Abstract

A 3-month-old male kitten had a history of dysuria, stranguria and episodic urethral obstruction. At referral, the penile urethral meatus was not patent and, when the bladder was compressed, urine leaked from a fistula in the scrotum. A narrow-necked urethral diverticulum in the region of the scrotum was demonstrated with voiding, positive contrast cysto-urethrography. Whilst it was not possible to classify this anomaly with certainty, a congenital urethral diverticulum with fistulation to the scrotal surface was considered the most likely diagnosis.
Case Report
A 7-week-old male domestic shorthair kitten presented to the referring veterinarian with haematuria. The owners commented that it also leaked urine whenever it was picked up. A palpably thickened bladder was the only abnormality noted on physical examination and the kitten was prescribed amoxycillin-clavulanate (Clavulox; Pfizer; 25 mg twice daily orally). The next day constant straining was observed but only small drops of urine were voided. The kitten was anaesthetized and the penile urethra catheterized with a 24 gauge intravenous catheter. Thirty mililitres of lactated Ringer's solution were instilled through the catheter and bladder compression after catheter removal resulted in a good urine stream. A single dose of dexamethasone sodium phosphate (Dexadreson; Intervet Australia; 0.4 mg) was administered subcutaneously and amoxycillin-clavulanate was continued for 10 days. At recheck, the kitten was vaccinated against feline panleucopaenia, feline herpesvirus and feline calicivirus. The bladder was still palpably thickened so amoxycillin-clavulanate therapy was continued. Ten days later, the kitten was again presented with signs of urethral obstruction. Under general anaesthesia, the penile urethra was catheterized with difficulty and a ‘urethral fistula’ was noted between the testes. The kitten was observed to urinate through both the penis and the fistula over the next 2 days. A week later, it was referred to The University of Sydney Veterinary Centre (Sydney, Australia) for evaluation.
Physical examination was unremarkable except that a 1 mm diameter opening was observed in the scrotum between the testes. When the bladder was expressed manually, urine was voided from this opening and not from the penis.
The kitten was anaesthetized for contrast radiography and more detailed examination of the penis and scrotum. The penile urethral orifice was not discernible. Iohexol was injected percutaneously into the bladder. The bladder was then expressed manually to simulate voiding and fluoroscopy was used to evaluate the lower urinary tract. Urine passed from the bladder into the preprostatic urethra and then into a dilated intra-pelvic urethra. At the level of the ischial arch, the urine stream diverged. Urine passed into the terminally blocked penile urethra and also into the scrotal region where it pooled in a diverticulum before exiting through the small scrotal orifice (Figs 1 and 2).

Voiding positive contrast cysto-urethrogram. Note the dilated intra-pelvic urethra, the diverticulum at the scrotum and the thin stream of contrast agent in the distal penile urethra.
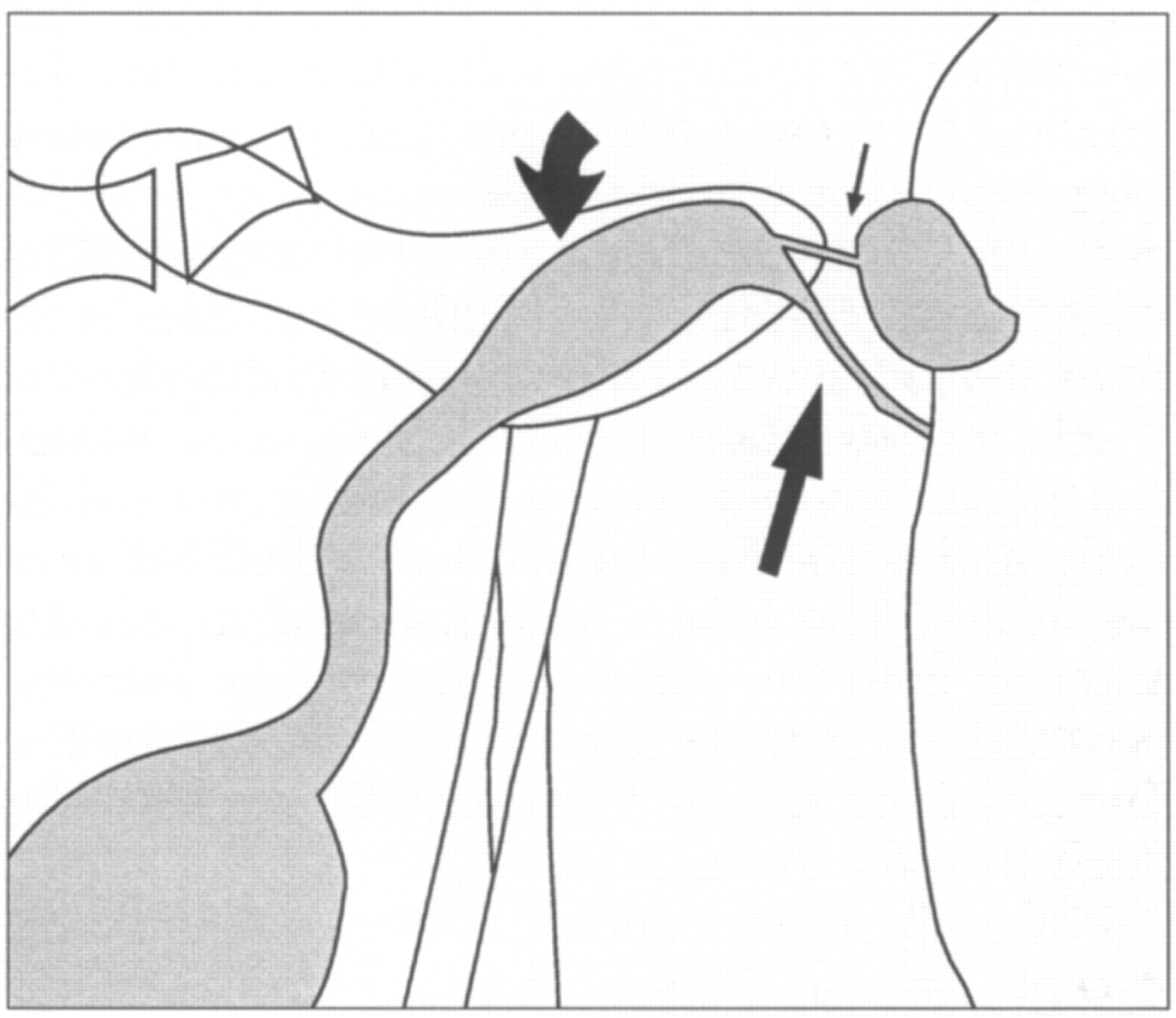
Diagrammatic representation of the voiding cysto-urethrogram. Urine passes from the bladder through the dilated intra-pelvic urethra (curved arrow) and into the penile urethra (long arrow). Urine also passes via a narrow tract (small arrow) into the urethral diverticulum within the scrotum.
