Abstract
Automation requires significant capital investment and for contract laboratories this funding may be difficult to justify. However, with increasing commitment to the automation of dosage form analysis by pharmaceutical companies and the adoption of automation earlier in the development process, outsourcing of automated methods will be an important requirement in the future. Contract laboratories that do not have automation equipment will not be able to compete as effectively for business as those that have this capability. The benefits to the customer of outsourcing automated methods will be discussed together with the potential problems for the contract laboratory in forming a strategy to meet this need. The adoption of new technology in other development functions (e.g., bioanalysis), by pharmaceutical companies and contract facilities has been very successful. The learnings from this situation will also be briefly outlined to provide some perspective around the implementation of automated methods.
This presentation was given at the 2000 International Symposium for Laboratory Automation and Robotics (ISLAR) held in Boston, MA, October 15-18, 2000. The full manuscript is available on CD-Rom and can be acquired by contacting Christine O'Neil, 508-497-2224; email
INTRODUCTION
The ultimate goal of outsourcing in drug development is to reduce the time and cost of bringing a new drug to market, to focus the company's efforts on exploiting its own proprietary technology, and to make the most efficient use of the company's resources and personnel. CROs revenues increased by about 120% from 1995–1997, whereas pharmaceutical companies R&D budget increased on average by only 14% which illustrates the growing trend towards outsourcing. This is supported by a recent report 1 that CROs accounted for 26.8% of a development budget of $19.9 billion. The same report forecasts that CRO penetration would, by 2001, increase to 34% of a budget of $26.4 billion. For CROs the continuing merger activity by pharmaceutical companies will provide new challenges and opportunities. The large pharmaceutical companies will continue to take advantage of the reorganisation that inevitably follows a merger and to rethink their approach to product development.
AUTOMATION IN SMITHKLINE BEECHAM
Pharmaceutical Technologies are responsible for the formulation and analytical development, clinical trials supply and technology transfer of new products to manufacturing facilities. The department is organised on a team-based structure in which the formulation and analytical development is performed by dedicated “Product” teams for a development candidate. More generalised activities such as microbiological and raw materials testing are the responsibility of “Support” teams (see Figure 1). Implementing robotics/automation is the mandate of the Automation Development (AD) “Support” team.
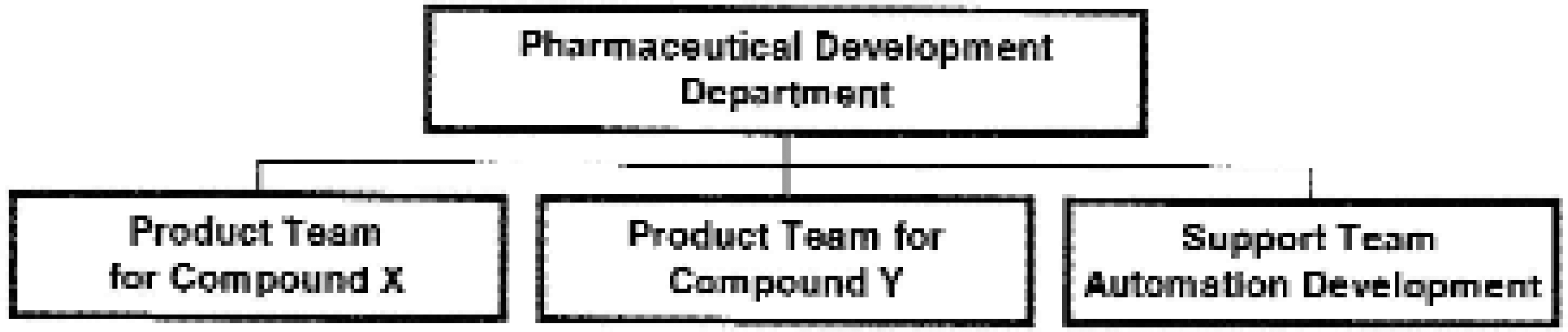
Outline of organisational structure
This central team works with the Product teams to implement robotic analytical methods employing the general process shown in Figure 2 using the following equipment: Zymark TPW™ IIs, Zymark MultiDose™ (USP2), Zymark MultiDose Plus™ (USP1), and Automated Dissolution Modules™ from Source for Automation (USP 1 and 2).
