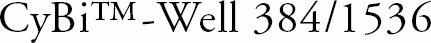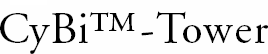Abstract

Jena, in Thuringia, Germany, is a city of contrasts. When approaching Jena on the Autobahn one is immediately struck by the large gray apartment buildings dominating the skyline standing as stark monuments to the communist era. But Jena today is a very different place. It has transformed itself into an extraordinarily progressive city blossoming with hightech industry. Building on the strong foundations of Carl Zeiss, who established Zeiss Optical, Jena has re-emerged as a leader in the field of automation solutions. We have selected one of the key biotechnology companies in Jena, CyBio AG, as this issue's cover and the focus of this article. Future papers in JALA will detail on a more technical level their products and applications. This article intends to provide the reader with a broad overview of the emergence of CyBio AG, and the future direction of its operations.
Less than two and a half years ago CyBio AG, then a division of Jenoptik AG, was a small organization focused on building instruments, sold through distributors. Jenoptik AG decided not to focus on Biotech as a strategic devision. At the same time the instrument market grew rapidly, therefore CyBio was spun off in order to get closer to their customers to understand customer needs. The realization was that the customer is the greatest resource for new ideas. Initially the sales team was formed in Germany. Within two years they had expanded to have a sales force in France and Britain. The next stage was to break into the US market, so they ended their relationship with their US distributor and opened offices in the US in December.
Petra Strümper is one of two CEO's of CyBio AG. She joined the forerunner company Opal Jena GmbH in 1994 when what is now CyBio was still the research and development wing of the parent company (Jenoptik AG). In 1995 she was appointed Managing Director of Jenoptik Bioinstruments GmbH (former Opal Jena GmbH), with a staff of 16 people. Today CyBio employs over 130 people. In 1999 Jenoptik allowed CyBio its independence. The initial public offering of stock (IPO) took place at the end of November, 1999, and was over-subscribed by 23.8% times. The proceeds from the IPO provided CyBio with strong financial resources to devote to implementing their bio-cybernetic concept: After automation, CyBio foresees a laboratory revolution in which intelligent and globally networked laboratories will increase the efficiency of finding new pharmaceutical agents.
Dr. Petra Strümper, CEO CyBio AG

