Abstract

Introduction
In 1995, the standard ISO/TC212 which applied to the Clinical Laboratory and External Laboratory System was established by the National Committee on Clinical Laboratory Standards (NCCLS). Later in 1996, with this as a catalyst for future standards development, the NCCLS set to work developing a Laboratory Automation Standard in order to increase the efficiency and interconnectivity of laboratory automation systems and to facilitate their engineering. The NCCLS also proceeded to work with other standards developers such as the American Society of Testing and Materials (ASTM), International Electrical Engineers (IEE), and Hospital Level 7 (HL7). These committees worked on creating laboratory automation standards from the period extending from 1997 until the end of 1999. The following standards have been completed by the NCCLS:
AUTO1-P: Standard of Specimen container / Specimen Career design and production
AUTO2-P: Barcode method for Specimen container discrimination
AUTO3-P: Communication standard of electronic date exchange of automation system
AUTO4-P: Standard for status information; system applying condition, special quality, and information element, etc.
AUTO5-P: Guide to electronic mechanical interface These standards have been adopted by other organizations such as the Japanese Clinical Chemistry Laboratory Society (JCCLS) and the International Federation for Clinical Chemistry (IFCC). AUTO3-P will also use the standard HL7 version 2.4 as the current HL7 standard.
OUTLINE OF HL7
HL7 standard is the electronic data exchange standard between used in medical facilities. It's called the Health Level Seven Standard because it is a standard for the medical industry.
In March 1987, HL7 was established by Pennsylvania University Hospital in order to facilitate easy connectivity between a hospital information system (HIS) developed in house, and the hospitals medical equipment. They submitted the original draft of version 1.0 on October 1987, and set up the interface structure for their admission, discharge, and transfer (ADT) system, order entry, indicate query. The final draft version was published as version 2.0 in September of 1988. In 2000, Version 2.4 was published after revisions concerning Laboratory Automation, Network Management, and Addition Personnel Management were completed. Version 3.0 is also going to be revised to be a standard that accepts object direction modeling and message design.
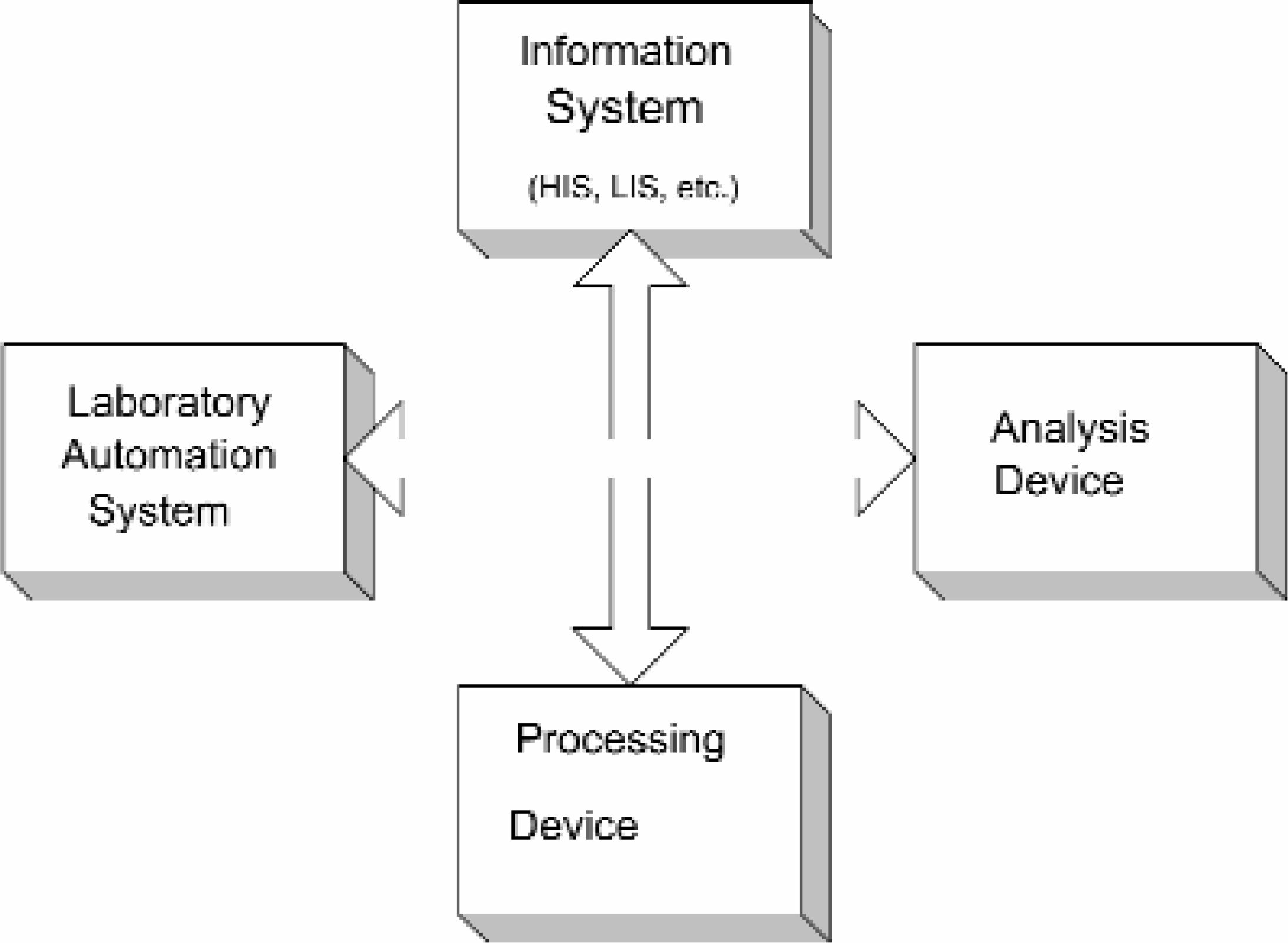
Elements of a Laboratory Automation System.
