Abstract

INTRODUCTION
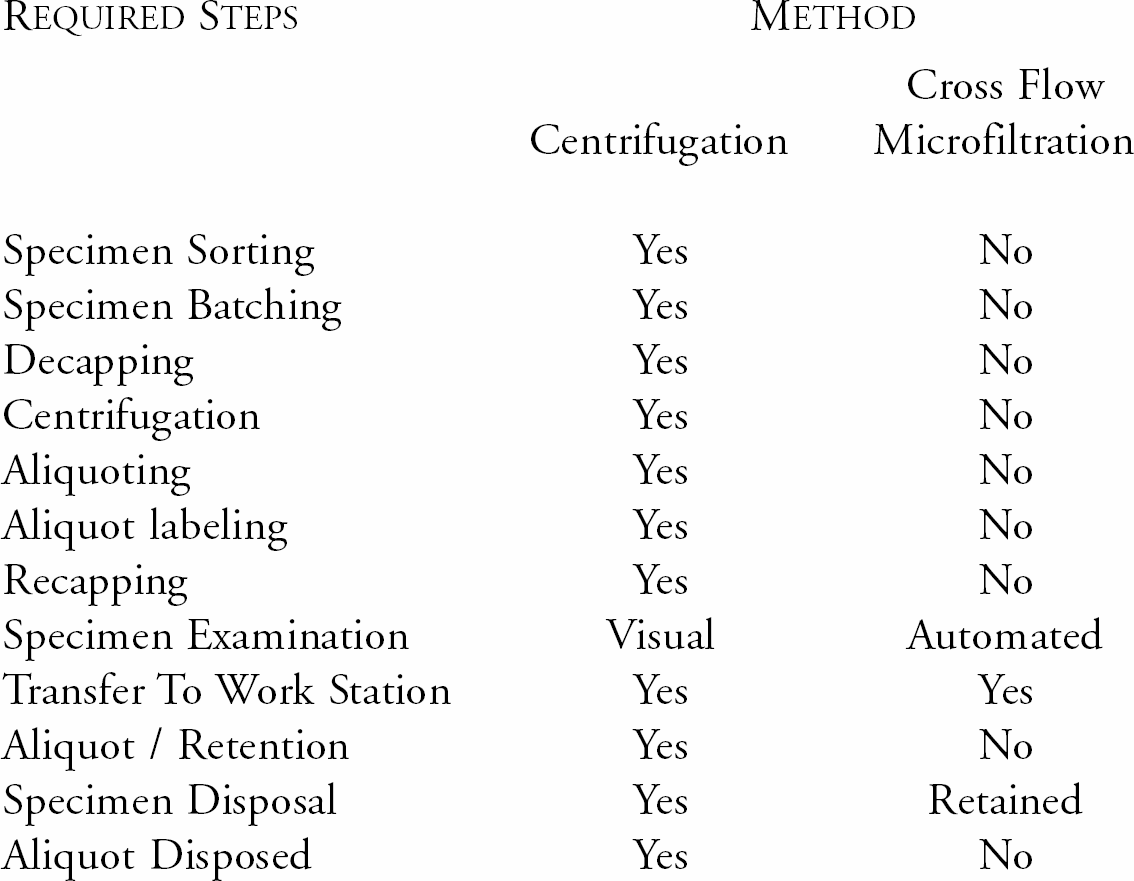
CENTRIFUGATION
Centrifugation is a traditional separation method for preparing analytical specimens. It is not the ideal method. About 75% of specimens are centrifuged. Centrifugation is the major delay in the laboratory's workflow. Specimen flow varies throughout the workday. Consequently, specimens are batched because centrifuges are in use; or delayed to collect a sufficient number of specimens to centrifuge. The result, in both cases, is delay. Each delay lengthens intra laboratory turn-around-time.
Like other methods used in the laboratory, centrifugation of specimens must follow specific procedures to achieve acceptable results. Centrifugation of whole blood to produce plasma or serum specimens for analyses can not exceed an RCF of 1500XG for more than fifteen (15), minutes without disrupting cellular integrity, thereby affecting test results. This precludes the use of high-speed short time centrifugation, or ‘STAT’ centrifuges.
