Abstract
In recent years Matrix-Assisted Laser Desorption/Ionization Time-Of-Flight Mass Spectrometry (MALDI-TOF-MS) 1 has grown rapidly in its importance in the areas of Genome and Proteome research. The received mass spectra have high information content and can be acquired in a few seconds. Another reason for doing MALDI-TOF-MS is the possibility of fully automated processing.
This presentation was given at the 2000 International Symposium for Laboratory Automation and Robotics (ISLAR) held in Boston, MA, October 15-18, 2000. The full manuscript is available on CD-Rom and can be acquired by contacting Christine O'Neil, 508-497-2224; email
INTRODUCTION
MALDI is based on the laser desorption/ionization of a crystallized mixture of analyte/matrix by a short laser pulse. The ions are accelerated inside an electrical field to give all ions the same kinetic energy, which is equal to 1/2m/z v2, where m/z is the mass-to-charge ratio and v is the ion velocity. By measuring the time-of-flight between the laser pulse and the event when the ions hit the detector, m/z can be calculated. A schematic diagram of a MALDI-TOF instrument is shown in Figure 1.
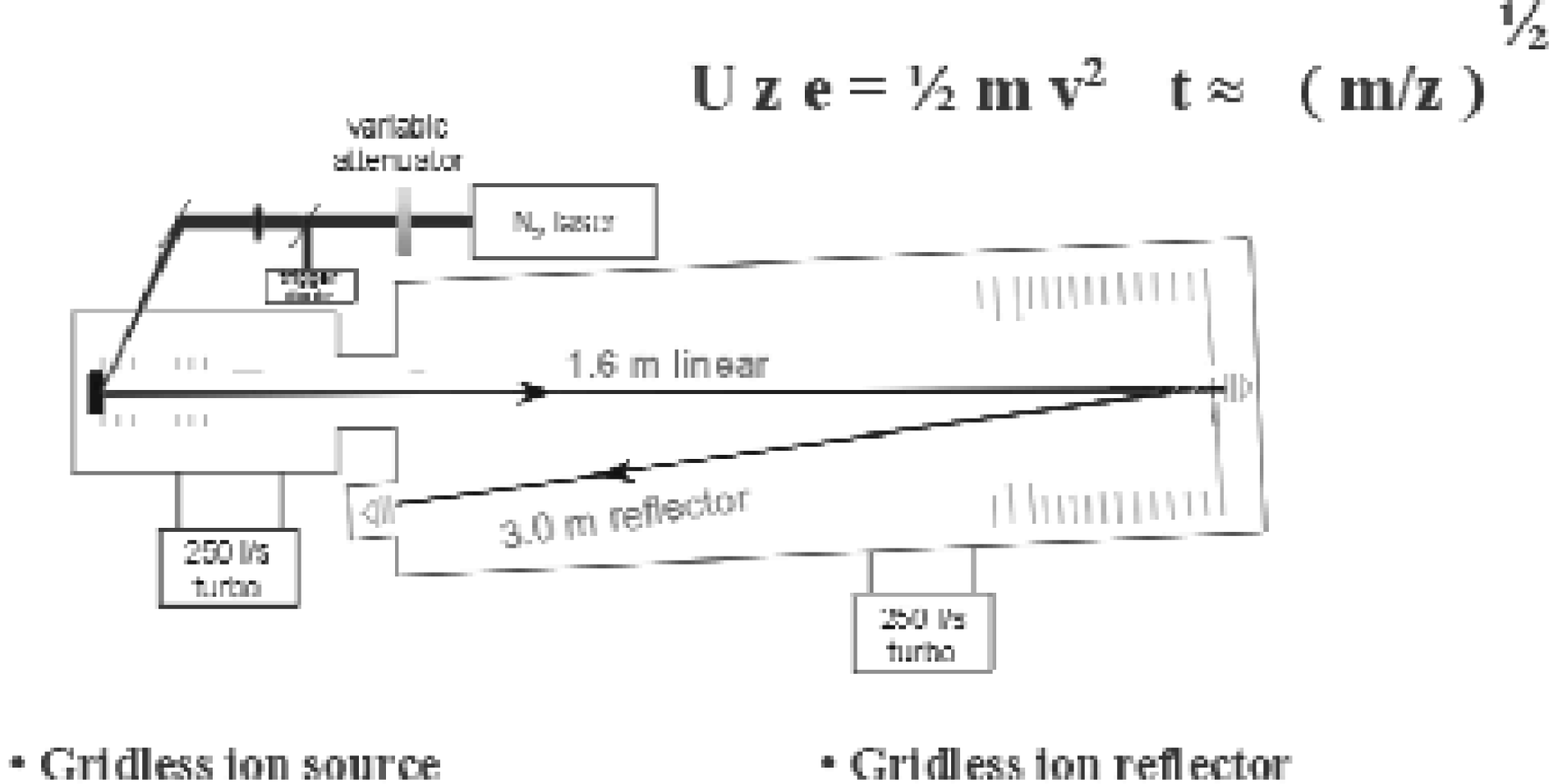
Schematic diagram of a MALDI-TOF-MS
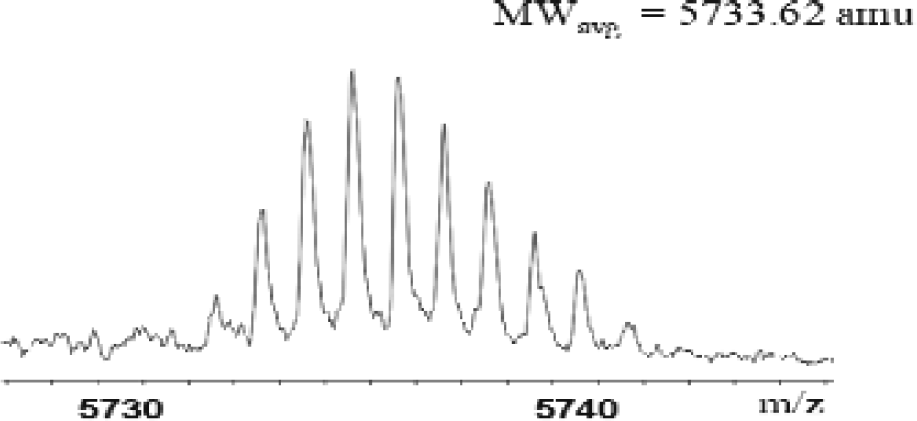
Figure 2 is a mass spectrum of a high molecular weight glycoprotein. Low mass resolution and mass accuracy are typical for this mass range. In the mass range below 10000 amu, Figure 3 shows an example of bovine insulin. A mass resolution of over 10000 (FWHM) and mass accuracy in the low ppm region can be achieved.
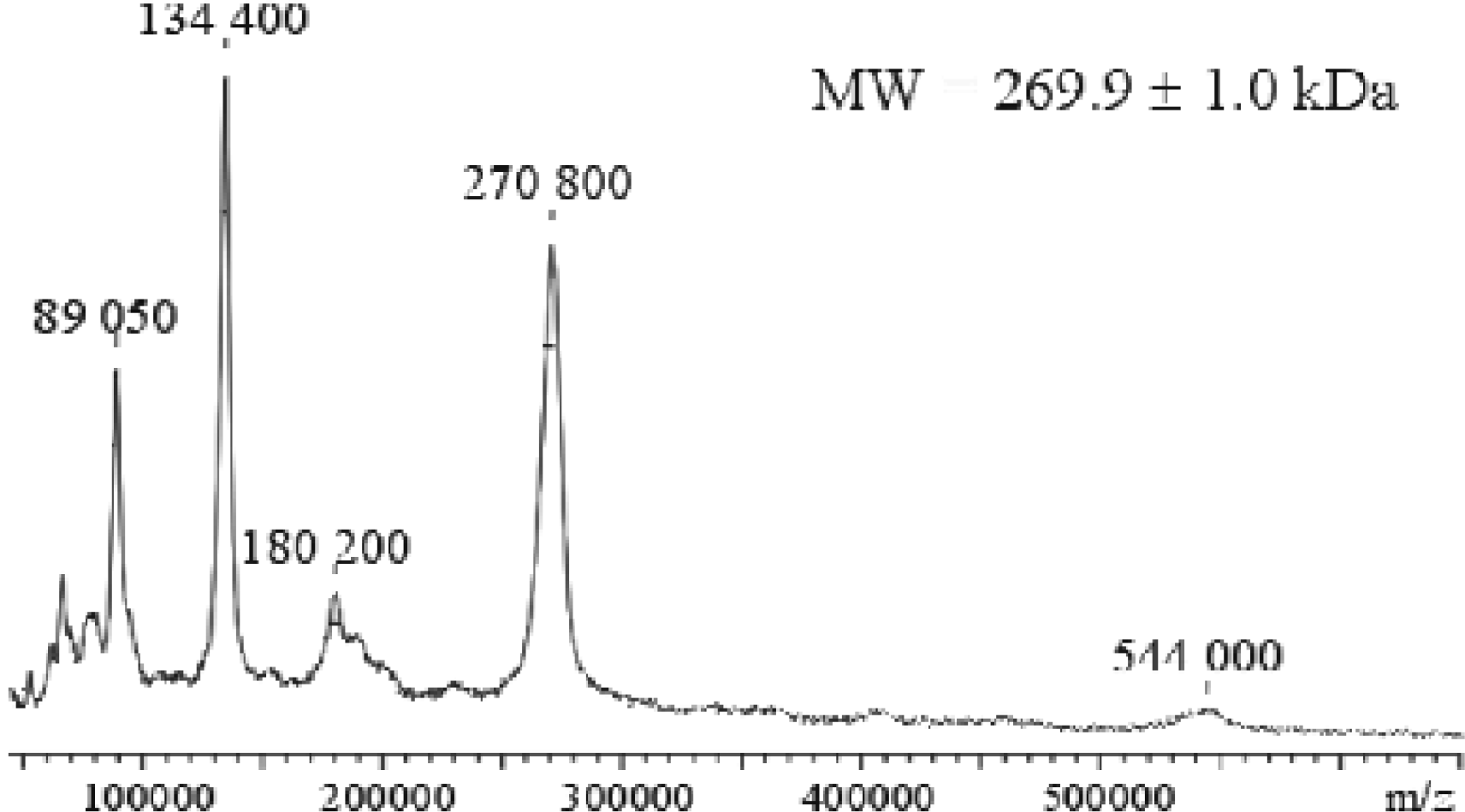
Mass spectrum of a large glycoprotein
MALDI is based on the idea to incorporate the analyte into the crystal structure of a matrix. The matrix, generally a derivative of cinamic or benzoic acid, absorbs the light of a short laser pulse to be desorbed and to simultaneously desorb and ionize the analyte. The sample preparation is carried out by mixing 0.5 μl of analyte solution with 0.5 μl of the matrix solution. The total amount of 1 μl is dropped onto a stainless steel surface and allowed to dry.
RESULTS
Figure 4 shows the result of such a preparation (“dried-droplet”), where the dot represents the focus of the laser beam. When a droplet is prepared with an overlap on a hydrophilic anchor surrounded by a hydrophobic area, it shrinks down, with time, exactly on top of this anchor.

“Dried-droplet” (left) vs. AnchorChip™ (right) preparation
So “Sweet-spot hunting” is no longer necessary. Furthermore, this AnchorChip™ technology provides the possibility to use standard liquid handling systems. No nanodispensers (like ink jets) are necessary. The sensitivity is also increased, or better, the total load of sample can be reduced. This is shown in Figure 5 by the comparison of a “dried-droplet” vs. an AnchorChip™ preparation of 1 to 100 fmol total load of a tryptic digest of myoglobin, where a sensitivity increase factor of 100 can be observed. For totally unintended high quality MALDI-MS acquisition the laser power has to be adjusted. This is done by an intelligent fuzzy-logic algorithm, which takes the mass resolution and signal-to-noise ratio into account in deciding whether or not a mass spectrum is co-added. The sample preparation can be performed on almost any standard liquid handling system.