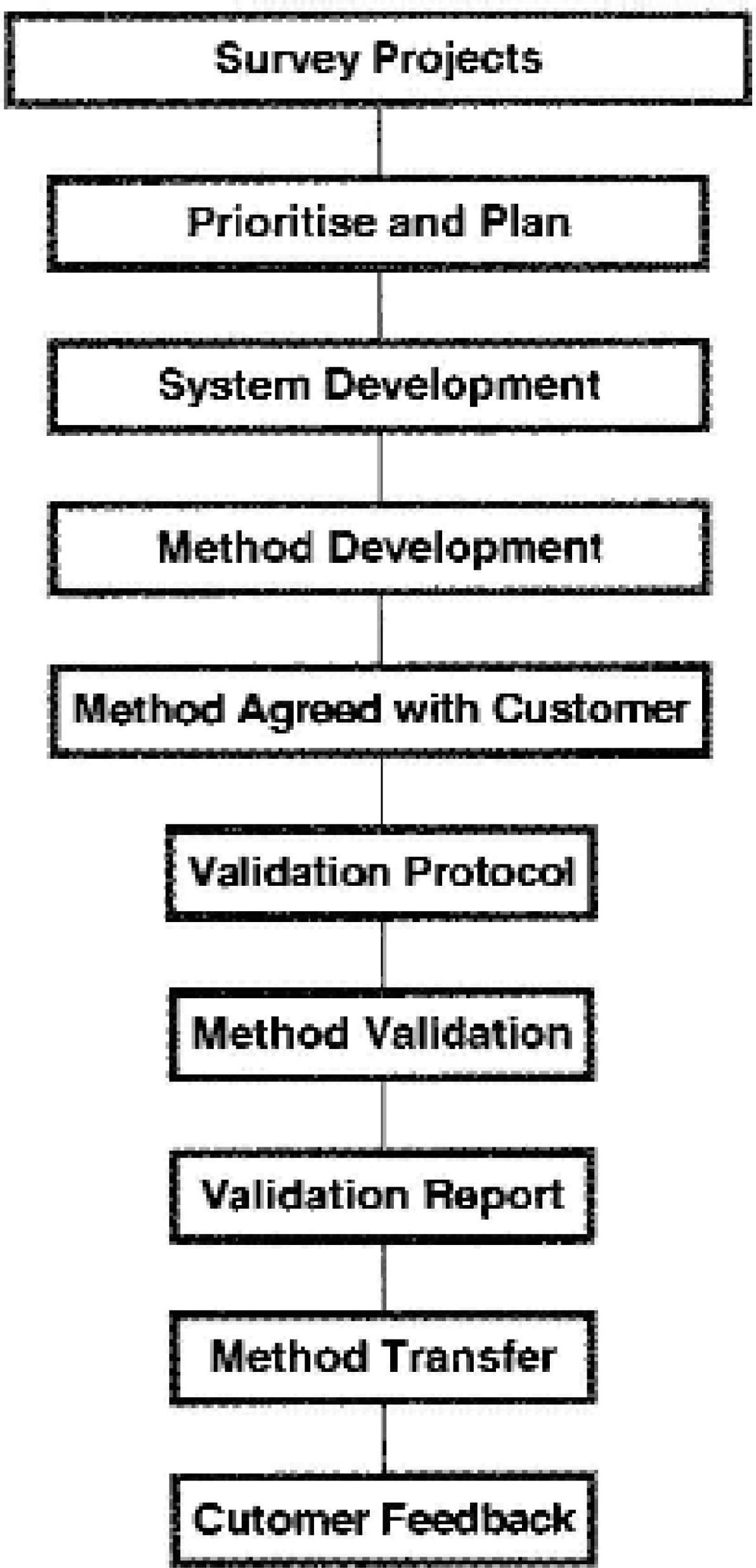
Implementation Process for Robotic Automation
Some of SB's Production sites have invested heavily in automation mirroring the equipment in Research and Development and are now requesting that analytical methods (i.e., assay, content uniformity and dissolution), transferred are automated. Estimation has also been made of the cost benefit to production sites of the provision of ready validated robotic methods (see Table 1). In the future it is planned that implementation of robotic methods will be rapid enough to make it cost effective to automate projects from Phase 1.