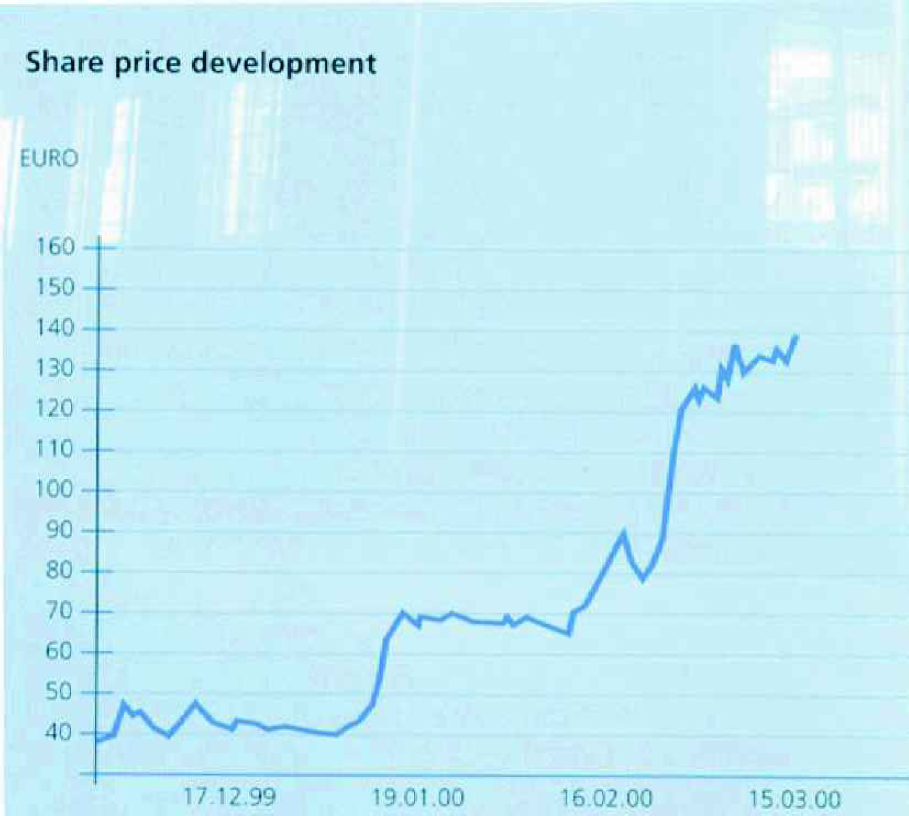
CyBio AG Share Price Development
Petra Strümper cites three reasons for their success, 1) Development of products based on market feedback; 2) International distribution; 3) huge investment in Research and Development. She further stated that they have the advantage of being in a niche market, which she defined as “knowledge of total Process”. Forty percent of CyBio's staff are employed in Research and Development. This allows the company great flexibility in adapting its products to meet market demands in a timely manner. In 1999 they spent almost 2.5 Million Euros on R & D, up from 70K in 1997. In the past they had to rely on public funding for research, now CyBio is entirely free to fund its own research. CyBio has engaged, and will continue to engage in collaborative research projects, and shall also consider the purchase of research-oriented companies which would compliment their business.
CyBio opened offices in North America since they view North America as the largest biotechnology market in the world. CyBio AG also foresees significant growth coming through its various subsidiaries CyBio Instruments GmbH, CyBio Software GmbH, CyBio Screening GmbH, among others. Diversifying the company through subsidiaries allows each company to focus exclusively on research and development in its own specialized area. A good example of how this works is CyBio Software. Control software is vital to insuring the functioning of the HTS laboratory. CyBio's software, CyBioControl, enables the pippetting, scheduling, stacking and various other functions to be run from one PC, a function that is vital to insuring optimal performance from automated high throughput screening laboratories. CyBio adapts the software for the individual needs, including the integration of systems from other companies. CyBio Software's role is to use the feedback information from customers to constantly update, adapt and improve the software, according to the daily requirements of laboratories.
The management of future directions of research and development is in the hands of the Product Coordination Division. They interface with the sales force and the researchers, their job is to market justify any new idea or concept. Put simply, their role is to determine if an idea can be turned into a product at a marketable price. This team consists of scientists with a background in both biology and engineering, which CyBio feels is the essential combination in their industry. They are also responsible for the on-sight set-up of new machines, and work with buyers at their site until the system is running to the customer's satisfaction.
“In this business you need a perfect service for customized solutions more than sales people”, said Wolfgang Lerch, head of International Sales for CyBio AG. CyBio offers its customers immediate call-back service to insure that any problems do not lead to a serious delay in the HTS process. This process builds the bond between CyBio and its customers that insures the company remains in touch with immediate market needs. Their sales team are all scientists, biologists, molecular biologists or biochemists, trained in the usage and maintenance of the equipment. Lerch added, “Nearly all of the instrument and software produced by CyBio had the origin in customer needs”.
CyBio's sales have remained stable in terms of the markets into which they sell. North America accounts for about 50%, Europe 45%, and Asia 5%. Within Europe Germany and Britain are the biggest markets, though Switzerland, Sweden, France and Belgium are also important. As part of their marketing strategy, CyBio exhibits at important trade shows around the world. This offers them the opportunity for direct contact with many customers. It also provides a forum for getting customers together to discuss what they like or do not like about the company's instruments. At each major trade show CyBio sets highlights on new products or features. As they already know who their customers are, it is a useful way of showing them a new system as it operates.
CyBio prides itself on the fast development of its instruments, from conception to product launch. The average time is six to nine months, an extremely fast turn around time. The process by which decisions are made to develop a new instrument is entirely based on feedback from customer needs. The Sales Manager can initiate the process which then is strategically decided together with the Board. This they believe makes them quite unlike many other companies. It puts the Sales Manager to the fore in deciding on new products, based on information from the field. Frequently, when presented with the necessary evidence, the board will give the go-ahead to a new project within a few hours. At the present time there are many projects in the development phase. Of these, more then 50% will eventually come to market.