Temporary relief of the urinary tract obstruction was provided by enlarging the exit portal for urine. A 4 mm incision was made in the scrotum and a mucosa-lined cavity was observed through the incision. Amoxycillin-clavulanate was continued (62.5 mg twice daily orally) and the kitten experienced no difficulty voiding urine through the enlarged scrotal orifice. Six days later, the perineal area was explored surgically and a large, mucosa-lined diverticulum communicating with the pelvic urethra via a narrow tract was visualized. No communication between this structure and the penile urethra was evident. A congenital narrow-necked urethral diverticulum with acquired scrotal fistulation was considered the most likely diagnosis. The kitten was castrated and a perineal urethrostomy performed. A further 2 weeks of amoxycillin-clavulanate were prescribed and the kitten recovered uneventfully and remains well at the time of writing (7 months after surgery).
Discussion
Congenital anomalies of the urethra occur infrequently in dogs and rarely in cats (Osborne et al 1972, Holt 1992, Krawiec 1995). The malformations described include urethral agenesis (Kruger et al 1996), urethral hypoplasia (Holt & Gibbs 1992), urethrorectal fistulas (Osborne 1977, Van Den Broek et al 1988) urethral duplication (Masetti et al 1985), hypospadias (Hoskins 1995) and ectopic urethra (Lulich et al 1987). No reports of congenital urethral diverticula in cats could be found in texts or review articles (Lulich et al 1987, Holt 1992, Hoskins 1995, Kruger et al 1996).
The male urethra is derived primarily from the urogenital sinus. The epithelial lining of the caudal portion of the urogenital sinus expands along the elongating genital tubercle as a solid cord, the urethral plate. The urethral plate then hollows to form a canal and proliferation of mesenchyme on both sides of the plate enlarges the urogenital folds and leads to the establishment of a median urethral groove on the ventral surface of the genital tubercle. The elongating genital tubercle develops into the penis and the urogenital folds fuse ventrally along the entire length of the genital tubercle establishing the penile urethra. Simultaneously, the prepuce develops as an epithelial invagination and covers the penis. The scrotal swellings which arise lateral to the urogenital folds, in the inguinal region, gradually move caudomedially and the caudal parts enlarge and fuse in the midline to form the scrotum and the scrotal raphe (Dennis 1979, Noden & de Lahunta 1985). The erectile tissue of the corpus spongiosum is formed by differentiation of the mesodermal tissues of the urethral folds and advances along the urethra as fusion of the urethral folds progresses (Dorairajan 1963). Any aberration in this complex sequence may result in malformation.
Urethral diverticula are tubular or sac-like dilations opening into the urethral canal through an ostium at any point on its course. Congenital urethral diverticula are lined by urethral mucosa and contain a striated muscle layer. Acquired diverticula result from trauma, suppuration, urethral calculus or urethral stricture and are lined by granulation and fibrous tissue; muscle fibres are absent (Maged 1965, Mohan et al 1980).
