Abstract

This presentation was given at the 2000 International Symposium for Laboratory Automation and Robotics (ISLAR) held in Boston, MA, October 15-18, 2000. The full manuscript is available on CD-Rom and can be acquired by contacting Christine O'Neil, 508-497-2224; email
INTRODUCTION
With the level of high throughput screening (HTS) achievable today there is an ever-increasing demand for the production of serially diluted compound assay plates for secondary screening, structure-activity relationship (SAR) development, and compound specificity across biochemical targets. The latter task is addressed by optimizing the production of serial dilutions and making copies from that dilution scheme. A Tecan Genesis that executes an established serial dilution protocol is combined with a Zymark track system to accomplish the entire task on a single machine.
CONCEPT OF THE AUTOMATED PROCESS
The system is comprised of a Zymark XP robot on a 2-meter track that includes 3 plate carousels, a RapidPlate 96/384, a Tecan Genesis 150 with disposable tip adapters, and a Zymark plate sealer. The controller language for the Zymark system is Easylab (Zymark) with PCS (Productivity and Communications Software) as the process scheduler. The Tecan was programmed with Gemini v3.1.
A compound master plate is retrieved from the first carousel and placed on the deck of the Tecan for serial dilution. The RapidPlate is loaded with tips and the previous serially diluted plate (from the Tecan). The subsequent copy plates for assays are then retrieved from the carousels and copies of the dilution are made as the next serial dilution is proceeding. Each plate is sealed and returned to its original location. As each plate is retrieved and/or returned, the RapidPlate is executing a transfer function. This process has the gripper/arm traveling occupied during 75% of its moves.
The general order of mechanism is quite simple but when coordinating and executing this with a single arm to transfer a single plate between five peripherals the time delays can accumulate rapidly. Although data quality is the primary issue, all mechanized processes should be optimized for time.
System
Serial dilutions are commonly performed in DMSO to avoid compound precipitation. Assay stringencies were achieved by transferring 1μL samples from the serial dilution plate for the production of multiple copy assay plates. This approach accommodates a range of low-volume assays with a standard volume of compound while maintaining a low percentage of DMSO. These identical copy plates were then distributed to assays within a systems-based research area for the purpose of compound specificity screening.
An important component of this automated process is receiving compounds as liquid samples from a central distribution group as a standard format of compounds on a master plate. From earlier work (unpublished) it has been demonstrated that compound distribution as liquid sample (chemistry dissolved in DMSO) does not significantly effect compound stability. The format that simplified and most benefited the automated process was the “column-daughter” arrangement. This is a 96 well plate with the chemistry in the first column only. This plate format was designed with consideration for robot handling and to facilitate programming. For ease of plate handling and deck efficiency, the “column-daughter” format eliminated the need for a master plate, containing 80 compounds, to be distributed to a column daughter equivalent. The easiest step to automate is the step that you don't do.
Another consideration in simplifying and increasing the efficiency of the process is recognizing that on a track or rail system the gripper/arm is the bottleneck. By minimizing the number of moves the arm travels while unoccupied, the efficiency of the system increases. This consideration led to the coordination of three carousels and a single RapidPlate to rapidly produce the copy plates. The following is a description of how each peripheral was optimized.
Carousels
The goal was to retrieve the plates from the carousels in an order that eliminates the carousels' PCS start-up/shutdown times from the plate acquisition process. This elimination of startup/shutdown time was achieved by making the next plate to be handled in the process always come from a different carousel than the last plate (Figure 1). With this arrangement of carousels, such that the next plate to be put into process would never interfere with the last plate returned, the time lost in carousels processing plates was minimized; i.e., Carousel #1: returned plate; most recent copy; Carousel #2: provided the plate active in RapidPlate; copy being made; Carousel #3: next plate to enter process, ‘start-up’ doesn't interfere with retrieval of this plate.