In mid 1998 CyBio AG shifted their emphasis from instrument providers in the liquid handling market to being the developers of complete solutions for High Throughput Screening -from instrument components, up to service providers for a modular technological platform. This important shift also required the shift in sales strategy as it required CyBio to be closer to their customers, and to cease selling through a distributor.
Today CyBio's range of modular products to a full screening platform include the following:
For fast and efficient handling of high throughput screening jobs, CyBio has the CyBi-Well 384/1536 which makes even the most complex high throughput screening process easier. With 384 simultaneously operating pipettes, this system is significantly faster than a conventional 96 well automated pipettor. A 384-well microplate can be filled in one process, taking under 20 seconds to complete. It takes just 30 seconds to fill 1536 wells in four steps.
The CyBi-Well 384/1536 is compatible with the entire CyBi-Well family. It is a highly specialized piece of equipment, yet is supported by CyBio Control software. In just a matter of minutes, a user could have a program typed in and the CyBi-well 384/1536 running all from one PC interface.
Other features include an extended extractor that allows the use of shallow well microplates in deep-well units, and a convenient location of function keys. The CyBi™-Well can handle several types of plates, reformatting microplates, and the introduction of reagents. An integrated control unit allows for addition of a plate gripper, making plate handling more flexible than ever.
CYBI™-DROP
For a fully automated high throughput screening laboratory, CyBio makes the CyBi™-Drop system, a precision instrument featuring fast operating speed in reagent dispensing. This dispenser features a liquid pump with precise distribution of liquids even at volumes as small as 0.5μL. The CyBi™-Drop dispenses liquid without contact, thus eliminating worries of contamination. Using eight channels or two sets of eight channels, fluid can be dispensed to either 96- or 384-well microplates in a user's choice of step-by-step or on-the-fly modes. A special pump system uniformly feeds even the smallest fluid quantities through all eight channels. The unit is made of material that is resistant to most organic solvents and works with both deep-well and shallow-well 96- and 384-well plates.

The CyBi™-Tower uses a heat transfer system to warm up test solutions in shallow-well microplates. Each position for plate storage has a water system running through it. Warm water circulates through the system and is capable of warming a plate from room temperature to 37 degrees in just five minutes. The CyBi™-Tower has space for 20 microplates with or without lids and accommodates both 384 and 1536 well formats. The system can operate as a standalone device, be accessed with a robotic arm, or be integrated into a CyBi™-Screen-Machine system. Access time for the plates is under 30 seconds and plates can be accessed at random. The software monitors the storage position status of each plate.
CyBi™ — Lumax 1536

CYBI™-LUMAX 1536
This luminescence reader works with all plate formats from all manufacturers and gives an accurate reading of glow luminescence in just five seconds under normal conditions. Previously reading time could take as long as ten minutes, thus slowing the entire high throughput screening process. The CyBi™-Lumax 1536 recognizes Luminol, Luciferase CDP-Star or CSDP and scintillation proximity assays (SPA). It can stand on its own or be integrated with an automatic screening system.
CyBi™ — Replicator