Relative cost benefit of manual and automated methods.

Note: Based on two analysts@£30 per hour each.
DISCUSSION
OUTSOURCING OF AUTOMATED METHODS
Product development teams have a number of options to handle excess analytical workload including:
Automation of the key tests (e.g., dissolution and assay/content uniformity).
Transferring extra in-house resources from other Product teams.
Hiring temporary contract staff.
Outsourcing work to contract research organisations (CRO).
In general the pharmaceutical development teams currently employ a mixture of all four options to meet the needs of our projects. The choice of approach is dependent on a number of considerations including:
Timeframes to complete the work
Type of work
comparator versus proprietary compound
routine, labour intensive, demanding
volume of work
technique available in-house or not
data for regulatory submission or not
Capability of CRO
Cost of using a CRO versus the other options
Outsourcing of dosage form analysis in SmithKline Beecham, Pharmaceutical Technologies is the responsibility of the Product Development teams. In the UK CROs are typically employed to perform work on comparators both for method development and stability testing. The analytical work on proprietary compounds is generally kept in-house even for large-scale stability studies. The rationale for this approach seems to be for the following reasons:
SmithKline Beecham is not an expert in the analysis of comparator compounds and does not see this as a core scientific requirement. This type of work is an ideal package for outsourcing.
The development of proprietary compounds is obviously a core business need for pharmaceutical companies even though there is a lot of routine stability testing performed particularly for qualifying batches. In some instances stability issues have become apparent at the latter stages of development which are best solved in-house by the Product development team. This problem has become more evident as product development times have decreased.
Over the past three years automated methods have been employed in-house to deal successfully with the high resource demands on stability testing.
BENEFITS OF OUTSOURCING
The potential benefits to CROs if they do invest in automated systems could be significant.
Technology transfer of the automated method from the pharmaceutical company to the CRO should be easier (i.e., a method on a disc). This assumes the CRO has the same type of automation equipment as their customer.
Peaks and troughs in workload can be met by the use of automated systems, thus offering great flexibility and capacity without the need to hire extra staff. Typically an automated system like the Zymark MultiDose™ can perform up to 12 dissolution runs in 24 hours for immediate release dosage forms. However, dedicated trained staff are needed to successfully run automated equipment.
Although the capital investment in automated systems for dissolution and assay is high, the cost per sample should be significantly decreased provided the equipment is well utilised. Cost savings obtained within pharmaceutical companies have been highlighted previously 3 .
There is the opportunity for CROs to offer a method development and validation service for automated methods provided their equipment matches that of their customer. This service could apply not only to new development candidates but also to old methods which pharmaceutical companies do not have the resources to update.
