Abstract
This presentation provides a concrete example of an application developed using the component-based framework [1]. A sample inventory and control system was developed for the purpose of aliquoting, distributing and tracking biologic molecules in pharmaceutical drug development. This presentation will provide an overview of the workflow involved, a description of the detailed functional implementation of the robotic aliquoting system, and a demonstration of the application with a real world implementation of the component-based framework.
This paper formed part of the Proceedings of the International Symposium on Laboratory Automation and Robotics (ISLAR), 1998. Reproduced with the permission of ISLAR (for further information, see: www.islar.com).
INTRODUCTION
Sample analysis for drug substance and drug product is addressed by a gamut of precise analytical procedures in Analytical Research and Development (AR&D) at Bristol Myers Squibb. In order to facilitate these procedures, the department supports a well-defined sample distribution process. This process has evolved as a consequence of several refinements instituted over the past few years. In order to address increasing sample load, it was decided to examine the distribution process to identify bottlenecks. Workflow modeling was used to document the process. After a careful analysis of the sample distribution process, several work activities were identified that were amenable for automation. The component-based framework described earlier [1], [2], was used to engineer, design, and develop an automated SDS. This paper provides an overview of the System, the rationale for automating the exiting manual processes and a description of the components of the system with emphasis on its implementation.
Sample analysis is dependent on the scientists having the appropriate materials and analysis information. The process of sample distribution is mission critical to the efficient operation of an analytical department. Poor sample distribution can cause unwanted delays, confusion, and poor work efficiency. It is for these reasons, AR&D has thoroughly defined its sample distribution process by evaluating its changing needs with the impact of new technologies in automation and information management. Currently, this distribution process involves the following steps: a batch of a specific lot of a drug product/substance is submitted for analysis, this sample is distributed into smaller aliquots and entered into an inventory control and task management system, the scientist responsible for each analysis is then notified of the sample analysis request. After the completion of analysis, the scientist enters the results into a Laboratory Information Management System (LIMS) to be approved and released to the client departments. In order to understand the process and to identify the rate limiting steps, it was necessary to model the process.
WORKFLOW ANALYSIS
Business processes can be broadly classified into two major categories. At one end of the spectrum are people-centric business processes that are largely defined by the users involved in the operation. Completion of the people-centric processes, which are generally project-oriented, is dependent on the involvement of a “knowledgable” worker(s). The methodology employed for the entire process, as well as for the tasks within each activity, is up to the discretion of the individual(s). Participants rely on a variety of information to perform tasks, and there can be little uniformity or predictability to the kinds of information required or the order the information will be reviewed. Other characteristics of people-driven business processes are relatively indeterminate processing cycle times and limited management accountability [3]. The success of the process is judged on the basis of tangible and intangible factors that are, again, very dependent on the individuals involved.
At the opposite end of the spectrum there are process-centric procedures, driven more by documents than by individuals or information. These processes are typically transaction-oriented and represent mission-critical business processes that are central to the firm's daily operations. Process-centric operations are very predictable: there are rigid rules for distribution, prioritization, and work completion. Even the way exceptions are handled is highly structured. These production-oriented operations tend to have short, fixed processing cycles and rigid deadlines. In addition, performance measurements for these processes are objective and quantifiable.
The sample distribution process was modeled according to the above classifications. People-centric tasks in the context of workflow are task assignment to specific resources, problem solving, project management, etc. However, tasks such as sample receipt and log-in, sample distribution and storage, analyst notification of workload, report generation, etc., are more process oriented. In order to enhance productivity, it was necessary to identify process-centric workflow and reduce or eliminate bottlenecks so that the flow of work can proceed in an efficient manner. The flow of work in the sample distribution process was modeled with the intent to identify process-centric tasks and institute procedures that eliminate redundancy and bottlenecks in operations that can be better served through automation. The result of this undertaking is depicted schematically in Figure 1.