The Lumax 1536 is equipped with a special CCD camera and an image intensifier that works like a night vision aid. Its detector works in either top-reading or bottom-reading mode, enabling the user to optimize for each experiment. This detector can work in either analogue or photon-counting mode. A convenient Windows interface makes the CyBi™-Lumax 1536 user-friendly and the software calculates the exact position of the well in the inserted microplate to determine the region of the image from which the measurement result originates. Optimum settings can be stored in method files for individual users and the output results can be accessed as either Excel or ASCII files with the option of calibrated colour code display.
CYBI™-REPLICATOR
The CyBi™-Replicator is a fully automated microplate copier providing the means for efficient assay preparation. It is capable of preparing up to 30,000 new samples in just two hours with a throughput of 140 microplates per procedure. Daughter microplates are filled from mother microplates at two separate pipetting heads each with either 96 or 384 tips. Minimum quantities of liquid are removed from the mother plate and transferred to daughter plates. A rotating arm delivers microplates from the stackers quickly and accurately. Twin, triple or even quadruple stackers are easily integrated into the system. The software accompanying the CyBi™-Replicator defines the number of mother and daughter plates and controls the duplication process. Labeling is done with an integrated barcode labeler, which automatically transfers the barcode on the mother plate to the daughter plate.
CYBI™-DISK
Taking the technology of the CyBi™-Well and adding a turntable to it results in the CyBi™-Disk. This turntable is based on the screening performance of the CyBi™-Well, but plates are transported via a turntable instead of along a system of tracks, carriages, and stackers. The system was designed with ten plate holders, each of which can accommodate a 96, 384 or 1536 well plate. Accessories such as reservoirs and adapters can be added to the carousel and the user also has the option of equipping it with a heatable reservoir. Other features include a drop catcher to eliminate unintentional dripping and a special lifter, which eliminates the need for height adapters. The CyBi™-Disk is useful in such diverse applications as magnetic bead handling and PCR.
CYBI™-SCREEN-MACHINE (COVER IMAGE)
The CyBi™-Screen-Machine allows the combination of the automated pipettor CyBi™-Well with other existing peripheral devices that provide fully automated ultra fast high throughput screening. At the core of the CyBi™-Screen-Machine is a rotating arm that provides an innovative mechanism for plate transfer. The arm rotates rapidly and positions plates reliably. Having the arm at the center of the CyBi™-Screen-Machine substantially reduces the amount of space the unit requires. As with other CyBio products, the CyBi™-Screen-Machine is user-friendly, has a proven control programme and has the advantage of being controlled from one PC.
FUTURE DIRECTIONS
Petra Strümper stressed her belief that CyBio AG is a pioneer in the field of bio-cybernetics. To this end they are developing new instrumentation that will use natural products to find effective therapeutic drugs in a cost-efficient manner. She believes it is critical that they be a leader in the drug discovery through the natural products field.
In September 2000 CyBio plans to unveil its latest system that comes out of its subsidiary, CyBio Screening. The underlying philosophy is to create the technology to bring nature to HTS. This is a project on which Dr. Ralf Thiericke, Managing Director of CyBio Screening Gmbh, has been working in the field of research for natural substances since 1984. He chose to come to CyBio because he needed, “a really good pipetting and screening machine”, from which to work. Previously he had been head of the “Screening Center” at the Hans-Knöll Institute, a government funded research body. CyBio had collaborated with them in their research efforts. Their focus was the integration of nature into HTS.
“Nine of the top twenty drugs on the market are derived from nature”, according to Dr. Thiercke. The gap in the market is the absence of screening with natural products. Bringing to HTS the technologies required to collect broad biological diversity and broad structural diversity, is presently impossible due to the absence of techniques on the market. CyBio Screening Gmbh believes it has an interesting new solution. This is the CyBi™-Xtract. This shall be discussed in greater detail by Dr. Thiercke in a future issue of JALA.
CyBi™ — Screen Machine

CyBio AG represents in many ways the face of the dynamic, high-tech era which has come about in the decade since the fall of the Berlin Wall. It is a company built on the strong foundations of the skilled engineering and scientific workforce employed in the region prior to the collapse of communism, blended with a driven and imaginative vision of competing in the global capitalist economy. With its dedication to research, its profound belief in rapid response to market needs, CyBio AG appears to be holding true to its vision of Research and Technology for Life as it seeks new solutions to the problems of the drug discovery process.
Dr Ralph Thiercke (left), and Conal F. Timoney, discuss CyBio Screening, a subsidiary of CyBio AG.