Congenital urethral diverticula are very rare malformations in human males (Maged 1965, Meiraz et al 1971, Freeny 1974, Mohan et al 1980, Dinneen & Duffy 1994). They are classified as saccular or diffuse. The most common type is a wide-mouthed, saccular diverticulum in the ventral urethra usually near the penoscrotal junction. Narrow-necked saccular diverticula are less common (Williams & Retik 1969, Colodny 1987, Bernstein & Churg 1992). Rarely, a diffuse urethral diverticulum or megalourethra is seen as a diffuse elongated expansion of the lumen which tapers to a normal diameter proximally and distally (Dorairajan 1963, Williams & Retik 1969, Meiraz et al 1971). Diffuse urethral expansion can be difficult to distinguish from a large saccular diverticulum and they may be variants of the same malformation (Dorairajan 1963).
The embryological basis for urethral diverticula in humans is unknown (Freeny 1974, Moffat 1990, Bernstein & Churg 1992). Hypotheses include defects in the corpus spongiosum, faulty closure of the urethral folds, persistence of embryonic para-urethral glands, distal urethral obstruction in the foetus and abortive urethral duplication (Johnson 1923, Dorairajan 1963, Sweetser 1967, Meiraz et al 1971, Moffat 1990, Bernstein & Churg 1992).
Clinically, males with congenital urethral diverticula usually present with a poor urine stream during attempted voiding and a soft mass at the penoscrotal junction. Involuntary dribbling of urine from the urethral meatus can occur, especially after voiding, with jerky movements or with pressure on the perineum (Maged 1965, Meiraz et al 1971, Mohan et al 1980, Kirks & Grossman 1981). If there is urinary stasis without obstruction, urinary tract infection is the most usual clinical manifestation (Kirks & Grossman 1981). Narrow-necked diverticula are not usually obstructive but stagnation of urine can lead to stone formation, urinary tract infection and fistulation to the skin surface (Mandler & Pool 1966, Maged 1966, Dinneen & Duffy 1994).
Historically, this kitten had demonstrated urinary incontinence when picked up by the owners, a sign consistent with the presence of a urethral diverticulum. The cause of the urethral obstruction and haematuria was not determined as urine sediment examination and culture were not performed. It is feasible that urine pooling in the urethral diverticulum predisposed the urinary tract to infection or to feline lower urinary tract disease (FLUTD) and led to the signs of dysuria, haematuria and ultimately urethral obstruction. The scrotal fistula was not noted during the first catheterization of this kitten by the referring veterinarian. This suggests that it was an acquired fistula, formed as a result of back-pressure secondary to obstruction of the penile urethra or as a result of manipulations performed to relieve episodes of urinary obstruction. Although the penile urethral meatus was originally reported to be patent, it was completely stenotic at the time of referral. The combination of catheterization trauma and inadequate urine stream were most likely responsible for the stenosis.
As no histopathology was performed, it is not possible to state with certainty that the diverticulum was congenital. However, despite the prevalence of urethral obstruction in cats, acquired diverticula have not been reported in this species so it is unlikely that FLUTD in a young kitten would result in this abnormality. The gross appearance of a smooth, glistening, diverticulum lining, more typical of mucosa than granulation tissue, also supported a congenital aetiology.
Congenital urethral diverticulum with an acquired scrotal fistula was the most likely diagnosis in this kitten. If, however, the fistula was congenital rather than acquired, then the condition would have been classified as urethral duplication, possibly similar to the Y-type in humans. A Y-duplication is an incomplete urethral duplication in which the urethra splits to form an accessory channel opening to the perineum or anus (Williams & Bloomberg 1976, Colodny 1987, Bernstein & Churg 1992). This type of duplication is often associated with stenosis and hypoplasia of the normal penile urethra, a factor that could contribute to the complete stenosis of the urethral meatus (Williams & Bloomberg 1976, Bernstein & Churg 1992). If the malformation represented urethral duplication, the diverticulum observed in the scrotum would be best described as a diffuse urethral diverticulum or megalourethra.
Whilst congenital urethral abnormalities in the cat are undoubtedly rare, lack of recognition may mask their true incidence. Despite the owners having noticed urinary incontinence, the kitten was not presented to a veterinarian until dysuria occurred and it is unlikely that dynamic contrast studies would have been performed had it not become obstructed and developed a scrotal fistula.
Footnotes
Acknowledgements
We thank Dr Rhondda Canfield and Dr Jenny Charles for their contributions to the manuscript. Susan Foster is a Lionel Lonsdale Clinical Fellow. Richard Malik is supported by the Valentine Charlton Bequest of the Post Graduate Foundation in Veterinary Science of The University of Sydney.