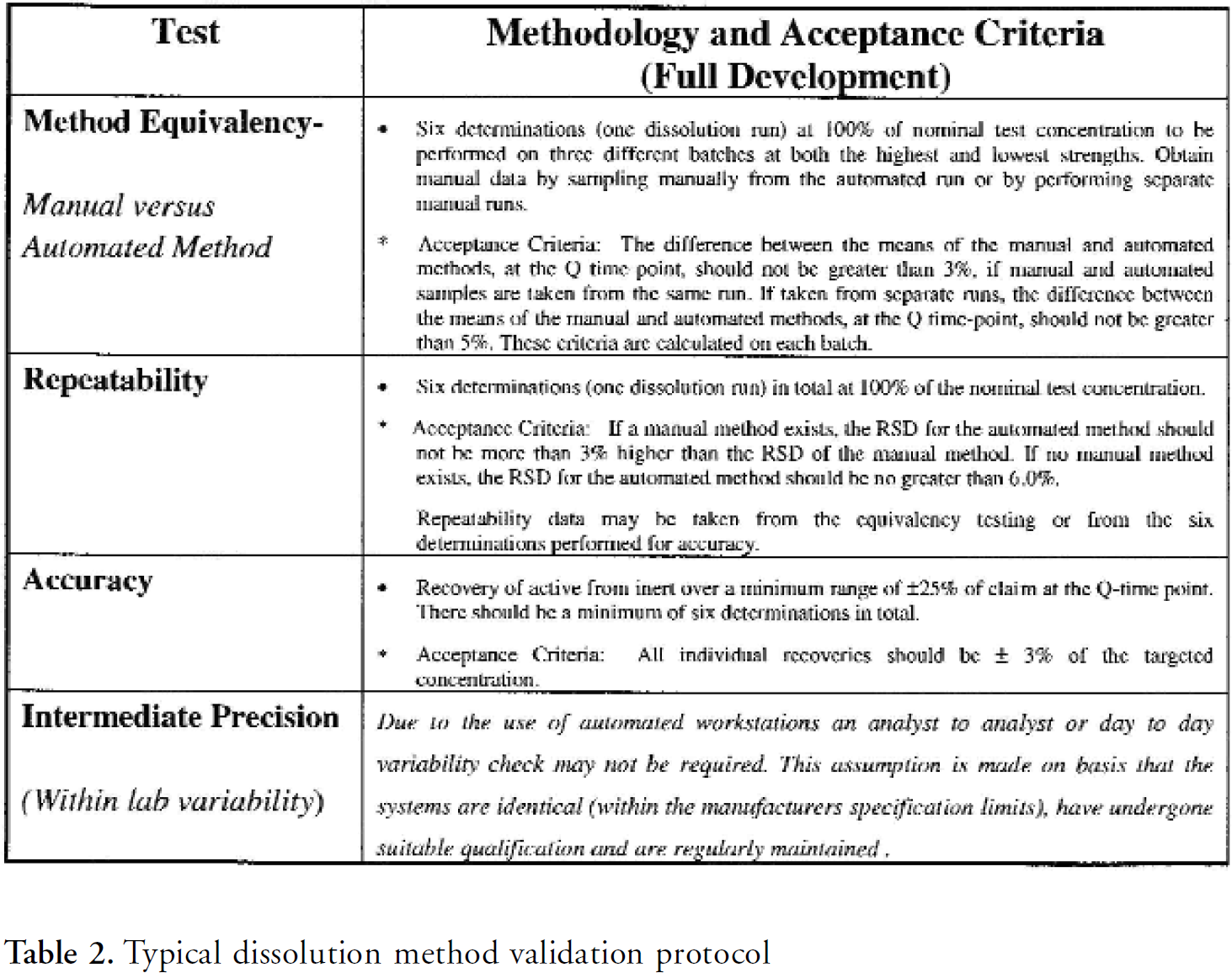
The effort needed to develop and validate automated methods, particularly for dissolution, is relatively small and should make it possible for CROs to use automated methods for all products that require significant numbers of dissolution tests. Most automated dissolution systems mimic the manual process in the way the test is performed which means that there should a high chance of performing the validation successfully. It should also be noted that validation guidelines for automated methods have been published by the Pharmaceutical Analytical Sciences Group Automation Sub-Group 4 . A typical example of a dissolution method validation protocol is shown below in Table 2.