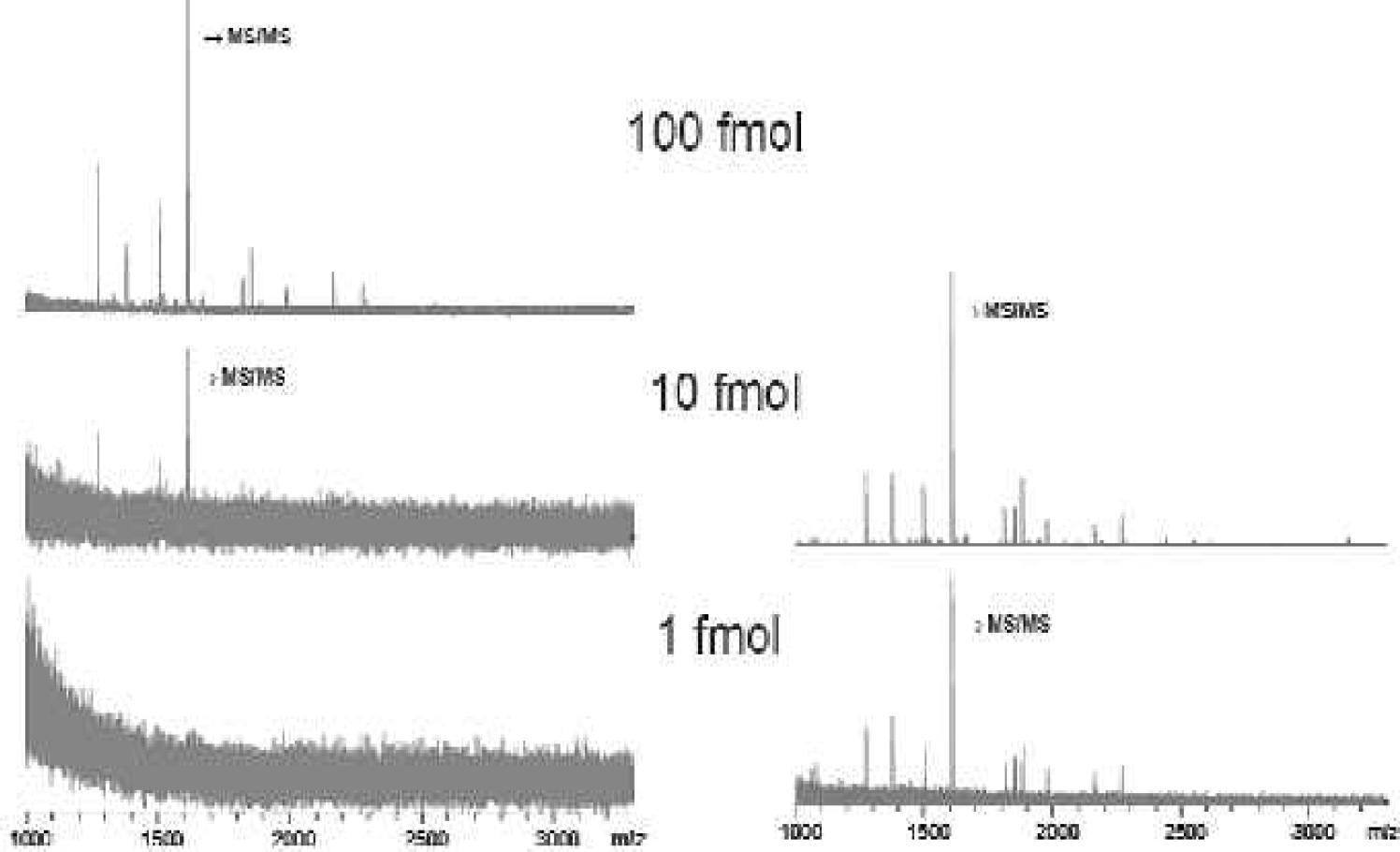
“Dried-droplet” (left) vs. AnchorChip™ (right) preparation of a tryptic digest of myoglobin
The MAP II/8, which is shown in Figure 6, prepares 8 samples simultaneously with disposable tips to avoid carry over. Special preparation procedures (“dried-droplet”, “thin-layer”, a.s.o.) or clean up procedures (“washing”, “ZIP-Tip™ * ”, a.s.o.) are integrated into a software package which is totally under Microsoft-Excel control.

MALDI-Auto-Preparation system with 8 disposable tips (MAP II/8)
The whole measurement setup can be edited and viewed by an Excel-Sheet to give an overview of where the sample came from, how it was prepared and measured (AutoXecute™ software) and how the data were processed (XMASS™, MSBioTools™ or GenoTools™ software) or how the results were correlated (Mascot ** ) with a protein database. Where the ExcelSheet can be found, stored inside is a transponder, which is incorporated inside the MALDI target. At the very end all data are exported/imported in a relational database.
The key is a novel MALDI-TOF-MS (autoflex™, Figure 7), which, together with a Twister™ *** allows the totally unattended operation of 20 AnchorChip™ targets (20 × 384 = 7680 samples, or 20 × 1536 = 30720 samples) within a day.

autoflex™ (MALDI-TOF-MS)
SUMMARY
The autoflex™ is the only MALDI-TOF-MS system on the market that was consequently developed to do totally unattended high throughput data acquisition in the field of Genome or Proteome research. This mass spectrometer together with the AnchorChip™ technology on a micro well plate format bridges the gap between sample preparation, done on liquid handling systems, and Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry.
TRADEMARKS
Footnotes
*
ZIP-TIP™ is a Trademark of Millipore.
**
MASCOT is a Trademark of Matrixscience.
***
Twister™ is a Trademark of Zymark.