Key Steps In Pre-Analytical Specimen Preparation
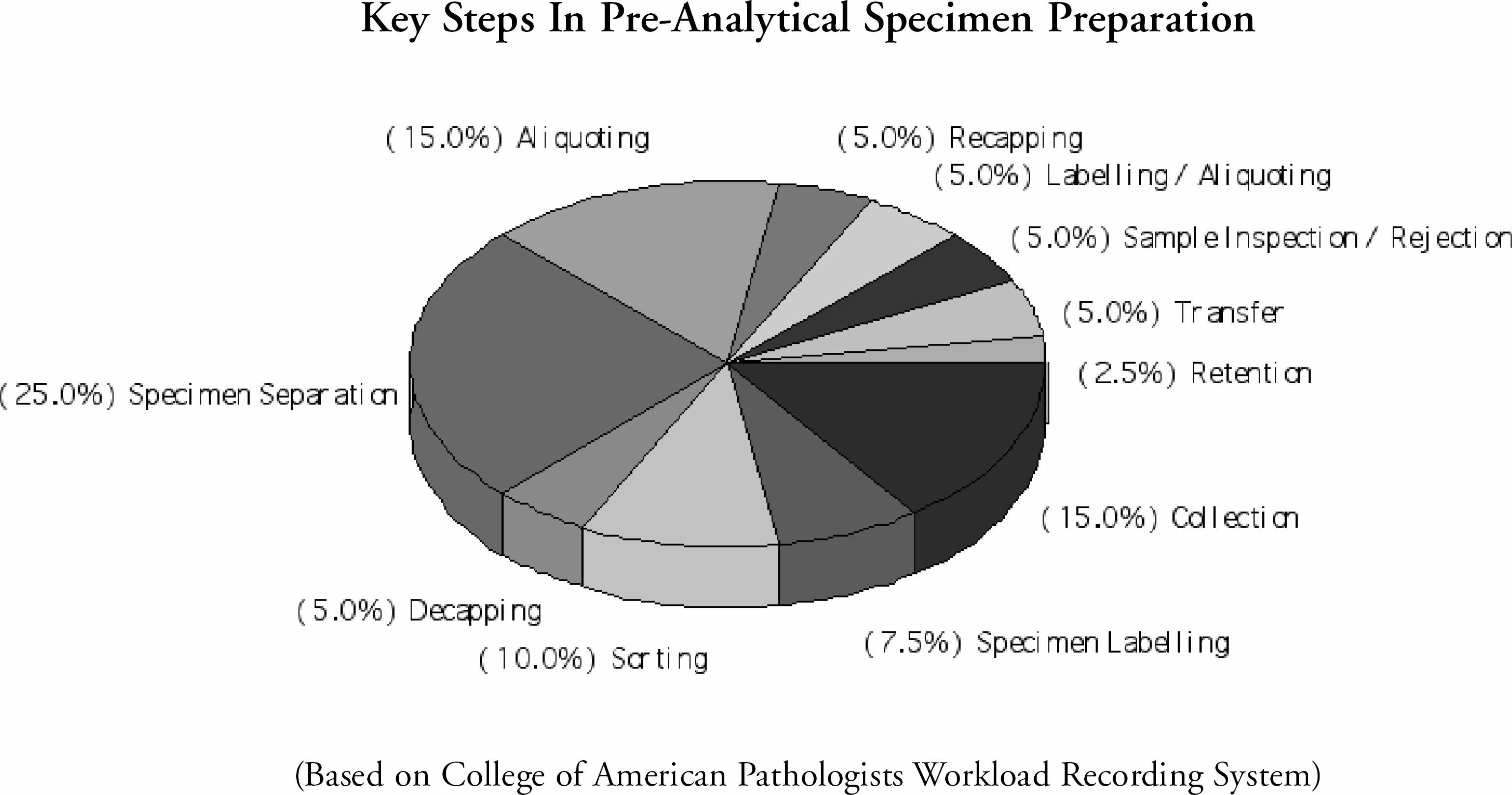
Cenrifugation As A Component Of Specimen Preparation: Specimen separation and its related functions comprise a substantial portion of the pre-analytical processes. It is also a major contributing factor in intra-laboratory turn-around-time.
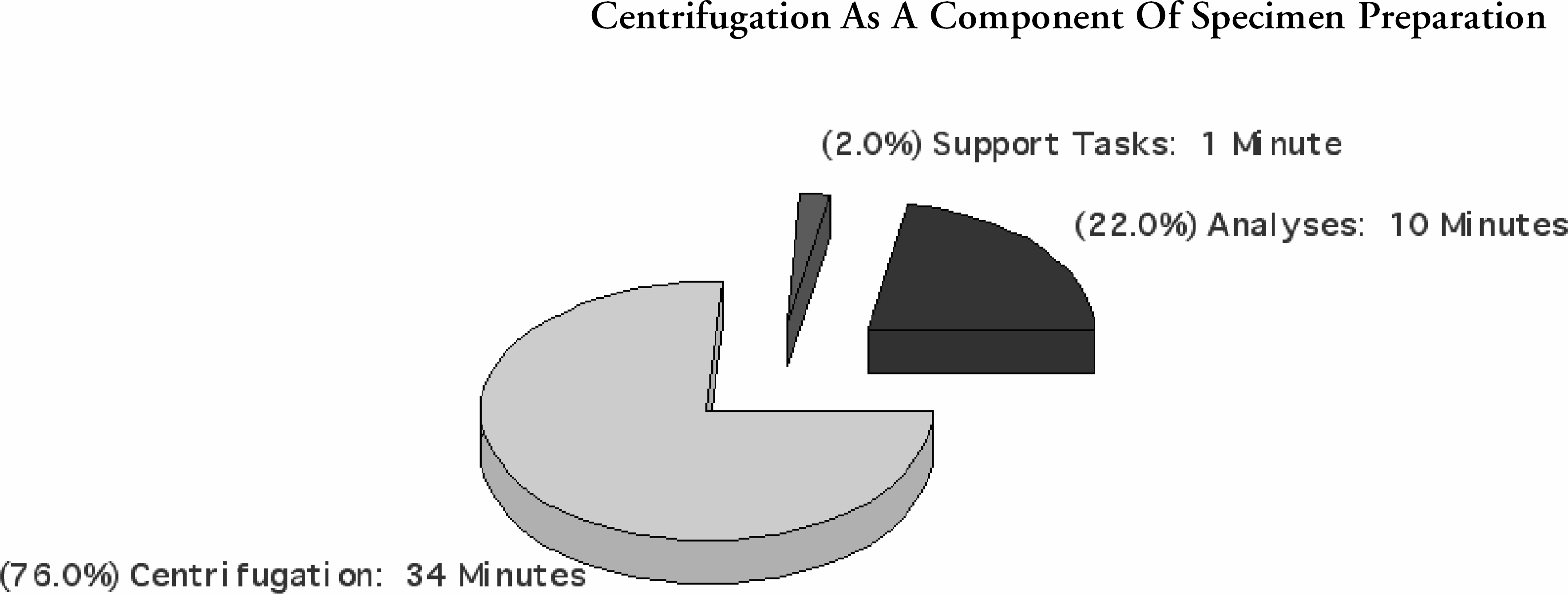
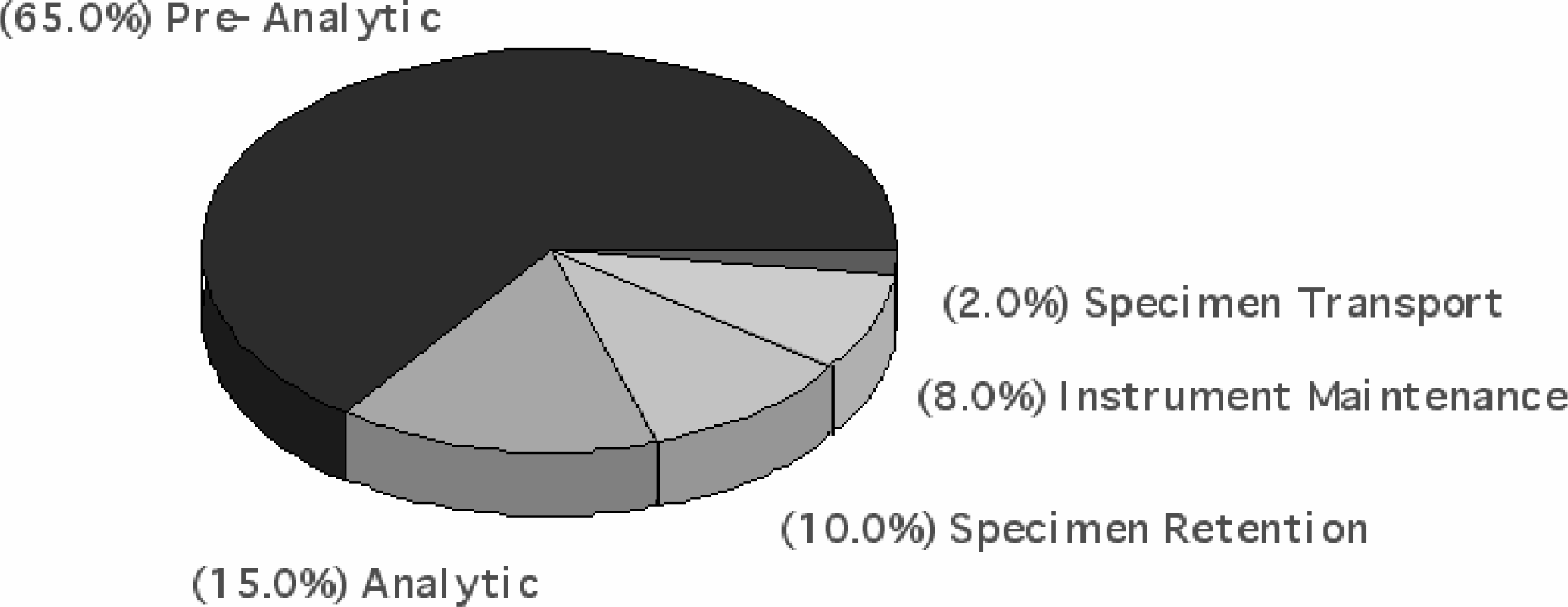
Unlike other laboratory procedures, the acceptability of a centrifuged specimen can only be determined at the end of the cycle or in the subsequent analytic phase. The lack of quality control processes for centrifugation result in variable specimen quality.
Centrifugation imposes significant limitations on the laboratory, affecting the general workflow. These limitations include:

Bio/Data Corporation has developed an alternative method for rapidly separating plasma from the primary patient specimen. This rapid method is based on precisely controlled, cross flow (tangential) microfiltration. Since 1985, this process has been employed in three generations of the Company's instrumentation. It is a validated technology.
Using this technology, a plasma sample is ready for analysis by standard test methodology in less than 30 seconds. Specimen volume and acceptability are also measured within the 30 seconds. In addition to the rapid separation, plasma separated in this manner is virtually free of cells, membrane fragments, cellular debris and cell contents. Such plasmas are, therefore, better suited for analysis than those prepared by centrifugation.
THE MICROFILTRATION PROCESS
Microfiltration is the separation of particles that range between 0.01μm and 20μm in size.
For the rapid separation of plasma from blood, a specimen is transferred between two reservoirs, flowing over a microporous membrane in alternating directions. By moving the specimen back and forth across the membrane, the membrane surface is continuously cleaned. This keeps the filtration efficiency at its peak throughout the separation process. As a result, very low pressure is required for precise control of the specimen flow and precludes cell disruption or migration. Plasma containing clinically significant analytes rapidly crosses the membrane. Typical anticoagulants do not affect the rate of microfiltration or the plasma volume. The maximum volume of microfiltered plasma is about a third of the whole blood specimen.
Microfiltration is the validated alternative technology to centrifugation. It allows rapid separation of analytical quality plasma directly from the unopened primary collection tube.
FILTRATION CELLS
A disposable filtration cell is the key component in the microfiltration process. The configuration of filtration cells can vary with its applications. Control and functions of the filtration remain the same. These include:
whole blood reservoir
microfiltration
plasma collection
specimen acceptability cuvette
analytical cuvette (in certain applications)
The components of a filtration cell are illustrated below.
