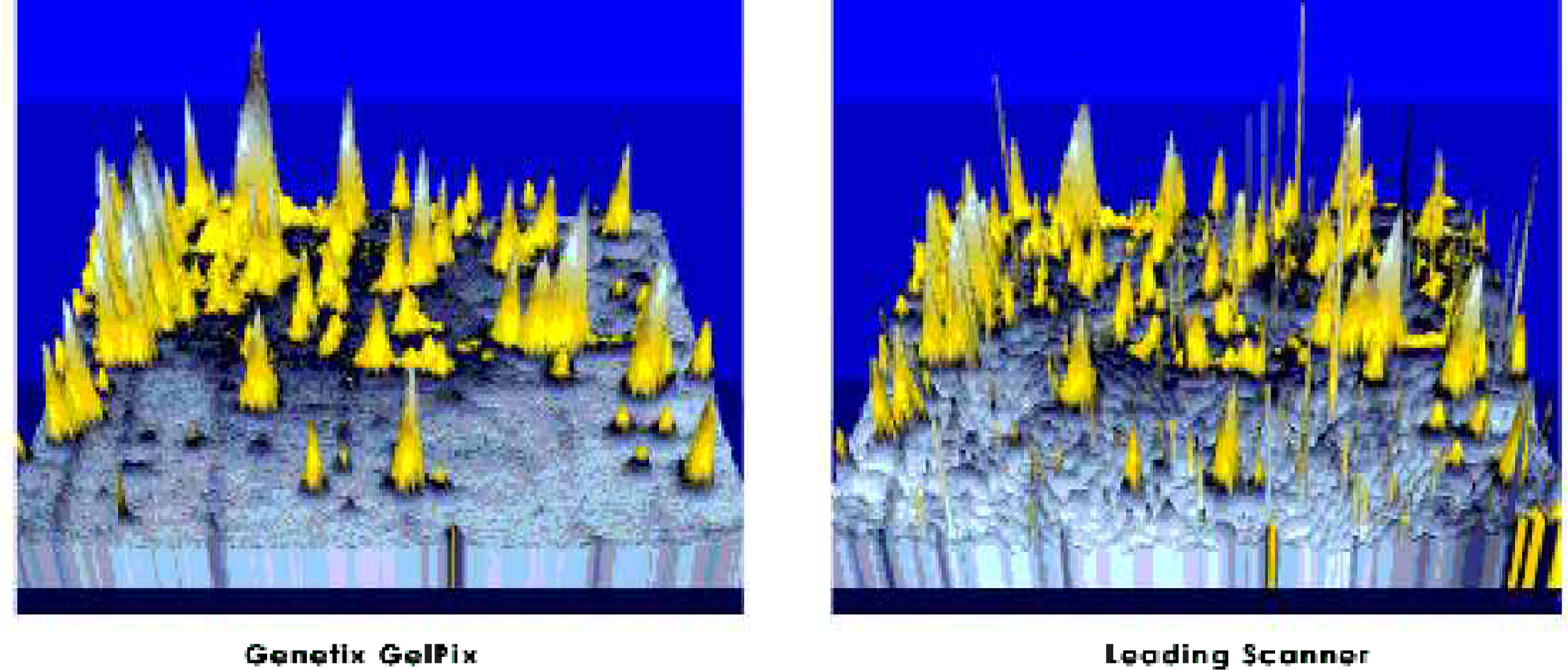
The unique fluorescent imaging system gives a clean background and prevents illumination of impurities seen with laser scanners.
A Sypro Ruby™ stained gel of whole E. coli cell extract was imaged on a leading brand of laser scanner and then on GelPix, at comparable resolutions. Instrument settings for the laser scanner were 532nm for excitation and 610nm for emission; exposure time 5 mins. GelPix imaging was completed in 1 min 40 seconds at F4. Pseudo-3D images were generated using Proteomweaver (Definiens, GmbH, Germany) without median filtering.