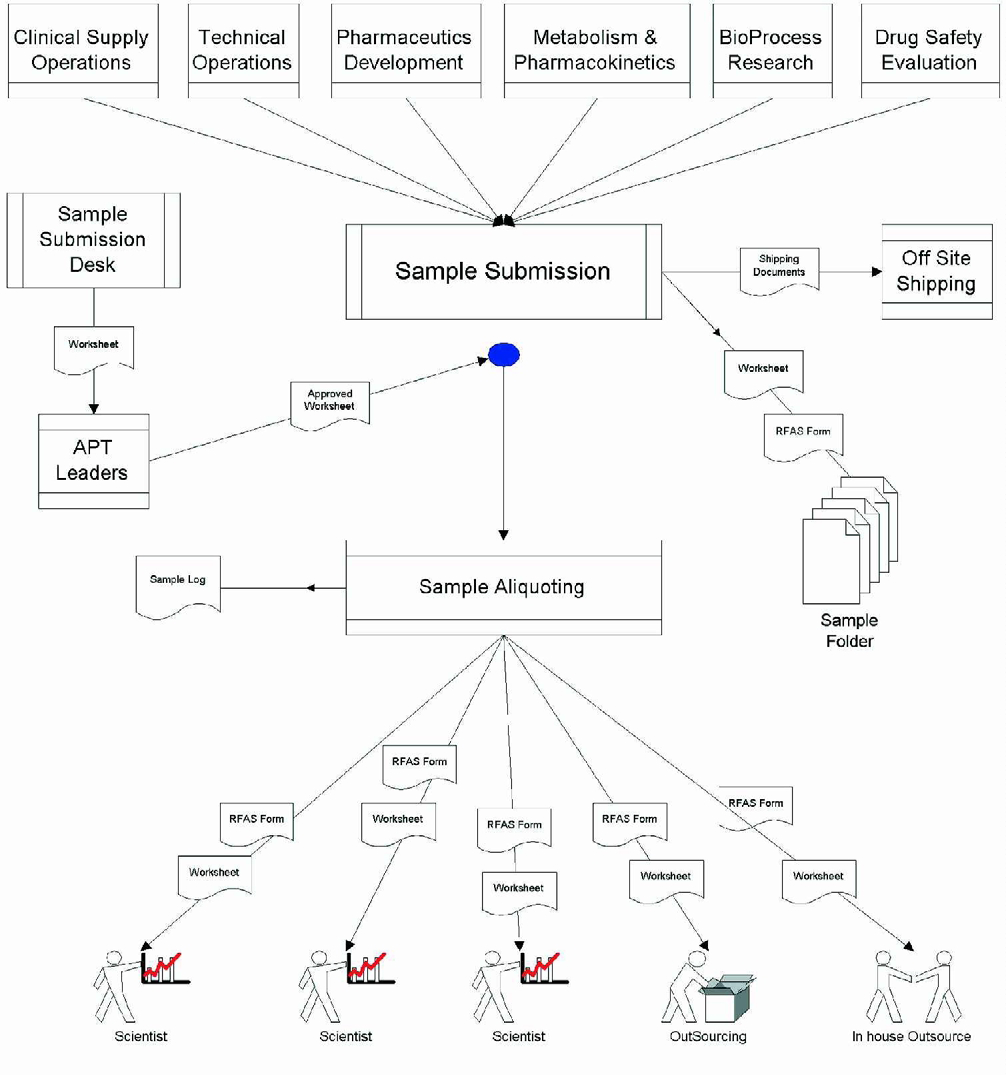
Current Sample Distribution Workflow
Figure 1 shows the flow of work in Analytical Research & Development as samples are submitted for analyses by our client/collaborators (i.e., Pharmaceutics R&D, Technical Operations, Clinical Supply Operations, Metabolism and Pharmacokinetics, Bioprocess Research and Drug Safety Evaluation). The current procedure is modeled on a centralized sample submission mechanism. In order to provide efficient flow of information and to aid quick decision making, this centralized mechanism has evolved over the past few years. This figure shows the process of submission of the sample to a centralized sample submission area.
A Request for Analysis form is created for each sample submitted for analysis. A worksheet containing the analyses specific tests is attached to the Request for Analyses form and submitted to the Analytical Project Team Leader for approval. Once approved, the sample is aliquoted into the appropriate smaller volumes and stored. A copy of the Request for Analysis form along with a copy of the approved worksheet is sent to each analyst responsible for performing a particular test on the sample.
A more in depth view of the sample aliquoting process is shown in Figure 2. The aliquoting process is comprised of several steps. These include the preparation of the source vial, the preparation of the destination vials (which includes the creation and application of vial labels), aliquoting the sample from source to destination vials, quick sealing and storing of the destination vials into the appropriate environment. Throughout each of these steps, environment controls (i.e., temperature, humidity, and sterility) are critical. A careful analysis of the model identifies two rate limiting steps; they are, the creation and application of the labels and the procedure of aliquoting of the sample (which includes uncapping vials, aliquoting the sample, recapping and storing the vials).