HL7 defines the definition of exchange data, exchange timing, application error communication. HL7 includes: patient reception, admissions and discharge information, change department and building, inquiry, appointment management, order entry, test result notice, accounting, master file management, consultation management, patient introduction, nursing recording, and medical information systems within cover field.
OUTLINE OF LABORATORY AUTOMATION SYSTEM COMMUNICATION STANDARD IN HL7 VERSION 2.4
Information System:
LIS: management of data about patient, test order, specimen
HIS: management of accounting, materials, employees, patient information
Laboratory Automation System (LAS): Part of the new equipment control center as an electronic interface
Analysis device: Elements for laboratory tests
Processing device: Elements for working except analysis to specimen, container, career device, etc.
FUNCTION MODEL
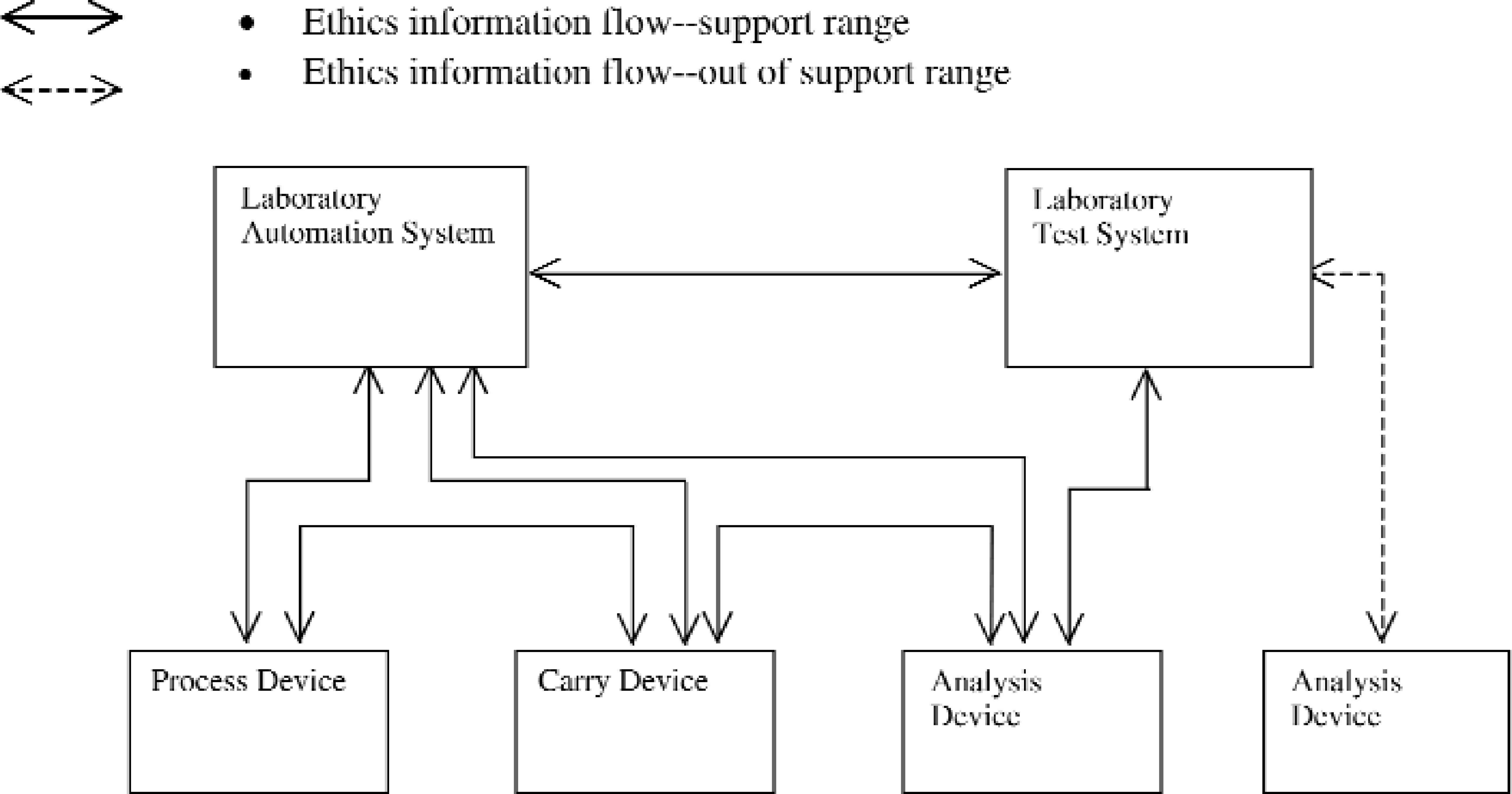
This model depicts the structural elements of the laboratory automation system (LAS) (Figure 3–2).