Typical dissolution method validation protocol
REGULATORY HURDLES
Over the past 10 years the regulatory burden on purchasing and using laboratory equipment and, in particular, automated systems, has increased significantly for pharmaceutical companies. In the early 1990s qualification of the equipment in the form of the 4Qs (Design, Installation, Operational and Performance Qualifications), were implemented. Following this in the mid-1990s Y2K issues became apparent resulting in significant investment in new software/laboratory equipment. More recently the FDA rule on electronic signature and electronic storage of data (CFR21, part11), has resulted in many vendors of computerised systems initiating development of software that complies with this rule. The net outcome from these various regulatory developments has been significant resources being expended by equipment suppliers and pharmaceutical companies both in terms of people and money. Unfortunately, if CROs are to continue to successfully develop their business for dosage form analysis then they too must conform to the same standards.
OUTSOURCING IN BIOANALYSIS VERSUS DOSAGE FORM ANALYSIS
CROs have been particularly successful in developing their business for the analysis of drugs in clinical samples. This investment has been in state of the art automated LC-MS systems. Numerous laboratories have set up over the past five years, both in Europe and North America, in which there are large numbers (10–30/company), of LC-MS instruments used which typically cost about $0.5 Million per instrument. This large level of investment must be done on the assurance of high sample throughput. The number of samples analysed in a clinical study can range from 400 to 6,000 and about 100 samples can be analysed in a 24 hour period. Perhaps the success of LC-MS technology in bioanalysis can also be attributed to a number of other benefits including:
The technique is easy to automate.
Method development time is reduced since the mass spectrometer is a selective detector and therefore samples require less clean-up.
The technology can be used by non-specialists.
Relatively reliable technique bearing its complexity to relatively reliable technique considering its complexity.
Improvements in technology driven by academia and instrument manufacturers.
In pharmaceutical development there has been an increase in the number of stability studies as a result of parallel development and global commercialisation of products. For example, for some indications special packaging and dose strengths are needed by Japan compared to other countries in the world. Also in commercial operations there is an increased need for flexibility in the supply chain which has resulted in extra stability studies to support a change in manufacturing site.
Typically, for most development products, 100 dissolutions, 100 assays and 100 degradation profiles need to be performed in about 10 working days for each 1, 3, 6, and 12 month timepoints for Qualifying batches for an NDA/MAA regulatory submission. Over a five year stability study about 600 samples are produced for each test with about 400 of those requiring analysis in the first 12 months. Clearly automated dissolution equipment is a necessity to complete this task. Automated assay and dissolution instruments can cost about $0.1 Million per instrument.
From the comparison above the success of CROs in bioanalysis can be attributed to the very high number of samples that require analysis. Although the cost of an LC-MS is about five times that of an automated dissolution system, this appears to be more than offset by the sample numbers. It seems, therefore, for more automation to be employed routinely by CROs in dosage form analysis, increased numbers of samples need to be analysed.
SUMMARY
Contract Research Organisations have been successfully winning higher proportions of the R&D budgets of pharmaceutical companies. However, compared with the bioanalysis sector of development, CROs have not invested significantly in the automation of dosage form analysis since sample numbers do not appear to justify purchasing the technology. In SmithKline Beecham most products are targeted for robotics during Phase 2/3 and these methods are used for stability studies to support regulatory filings. Comparator development and stability work is generally outsourced in SmithKline Beecham.
There are numerous benefits for CROs to invest in automation and the most important of these include; easier technology transfers, capability to overcome peaks and troughs in workload, faster throughput of samples, reduced costs and increased compliance with regulatory requirements.
At present few CROs have taken advantage of the benefits that robotic automation can offer. In the future it is likely that large pharmaceutical companies will have key analytical tests for all development products automated thus making the option of outsourcing difficult. If CROs fail to implement automation they will not be able to effectively compete for business.
ACKNOWLEDGEMENTS
Nigel North wishes to thank John Stanley, John Gostick and Les Brockhurst for their help in developing the robotic systems in Pharmaceutical Technologies.
The author also acknowledges Hank Surprenant and John Baldoni for their managerial support of automation in Pharmaceutical Technologies.
TRADEMARKS
Zymark TPW II™, Zymark MultiDose™ and Zymark MultiDose Plus™ are registered trademarks of the of the Zymark Corporation.
Automated Dissolution Module™ is a registered trademark of Source For Automation Inc.