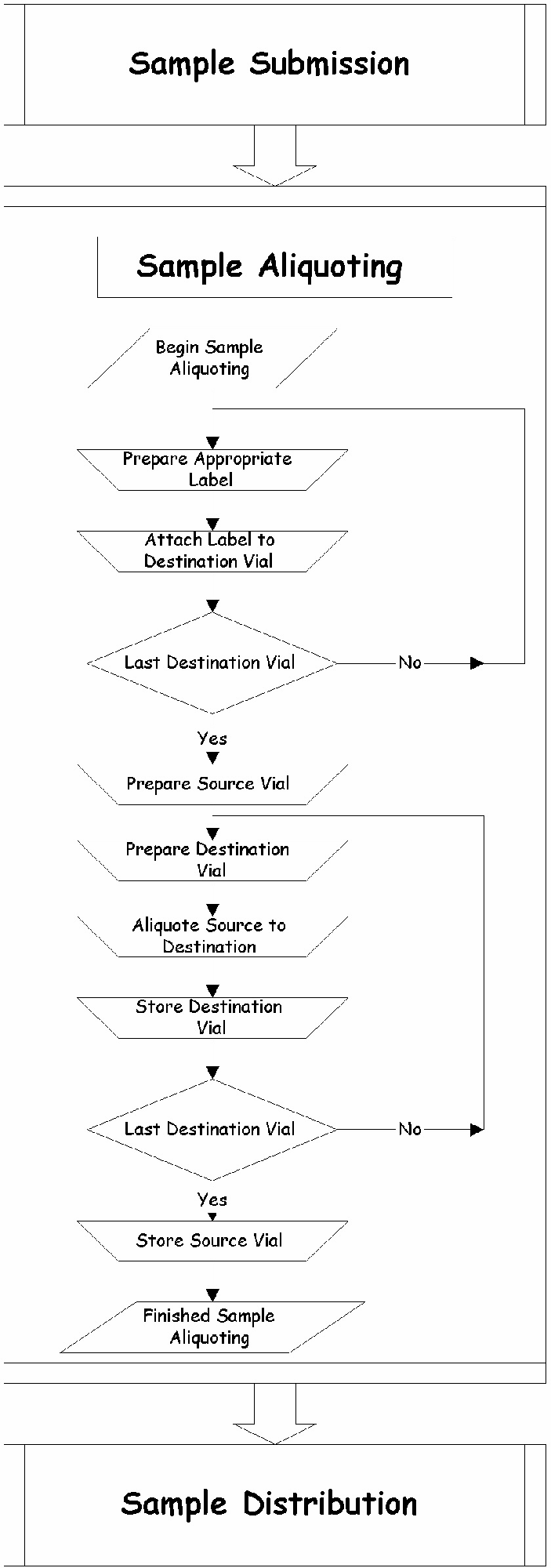
The Sample Aliquoting Process
Each batch of a drug substance or product requires a variable number of combinations of aliquot volumes and destination vials. The number of iterations for label creation and application increases significantly with increasing samples that require thousands of aliquots per sample. This was a people centric task, it is truly a process centric task. Until recently, there has been no other way to perform this extremely redundant yet well defined process.
The analysis of the sample distribution process involving people and process centric tasks lead to the elimination of the process centric rate limiting factors. The result was a system design that combines robotics, the world-wide-web for inventory control and sample status information management, and backend database technologies used to maintain and organize information. More specifically the process identified a robotic system for sample aliquoting (labeling, preparing, and storing vials), a system front end for the robotic system and for sample submission, and facilities for report generation, compound status, and other specific queries, all of which are supported by a centralized enterprise level database. This Sample Distribution System removes the bottle necks by applying appropriate technologies and automation tools to assist in making the process more efficient.
SYSTEM OVERVIEW
As a result of the modeling process, several bottlenecks were identified as amenable to automation. The sample distribution process with the proposed automation modifications is depicted in Figure 3.