GEL ANALYSIS
GelPix uses integrated image analysis software to generate the excision pick list. For 1D analysis, protein bands are detected by Phoretix 1D Advanced (Nonlinear Dynamics, UK). For 2D analysis, GelPix uses Phoretix 2D Advanced, Proteomweaver (Definiens GmbH, Germany), or Compugen software. Images of gels captured externally, e.g. using a scanner, can be matched to the GelPix image, allowing spot coordinates on the external image to be transformed into a pick list.
EXCISION
Excision is the process of collecting the desired gel pieces. A onetime calibration of GelPix is sufficient to ensure precise spot excision every time. Pre-defined settings ensure optimal excision from all gels up to 2mm deep (up to 1440 excisions). Each of the 8 excision pins is fired independently to collect its assigned protein spot [Figure 5]. After dispensing, all pins are washed simultaneously, greatly reducing the average cycle time. Each 96-well microplate is completed in 10 minutes.

Each excision pin is fired independently to collect its assigned protein spot.
GEL TRACKING
Gel Tracking ensures easy cross-referencing between microplate spot location and the original gel image. The data generated by GelPix is exportable in Excel format for downstream processing of excised spots.
GelPix Validation Testing
PRECISION
Proteins were excised from a backed 0.5mm gel. The excision process was then repeated twice. Figure 6 shows that GelPix repeatedly excised at precisely the same locations.
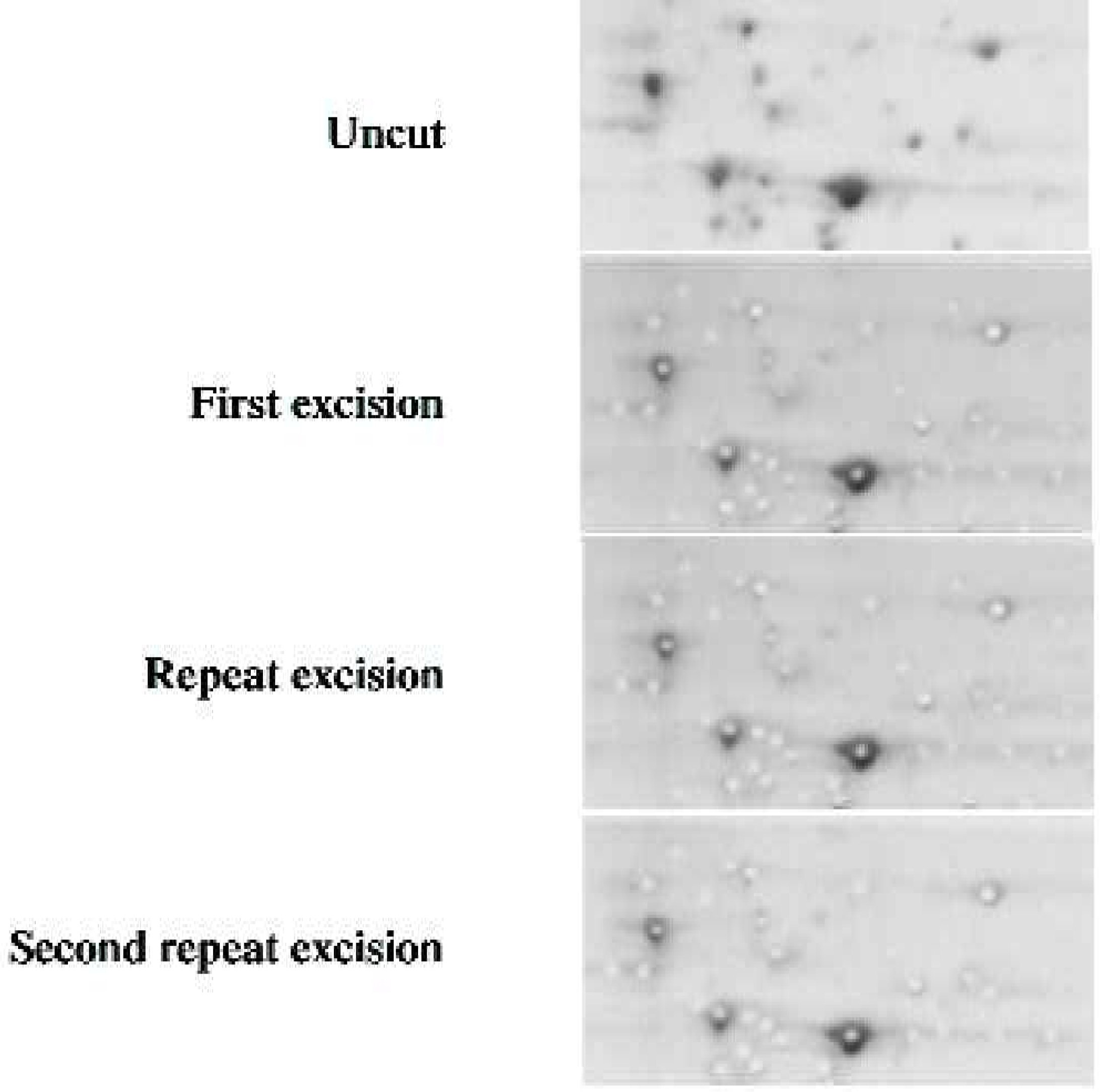
Repeated excision at the same coordinates shows the high precision of spot cutting achieved with GelPix.
EXCISION SUCCESS RATE
Using 1.4mm pins, 1440 gel spots (15 plates) were picked from an Amersham DALT precast gel (plastic-backed; 1mm; 12.5%). A total of 1431 spots were recovered, i.e. an excision success rate of 99.4%.
PROTEIN CARRYOVER
A major concern with excision robots is that each pin is used to excise many spots. We investigated carryover by GelPix after excision from intense Coomassie spots. Data was collected on a Micromass LCT ESI-TOF mass spectrometer (courtesy of Dr. Paul Skipp, Southampton, UK). Figure 7 shows that there was no detectable carry-over.
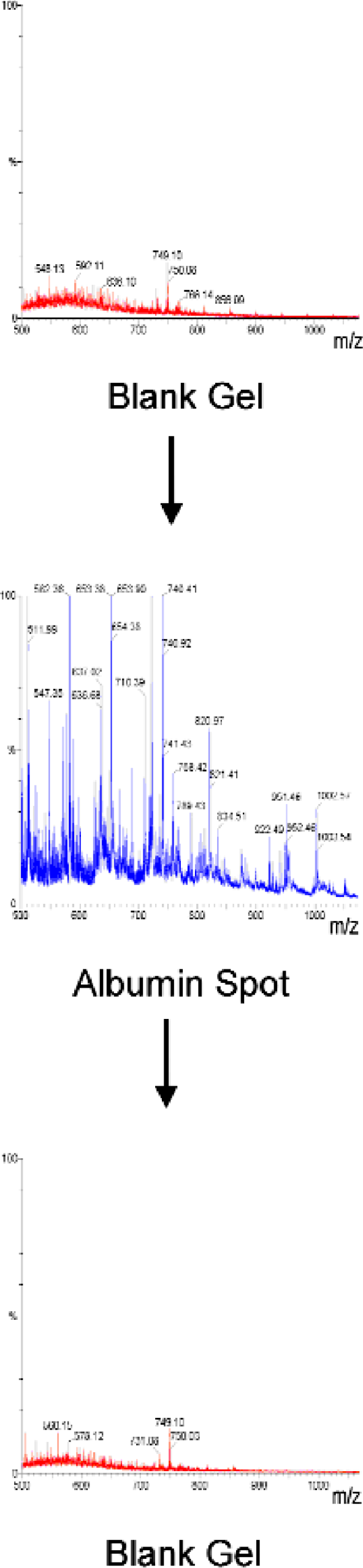
GelPix has no measurable protein carryover as determined by mass spectrometry.
Conclusion
GelPix is the only gel-excision robot with a multi-channel picking-head that can image, analyze and excise proteins from 1D and 2D gels cleanly and consistently without compromising accuracy. The excision rate of >600 proteins per hour will enable MALDI tandem TOF mass spectrometry to be used to its full potential.