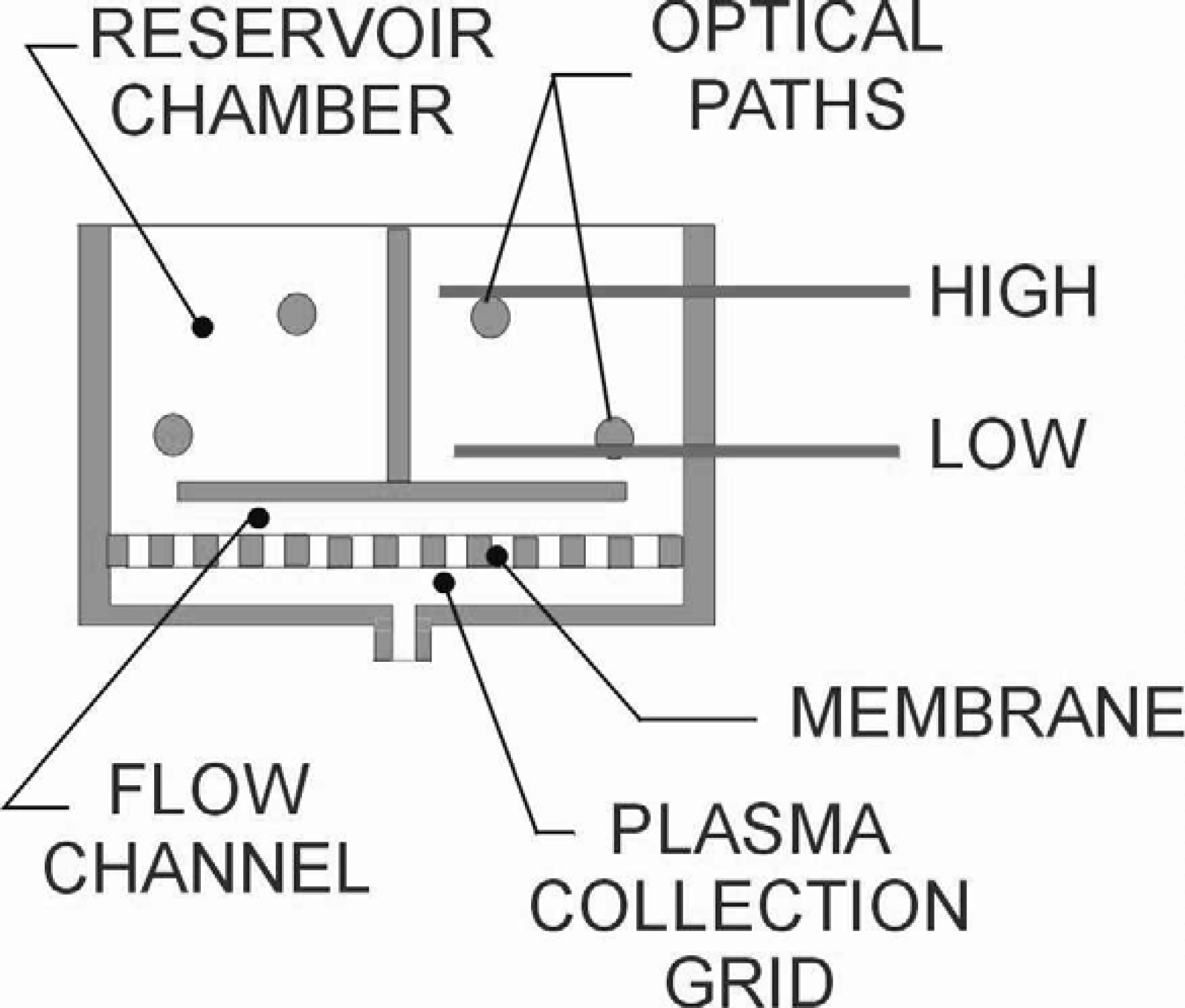
The diagram on the following page illustrates the microfiltration process and its controlling optical pathway. Each step takes about one second
The Microfiltration Process

Microfiltration eliminates the limitations and time delays inherent in centrifuge separation.
Specimen Preparation Method Comparison
PLASMA QUALITY
Microfiltration is a direct and gentle process. The primary collection tube remains unopened throughout the process, precluding pH and other artifactual changes in the specimen.
Microfiltered plasma is:
Immediately Available
More Physiologically Representative
Not Subjected To pH Changes
Not Subjected To Centrifugal Forces And Heat Stress
Not Contaminated By Cellular Components, Debris Or Lytical Residue
Automatically Checked For Analytical Acceptability
Not Affected By Hematocrit
Not Affected by Anticoagulant
Not Limited by Specimen Volume
Because they are virtually free of cells, cellular debris and contents, microfiltered specimens offer certain analytical advantages. This is important when assay detection methods are based on particle size or absence of cells, presence, or optical properties.
Effects Of Separation Methods On Plasma
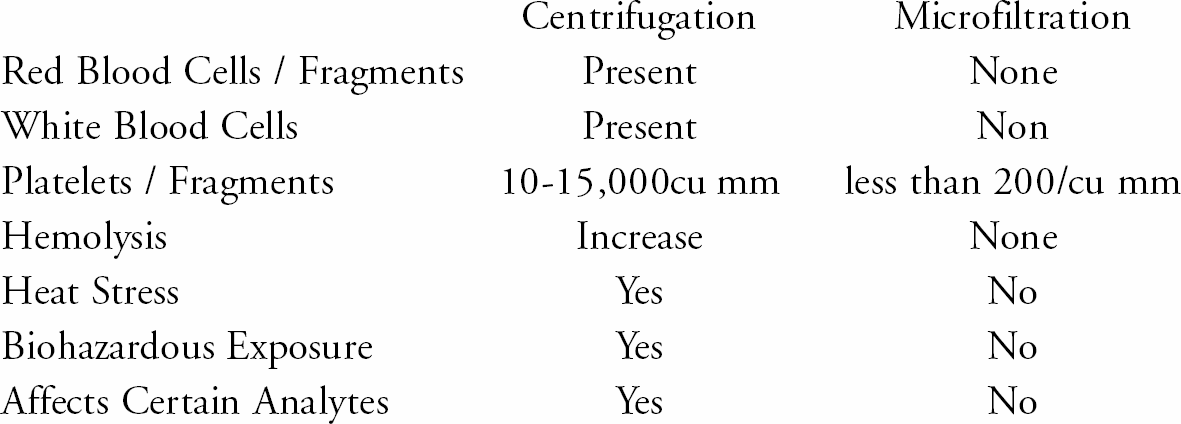
Recovery studies on analytes over a wide range of molecular size, weight, surface properties and detection limits averaged 99.7% on microfiltered plasma.