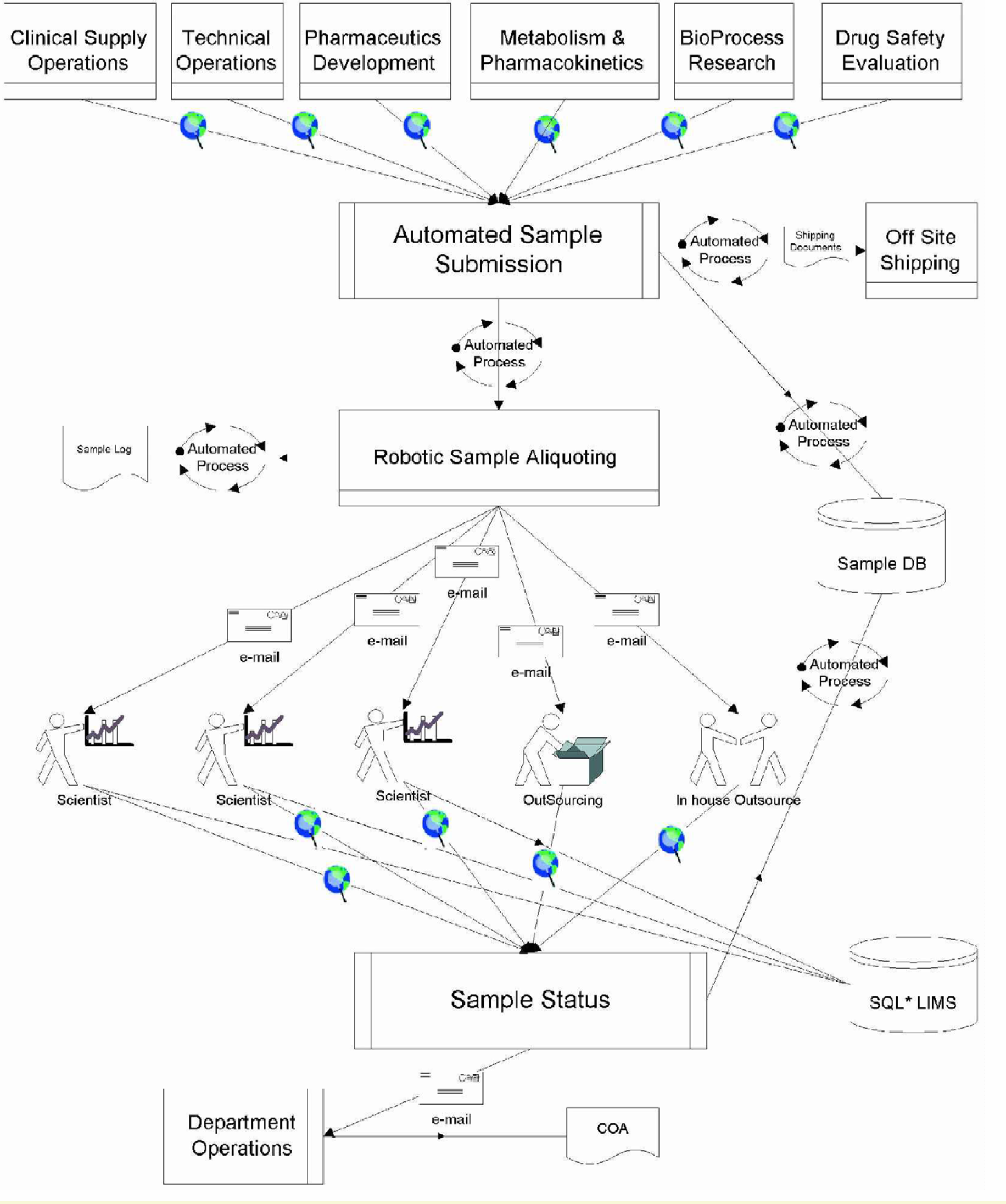
The Automated Sample Distribution Process
Essentially, the process was envisioned to include an automated robotic aliquoting system (RAS), an automated sample tracking an inventory control system (STS), and an electronic notification system (ENS). These three subsystems are schematically depicted in Figure 4.