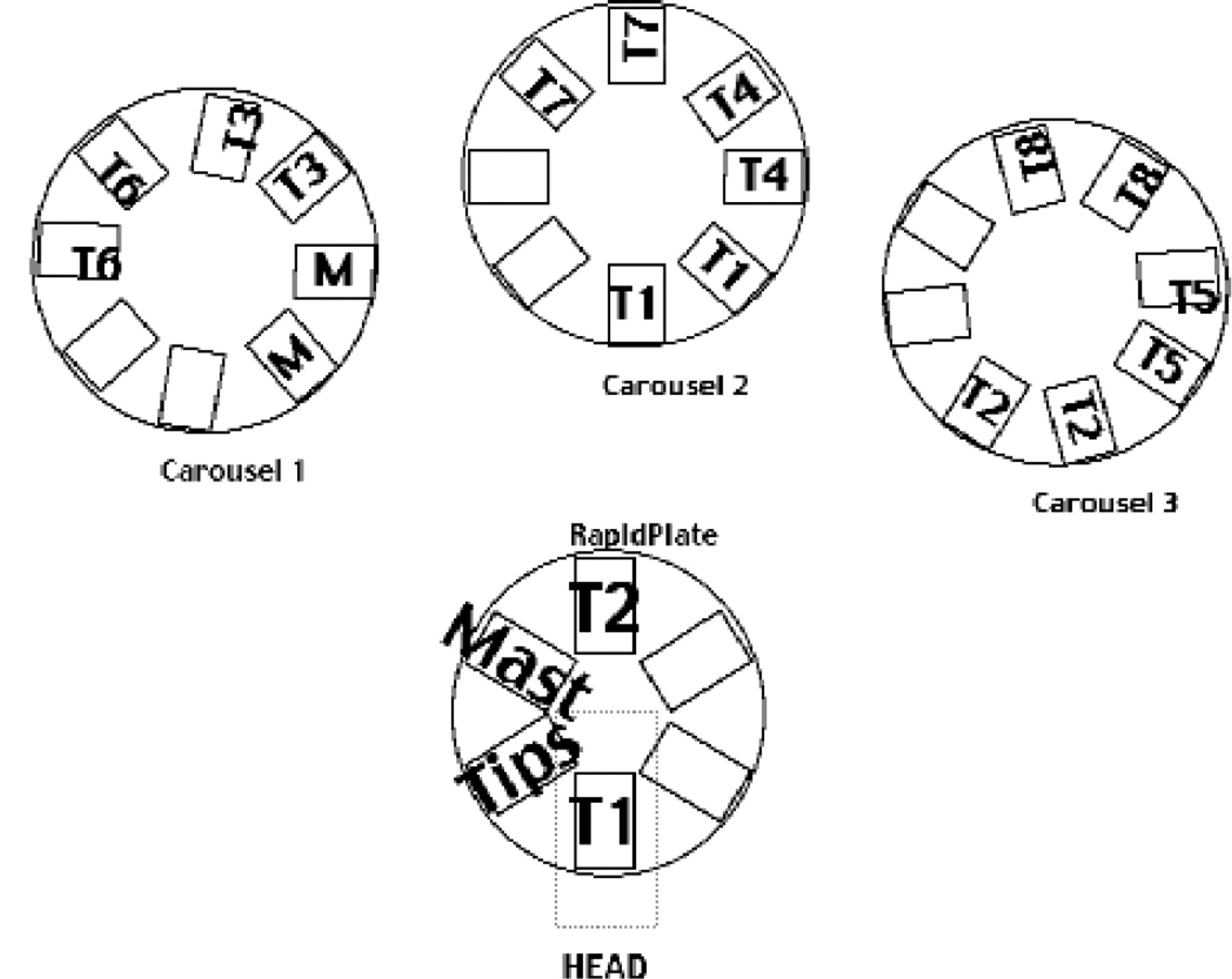
The racks that contain each defined target assay plate are staggered in the carousels to allow plate processing to occur without delay from ejecting or parking a plate. M; Master plate, T1; Target assay plate, Etc.
Tecan Genesis RSP 150
Serial dilutions using standard tips were easy to validate with dyes in DMSO. However, the accuracy of the serial dilutions became compromised with the introduction of real compounds because of carry-over issues specific to compound classes. While using standard tips, this process was requiring an escalating number of washes to compensate for carry-over, resulting in an increasing volume of liquid waste and a significant amount of additional washing time. A standard serial dilution application that would work with any compound class was needed to successfully automate this task.
Through many years of experimentation using Tecans for the production of compound dosing across five orders of magnitude the most successful and reproducible approach incorporates 250μL syringes and disposable polypropylene tips. 200μL tips are used for convenience and accuracy to fill the plate with the diluent DMSO. 50μL tips are used for accuracy in the serial dilution of the compounds, diluting across the plate for 11 concentrations and leaving column 12 to contain only DMSO for assay controls. The tips are not changed between individual concentrations but rather at the end of each serial dilution.
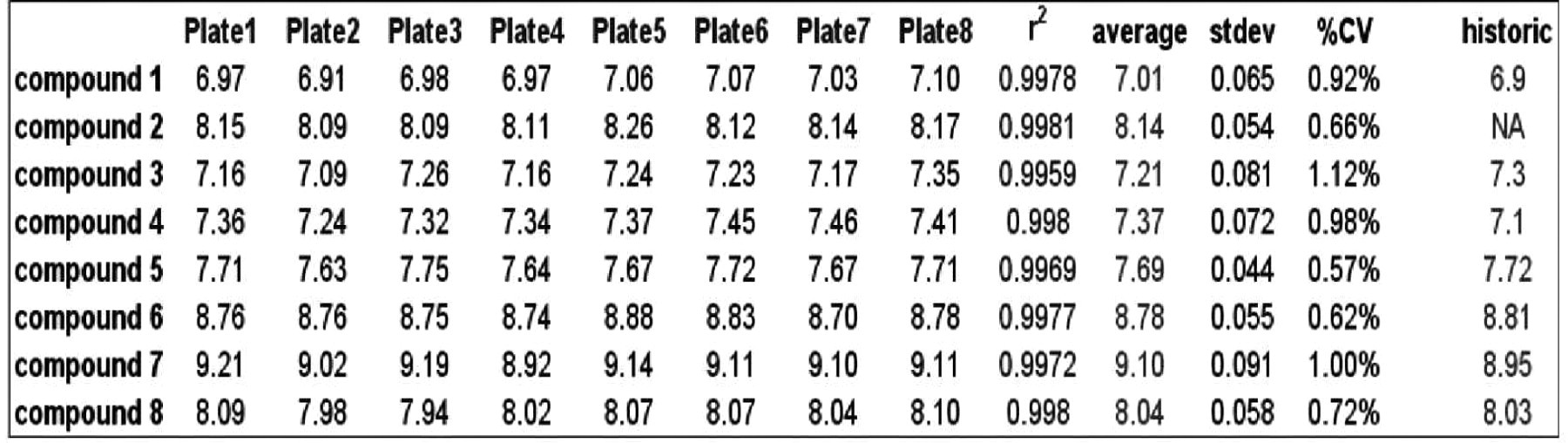
A Tecan Genesis 150 with Gemini (Tecan, Ag) as the controlling software for the serial dilution program allows for better control of the liquid handling and yields a more accurate result. Using real chemistry, goodness of fit analysis (r2 values from least squares curve fitting) and comparing historic IC50 data validate the process (Figure 2). We assumed that the historic data, that was produced manually, was the correct value.