SPECIMEN ACCEPTABILITY
The design of the filtration cells provide the opportunity to check the quality or acceptability of and measure the plasma volume. Bio/Data Corporation has developed an integral subsystem and software to perform these functions within the filtration cycle time. This function is called the Compromised Specimen Alert System.
Plasma is optically scanned for hemolysis, icterus and lipemia. Clots are detected through the flow control system. This compromised specimen alert system notifies the operator when specimens are not acceptable for analysis.
Additionally, the volume of plasma separated can be precisely measured or simply monitored.
MICROFILTRATION TECHNOLOGY SUMMARY
Microfiltration Technology is the alternative to specimen preparation by centrifugation.
INNOVATIVE: The first significant improvement since the centrifuge.
VALIDATED: Bio/Data Corporation has used Microfiltration Technology in its FDA licensed instrumentation since 1985.
RAPID: Filtration and compromised sample check completed in less than 30 seconds.
SIMPLE: Only four moving parts; no complex robotics required.
FLEXIBLE: Technology can be used in specimen preparation, point of care, analytical, automation, therapeutic and biological applications.
SERIAL WORKFLOW: Continuously accepts and processes samples, eliminating batching.
EFFICIENT: Pre-analytical specimen handling reduced to four steps.
COST EFFECTIVE: Single disposable replaces multiple items required for centrifugation separation and required sample transfer. Time saving 0.55 minutes / sample.
AVAILABLE: Refer to licensing program section for additional information.
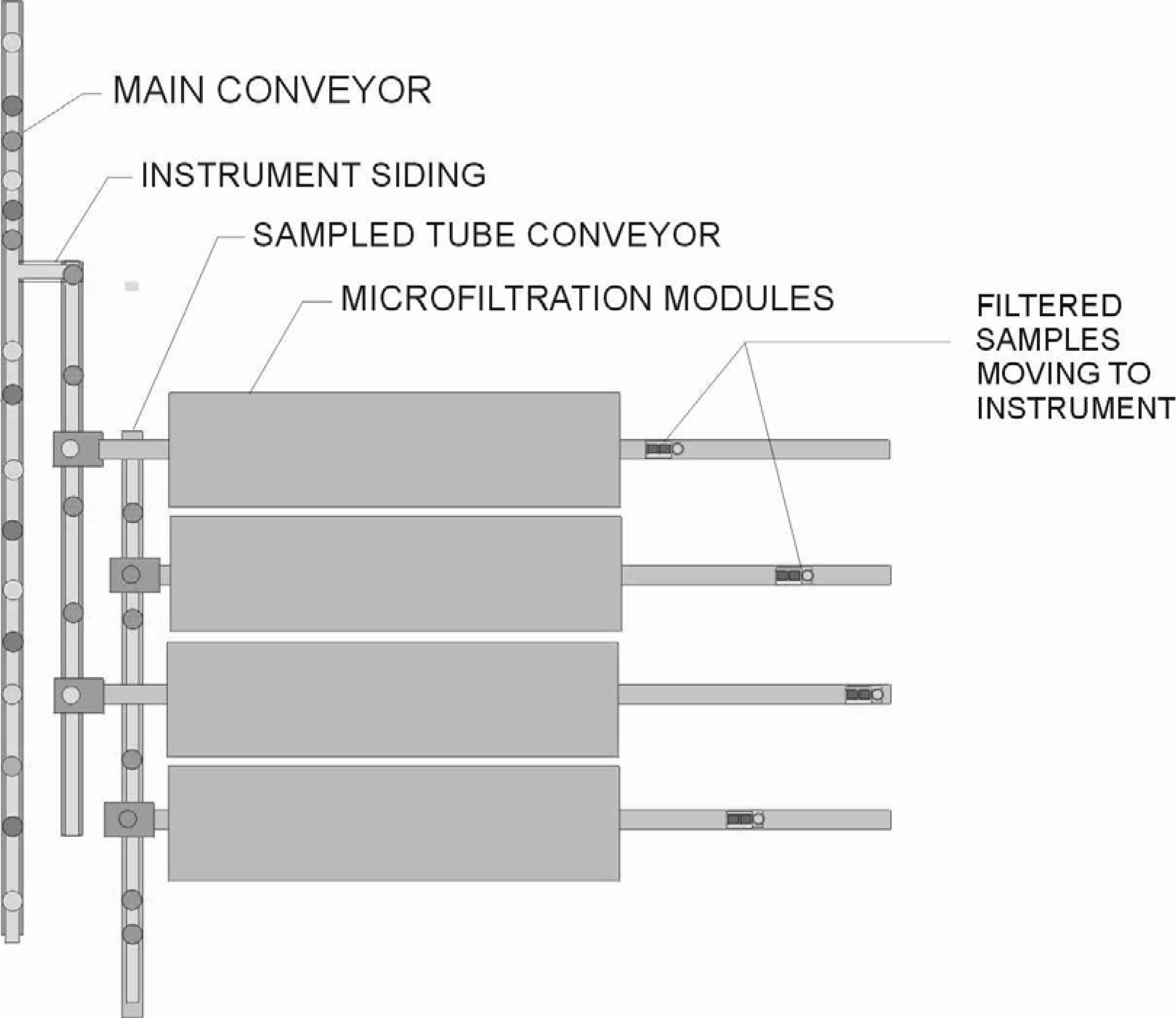
MICROFILTRATION SYSTEM
The primary components of a microfiltration system are:
Filtration Station
Optical Systems
System and Control Software
Disposable Filtration Cells
A microfiltration system can be used in conjunction with automated, cap piercing or non-automated sample transfer systems. The small foot print of a microfiltration station allows for very flexible applications. A microfiltration system can be:
Bench Top (accessory)
Integral to an Analyzer
Incorporated in a Workcell
Microsample Coagulation Analyzer™ Model MCA 310

Microfiltration Sampling System
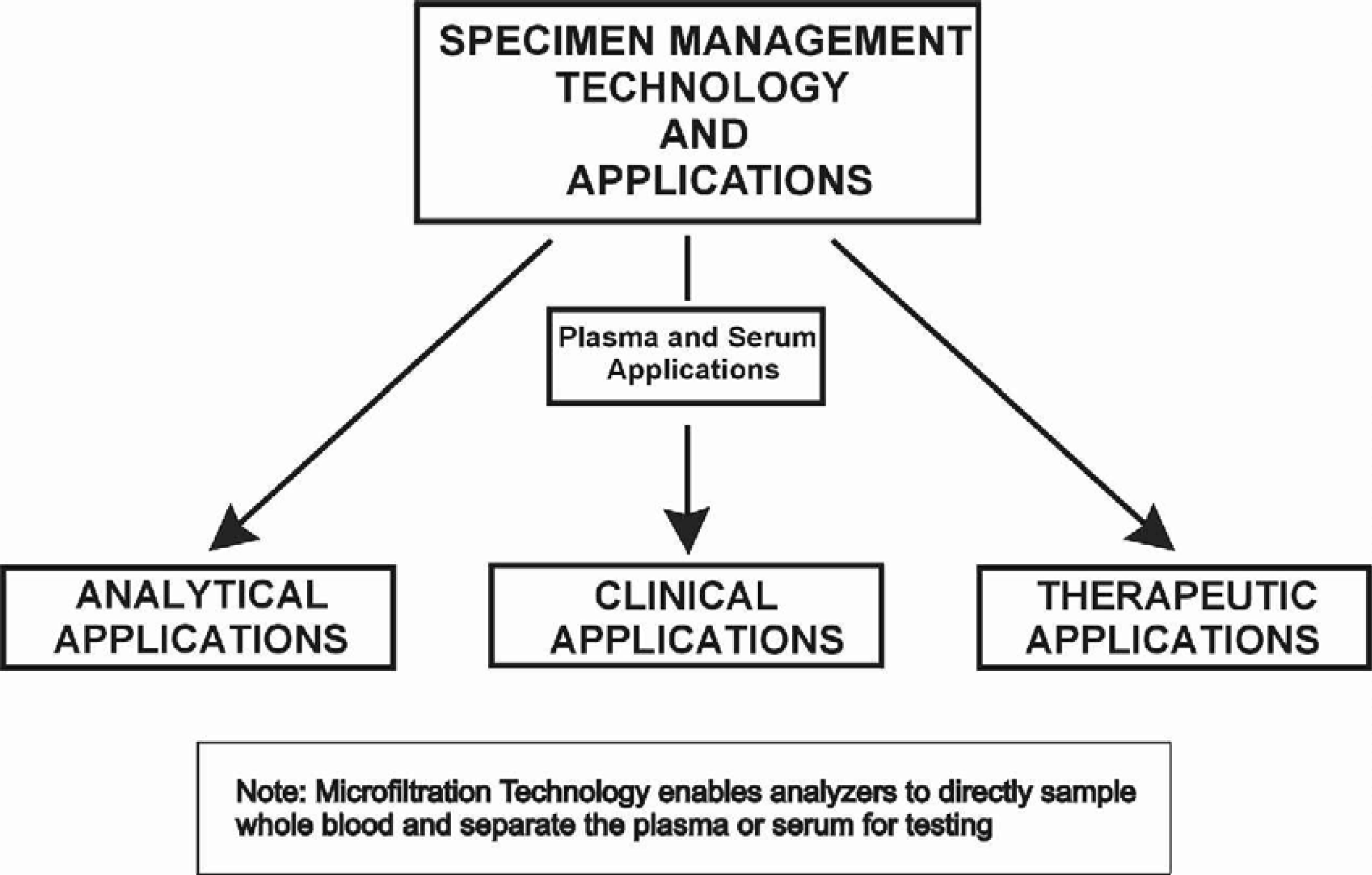
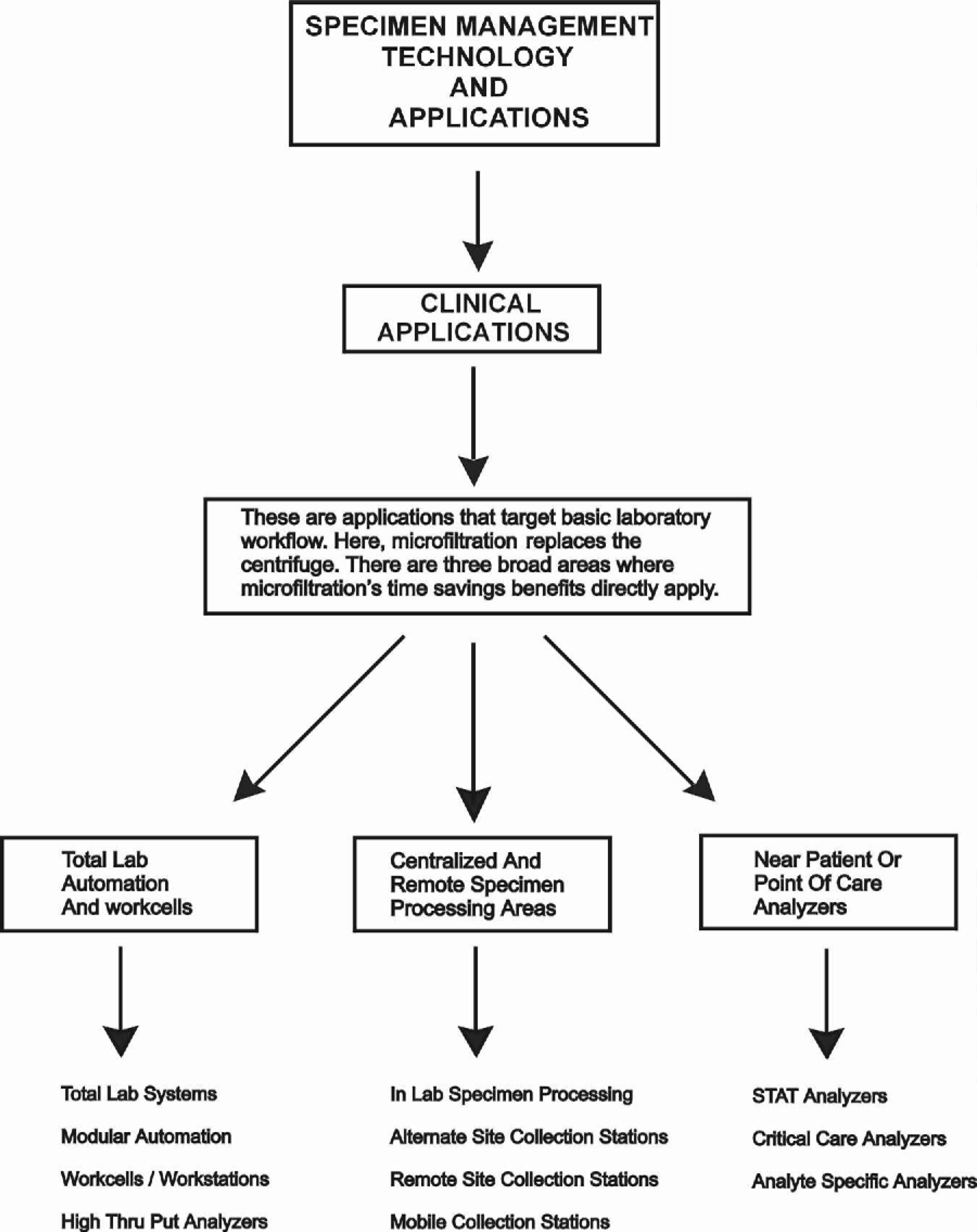
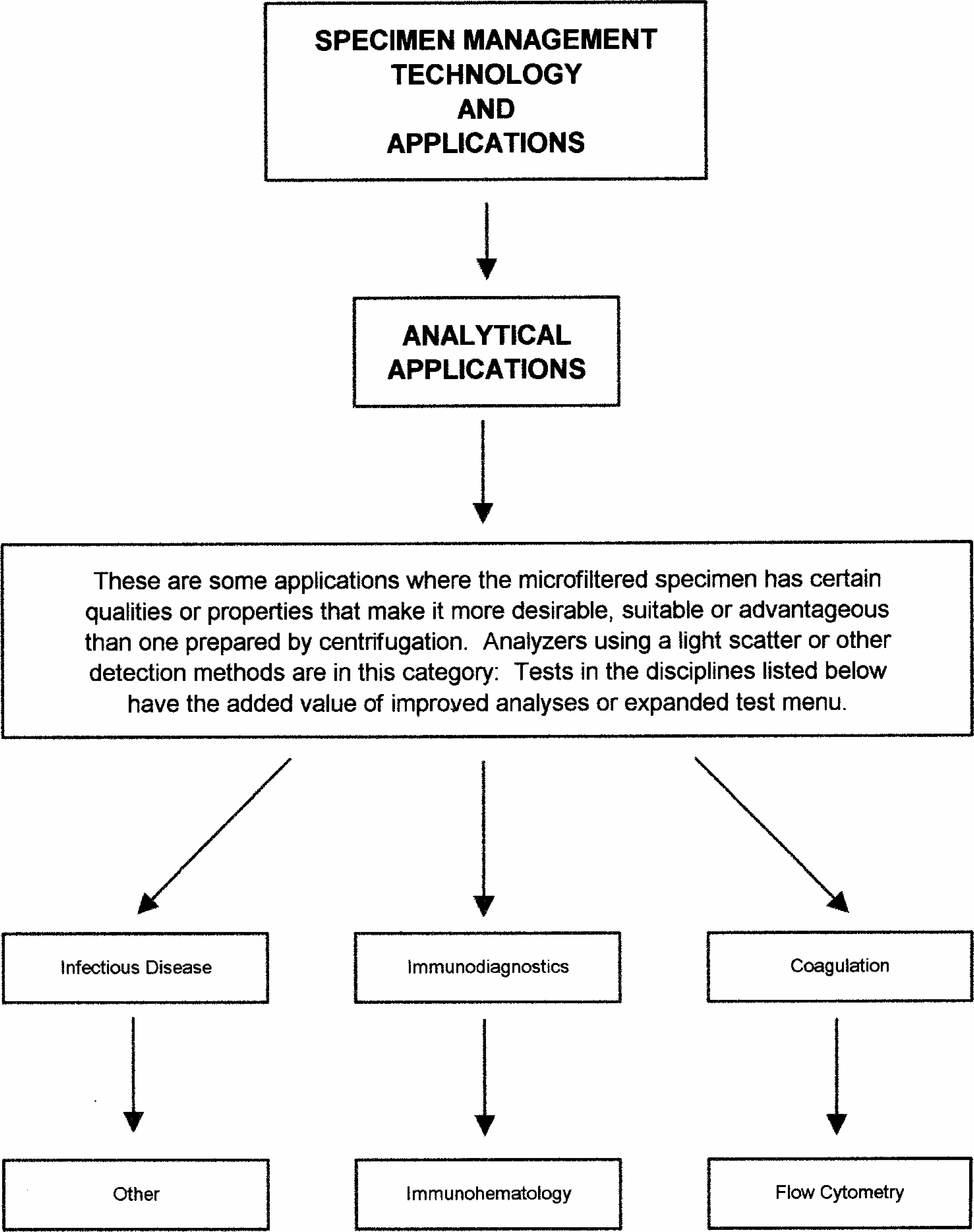
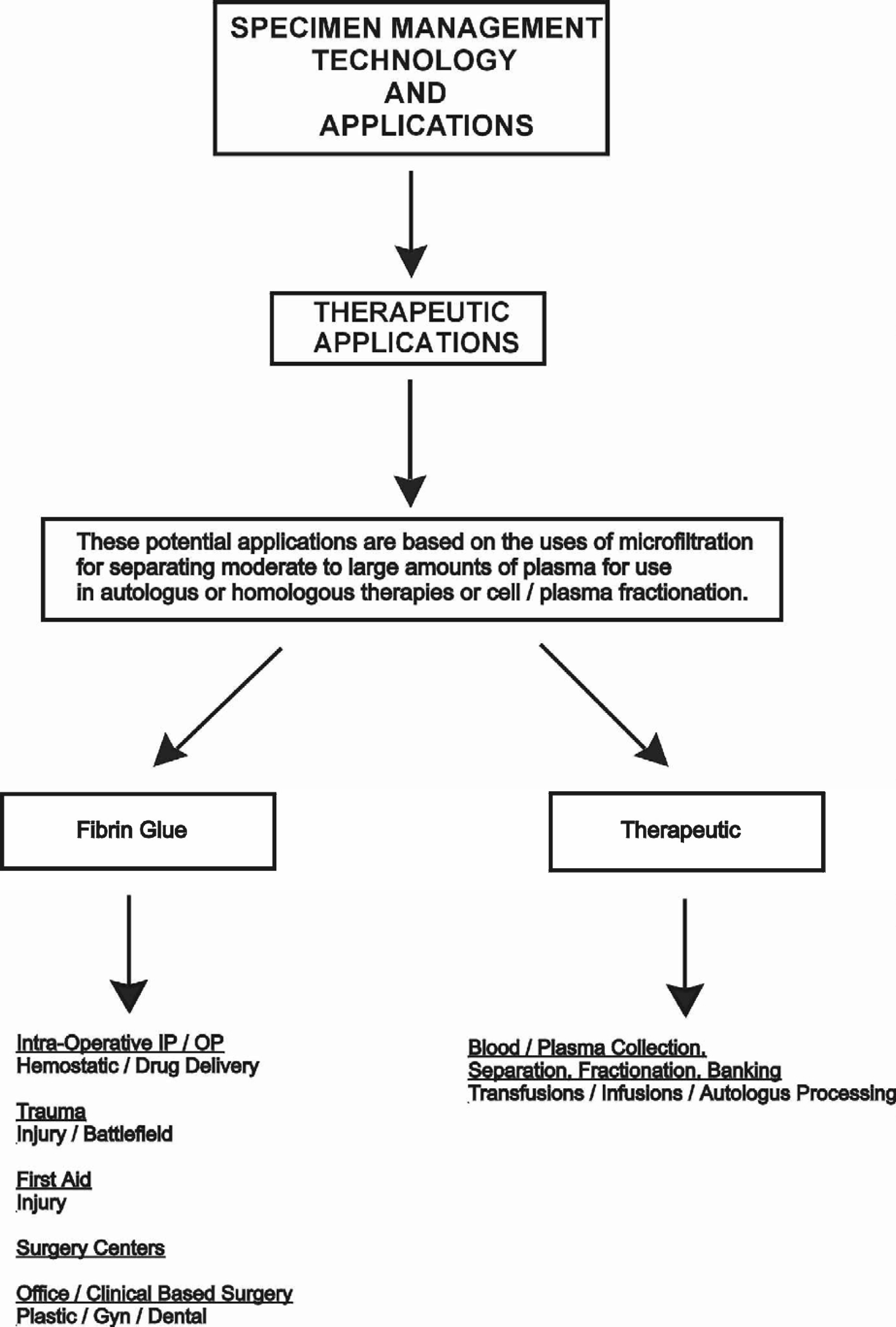
POTENTIAL APPLICATIONS FOR MICROFILTRATION TECHNOLOGY