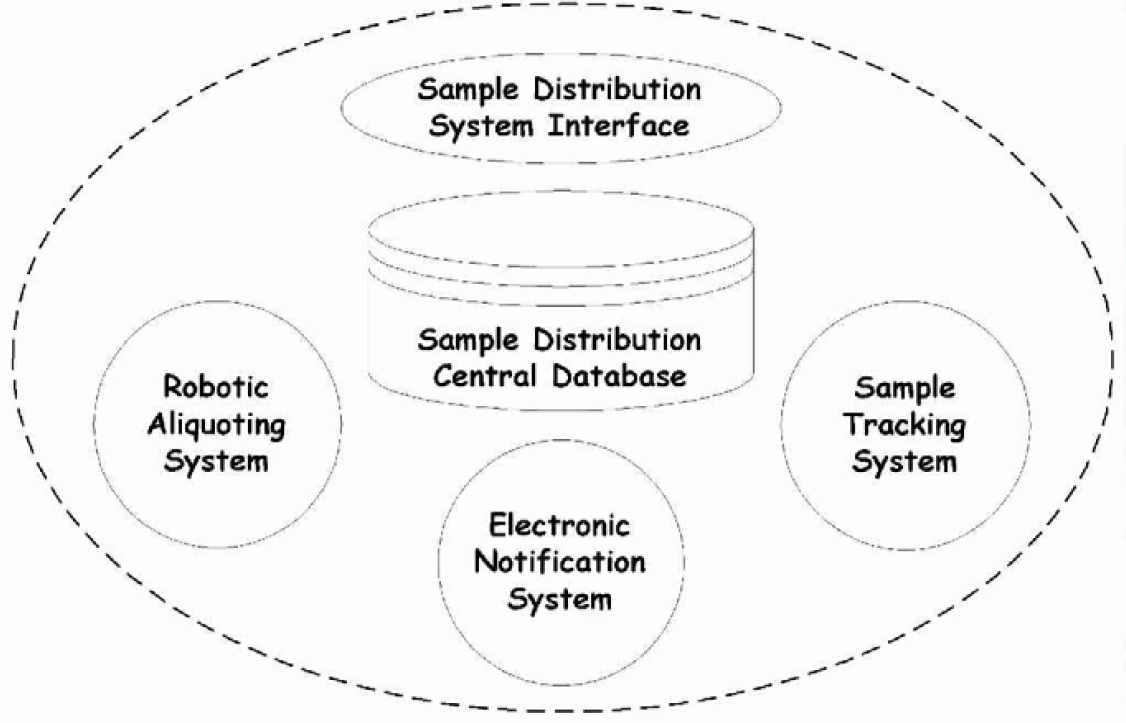
Functional Subsystems of Sample Distribution System
In order to carry the project from the vision to the implementation a team was formed, with representatives from the management, scientists familiar with the distribution process, and engineers and programmers from an in-house automation team. In order to remain project minded and progressive the team established a system life cycle and developed system requirements from which specifications were developed using the Unified Modeling Language (UML).
The seamless integration of information management provides one multitasking and multidimensional interface on the web that the clients/collaborators can use to enter sample information and query for status of analysis on submitted samples. The unified interface provided through the world-wide-web would allow for consistent sample information entry, inventory control, and sample status inquiries by both the department personnel and its collaborators.
Upon receipt of the sample, it is placed as input into the Robotic Aliquoting System (RAS). This subsystem creates and attaches labels to destination vials, aliquotes the sample, and places the destination vials into the appropriate storage. RAS acquires all of its sample information for labeling (compound name, date aliquoted, date received, lot number, freezer number, and concentration) and aliquoting (volumes, numbers) from the centralized database supported by the STS.
This minimizes redundancy of data entry and errors in operator input. Upon completion of the sample aliquoting procedure, the system updates the centralized database and generates a notification ENS (i.e., e-mail) regarding the requests for analysis to the respective scientists. Administrative level support for project management is attained through multiple security levels. This allows an authorized individual to perform a variety of queries over the web, including information about outstanding samples, date received, date aliquoted, compound name, or any other field in the database.
In order to incorporate the system and its components quickly into the operating culture, a prioritized development approach was used. In order to achieve seamless systems integration of the constituent components, it is necessary to have effective project management. This minimizes down time for the system, which results in maintaining a high level of productivity. The challenge is to balance the development of the system taking into account the use of already developed components toward mission critical processes. Figure 5, shows a component development and synchronization plan for the SDS. In this figure, each box illustrates a milestone of a usable component. It shows how from the project proceeded from concept to specifications, then to the parallel development of RAS and STS. The synchronization of these two subsystems is followed by the integration of the ENS.