Analysis devices can keep patient data, precisely manage data, and generate reports.
TRIGGER EVENT AND MESSAGE DEFINITION
Test order and test result message revising for laboratory automation systems. It is possible to send details and special qualities of Test order to Test order ORM messages and Test result ORU messages. Therefore, it is possible to add SAC and OBX segments before the ORC segment.
ESU/ESR: Equipment State Update / Request (Event U01 & U02)
This is the message for device or condition of equipment information. State Update can voluntarily transmit message as answer of “Specimen State Request” Trigger.
SSU/SSR: Specimen State Update / Request (Event U03 & U04).
This is the message for transmission/demand about specimen location and its information.
INU/ INR: Automated Equipment Inventory Update/Request (Event U05 & U06)
This is the message for transmission/demand about information of stock items (reagent, etc.).
EAC/ERA: Automated Equipment Command/Answer (Event U07 & U08)
This is the message for transmission/demand about support to device.
EAN/ACK: Automated Equipment Notice (Event U09)
This is the message for transmission to device notice.
TCU/ TCR: Automated Equipment Test code Settings Update/Request (Event U10 & U11)
This is the message for transmission/demand about information of measure code and parameter. Transmitted parameter set is automatically rearranged into the same parameter set as when receiving messages. You cannot choose “add” or “delete” within the new parameter set.
LSU/ LSR: Automated Equipment Log Service Update/Request (Event U12 & U13)
This is the message for transmission/demand about one side of log service or both.
SEGMENT DEFINITION
Additional segment in HL7 for Laboratory Automation System:
EQU: Equipment Detail Segment
This segment is necessary data for Equipment discrimination and maintaining of equipment state.
ISD: Interaction State Detail Segment
This segment contains data for a specific process state concerning specific equipment.
SAC: Specimen Container Detail Segment
This segment is data that maintains using a specimen con tainer in the system.
Example of Message
INV: Inventory Detail Segment
This segment is data for keeping record of stock of test materials (such as reagent, tip, and liquid waste).
ECD: Equipment Command Segment
This segment is data for notifying occurrences when receiving messages of organization elements.
ECR: Equipment Command Reply Segment
This segment is answering data just before command in the side of receiving message.
NDS: Equipment Notify Detail Segment
This segment is data for keeping inspection records and events notifications.
CNS: Clear Notify Segment
This segment is data for clearing notice in relation to receiving instruments.
TCC: Measurement Code Create Segment
This segment is data that is used throughout the automation system in relation to the Measurement Code for keeping or sending messages.
TCD: Measurement Code Detail Segment
This segment is data for operating, calculating, and assessing of the situation by the Clinical Test Automation System.
SID: Test Material Discrimination Segment
This segment includes data that is used test materials (such as Reagent) in an analysis results process for confirming, and it is used with the TCD segment. This segment can use reiteration with each of the test materials.
EQP: Equipment Log/Service Segment
This segment is data for keeping appropriate audit records that happen with events by specific Equipment.
EXAMPLE OF MESSAGE
This is an example of a message that is used to test order messages in the Clinical Test Automation System, and also this is an example of an OML message of plural specimen and plural test order.
FUTURE DIRECTIONS
Version 2.4 was published in 2000. In the future, HL7 is going to define conformance statements for Version 2.5, and plans to extend to do more high-grade inquiries. For example, there are queries for order information and transfers of additional information for automated processing. There is, however, no specific query for laboratory order information. Instead, the order information should be downloaded to the LAS either unsolicited or after an implicit trigger such as sample status update. Transfer of information for automated processing requires having additional instruments for the performing of automated processing based on automatic validation, such as the expected date of birth (Delivery Date), menstrual status, and medication history. Version 2.5 should consider using OBX segments and LOINC codes. Thus, our recommendation is to use the conformance statement such as to query the observation order of the patient for a specified container ID and SAC information; query the observation result for specified observation order within a date; and query the observation result of the patient for a specified container and other SAC information.
We hope that this standard will contribute to the development of true plug and play capabilities for laboratory automation systems of the future.