RapidPlate
Employing a RapidPlate arrangement that allows the hand to place a plate on the turnstile and return with a plate in hand can save time. Placement of these two transfer positions in opposing positions allows for movement of the active copy under the transfer head and exposure of the finished plate in position for hand access simultaneously (Figure 1). Now the arm can return to the carousel occupied with the finished target plate in hand to retrieve the next target plate. A single transfer is dispensed to the active plate while this last plate is returned and the next plate acquired.
Quality of the tip seal was maintained by attaching the tips a single time per master dilution plate for the 8 individual transfers to copy plates. The new RapidPlate software (96/384 standalone version 1.01), that allows for more control over pipetting functions, provided the control to achieve 3% CV's on 1μL transfers to dry plates (Figure 3). This accurate transfer maintains the quality of the original serial dilution and guarantees the assay data to be representative of quality data.

Uniform transfer of 1uL tartrazine/DMSO solution by a 96–well head RapidPlate to 8 replicate copy plates. Average volumes transfered to each plate are calculated against a standard curve. Replicate CV's are calculated across individual plates. The CV for average 1uL transfer to replicate plates is 3.99%.
CONCLUSION
With the high demand for compound selectivity within systems based research, there is a need to distribute compounds on a regular basis to several targets simultaneously. In order to produce data in a timely fashion, it is appropriate to distribute all compounds of a single serial dilution format to a set of multiple replicate assay plates. All the components necessary to handle these sets of selectivity compounds from serial dilution to distribution to assay plates are employed in this single-track system. A proven serial dilution procedure using a TECAN Genesis 150 for the serial dilutions was combined with a Zymark track system's carousels and RapidPlate.
Obstacles including an automated standard dilution application, small volumes, variable serial dilutions, speed, carry-over, and the future of 384–well formats were all addressed by using disposable polypropylene tips, low-volume syringes, and Gemini software for optimal liquid-handling control.
Increasing the efficiency of the machine by traveling between peripherals with an occupied hand is accomplished by using the RapidPlate as a turnstile rather than a fully loaded deck and the staggered carousel racks as a supplier of ready plates. Locating plates in opposing positions on the RapidPlate turntable allows the plates to rotate between the two positions, one under the dispense head, while exposing the opposite plate position to the hand. In this manner, the hand is constantly feeding and replacing the RapidPlate so that while it is dispensing to one daughter, the finished daughter is being sealed and housed and another plate is retrieved to replace the vacated position. The serial dilution of the next plate is being executed while distribution of the current compound plate is being completed.
Validity of the liquid handling of the entire system is confirmed by the quality of data produced. The accuracy is evident in the comparison of control data to historic values and goodness of fit (r2) statistics on new dilutions.
There are several advantages to distributing for secondary screening in this fashion. This program allows for minimization of compound stores required for distribution to targets. The amount of compound needed to distribute from one serial dilution of a compound to multiple targets is the same amount previously required by each individual target and therefore can extrapolate to 8–10 fold savings in chemistry stock. The reduction in the amount of compound distributed lessens the burden on the chemist and compound distribution services. There is timely turn-around of distribution from time of serial dilution to time of plating. The number of freeze-thaw cycles is significantly reduced since all assays are plated in one session, rather than plating on an as-needed basis. Because a single box of tips is used per master plate distribution to all copy plates, the cost of consumables is insignificant compared to the quality of the data generated.
Perhaps most importantly, this program yields good compromise for bench-to-data between chemists and biologists. By establishing a formal program such as this and dedicating a robotic system to the task, the time element is minimized on new chemical templates screened against a system-based target.
The current capacity of this system with three carousels is 44 masters, with eight different assay plate definitions, totaling 352 copy assay plates. The production rate of the system is 32 plates/hour, taking 11 hours to accomplish the full capacity of the system.