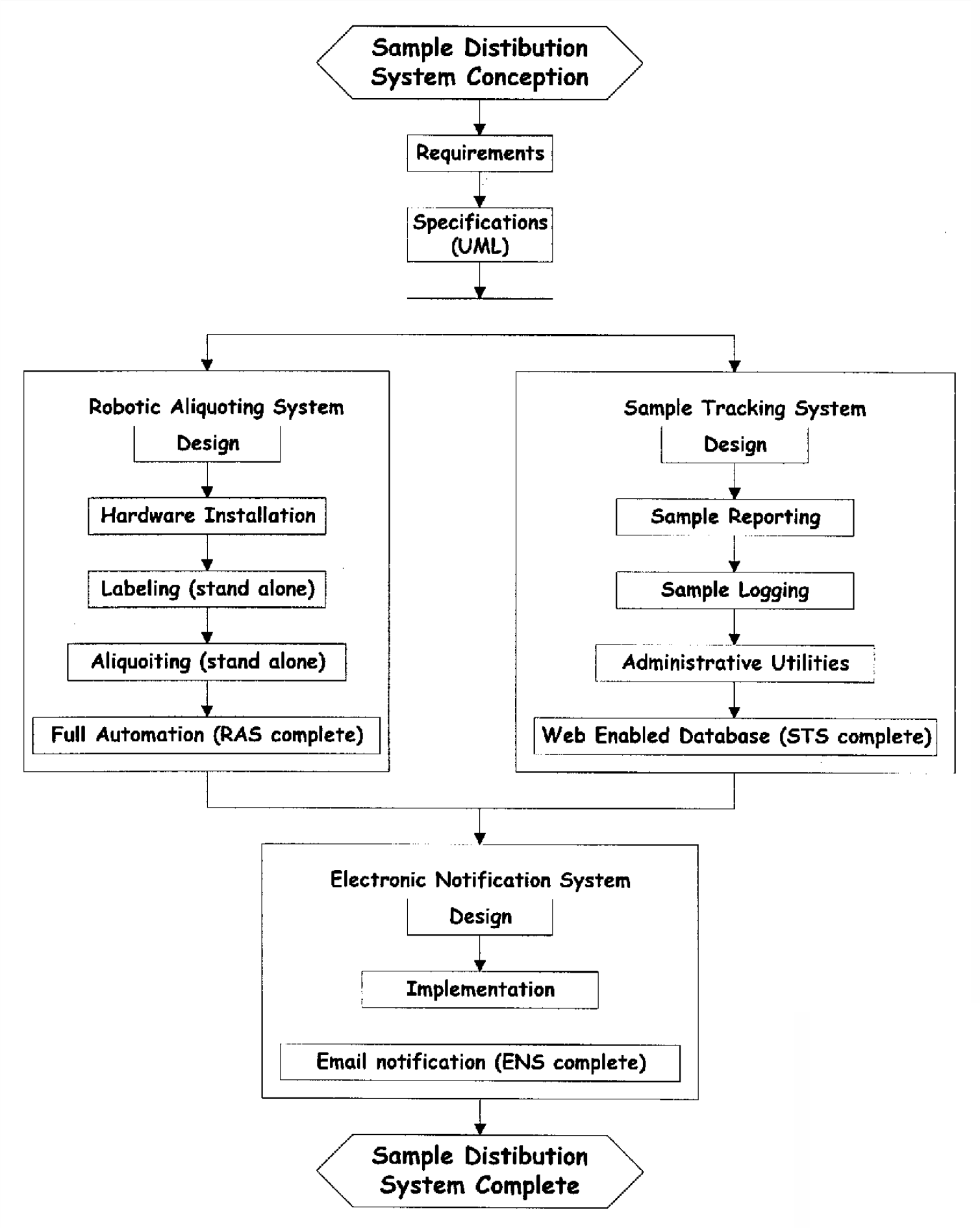
Component Development and Synchronization Plan for the SDS
The following discussion will use the RAS subsystem to illustrate the rationale for its design. The RAS is responsible for the aliquoting process (see Figure 2 for additional detail). The relationship between the components within this process is depicted in Figure 6.
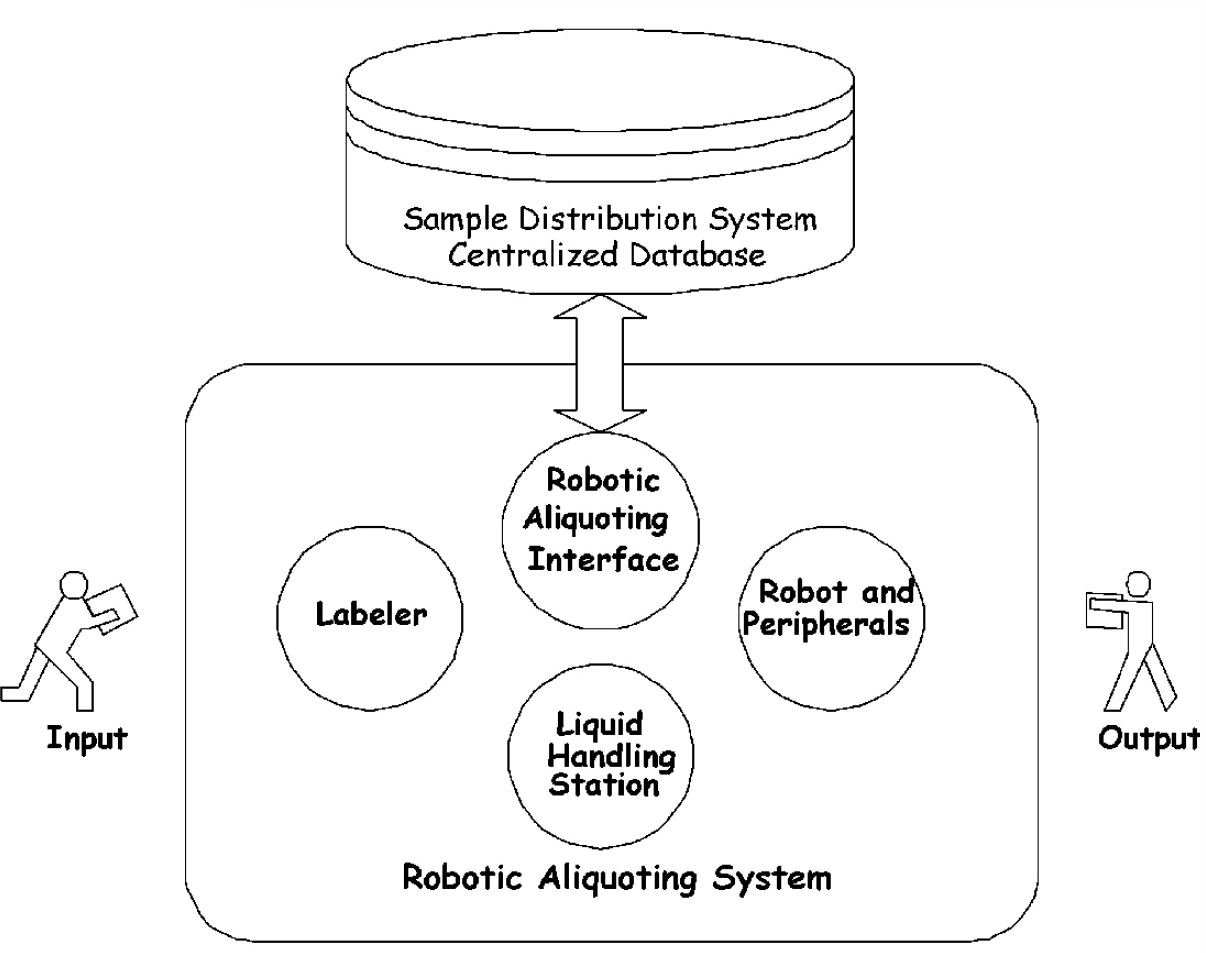
Overview of the Robotic Aliquoting System
The two most time consuming sub-processes involved with RAS are labeling of vials and aliquoting the sample into the vials. Of these two rate limiting factors, it was deemed that automating the labeling process would be of the most value, since this process was the most mundane and time consuming.
The equipment needed to perform the labeling was a Zymate XP Robot, a Zymate Carousel, and a Biomedical Devices (BDC) Bar Code Labeler. The design and integration of the Zymate peripherals (the robot and the Carousel) was addressed through the Zymate OLE Control. However, integration of the Bar Code Labeler into the labeling process involved the coordination of the functionality supported by the labeler with the Zymate peripherals. A batch scheduler was used to create task-lists to coordinate these multi-vendor peripheral components and to execute the Laboratory Unit Operations supported by them.
Consequently, to perform sample aliquoting, the coordination of the functionality supported by the Liquid Handling Station with the Zymate Peripherals (XP Robot, the carousel, the capper and the linear shaker) and the −20°C Freezer was addressed. Finally, these two capabilities were integrated to provide full automation capabilities of the system.
This approach to systems development and integration provides for increased productivity by allowing complete use of already developed components in semi-automated/workstation fashion while the system is being integrated. This is an example of the availability strategy [2] where the system is designed to be fault tolerant in cases where a particular component malfunctions and has to be taken off-line.
DISCUSSION
A specific architecture design was followed throughout the development of SDS. The following discussion addresses these architectural design considerations [2] and their impact on the design and implementation of the Sample Distribution System.
Workstation mode allows the user to use any hardware present on the system as a stand-alone workstation. Semi-automation mode allows the user to use specific functionality of the system, for instance, to create and apply a batch of labels to the destination vials. Full Automation allows the user to take advantage of complete functionality (Labeling and Aliquoting) of the system. The use case for the user services design of full automation is shown in Figure 7 Tools provided by UML such as system interactions and use case diagrams provided excellent modeling opportunities to design and develop the user services for the SDS.