This technology can be applied in three distinct areas:
Analytical
Clinical
Therapeutic
Within each of these areas, there are a number of individual applications.
The following charts outline selected application areas currently being developed.
MICROFILTRATION EVALUATION PROGRAM
THE PROGRAM:
Companies wishing to qualify microfiltration technology for a specified purpose can do so through this program.
Provided Materials:
Operations Guide
Microfiltration Evaluation Module
Software Communications Protocol
PC Software
Fifty (50) General Purpose Filtration Cells *
Fifty (50) Tap Vials
Direct Business Hours Access To A Product Development Engineer
REQUIRED INFORMATION:
Power Requirements must be specified on your Purchase Order.
TERMS & CONDITIONS:
An executed Non Disclosure Agreement is required.
Application description and a summary of test results must be provided to Bio/Data Corporation.
Agreed payment must accompany your order. Outgoing freight and insurance will be prepaid and invoiced upon shipment.
On site support by a Bio/Data Corporation engineer is available.
The module must be returned with freight and insurance prepaid at the end of the evaluation period.
LICENSING PROGRAM
Bio/Data Corporation has successfully developed and applied microfiltration technology for the rapid separation of biological samples since 1985. It has developed a body of intellectual properties including multi-national-patents, know-how and expertise in the practices and applications of microfiltration.
Microfiltration Technology
Software
Compromised Sample Alert System
Sample Transfer System
Automated Assembly Equipment
Licenses are only granted on a selective basis. Each licensee will be obliged to take a basic license. Content, limitations, and other terms and conditions will then be negotiated depending on the requirements of the license.
In addition to these licenses, the Company can provide:
Technical Support and Assistance
Application Development
Engineering Services
Contract Manufacturing: Hardware / Disposables
Automated Disposable Cell Assembly Equipment
A Microfiltration Evaluation Module and a supply of general purpose filtration cells are available, for qualification and development projects. A fee is charged for the Evaluation Module.
Footnotes
*
Please Note: Additional filtration cells may be purchased if required.
Acknowledgements
If you are interested in seeing more articles like this in JALA, please email