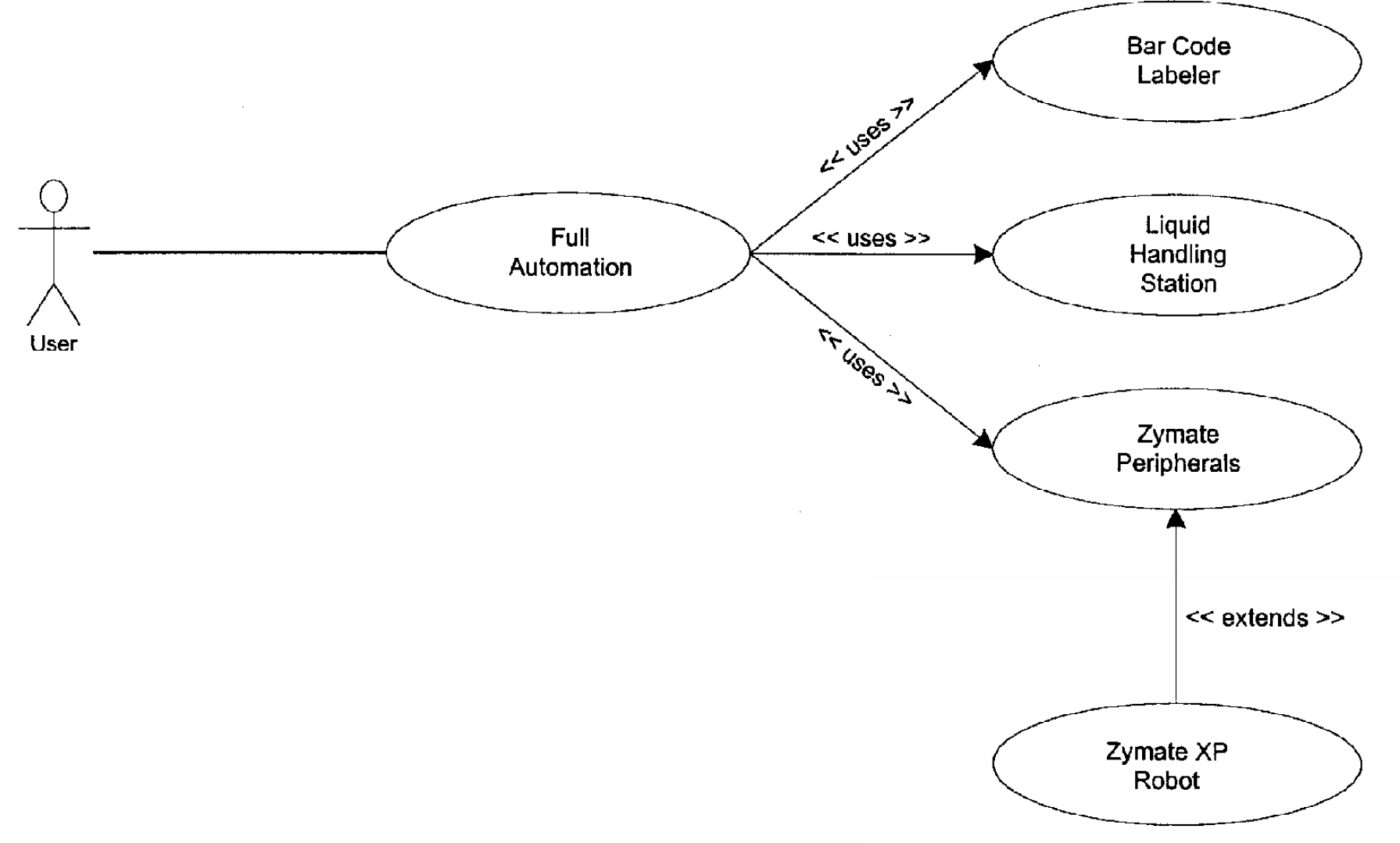
Overview of the Robotic Aliquoting System
CONCLUSION
The Sample Distribution System demonstrates how automation and scientific expertise can be exercised by selectively choosing processes amenable to automation. These processes were identified by means of thorough work flow modeling. The Sample Distribution System is an example of the integration of advanced technologies tailored to meet scientific needs, in a scalable fashion. It demonstrates how automation can assist with workflow and provide useful project management and scheduling tools. It also shows how analysis of a process can be performed and how analysis techniques can be useful during the prioritization of development.
To address decreasing timelines for drug development and changing needs of drug development processes, one strategy is to take advantage of existing and novel technologies in laboratory automation. It is advantageous to decrease the development time for an automated system by reforming the development strategy into usable multifunctional component layers that are strategically prioritized and initiated on the way to building a scalable system.
